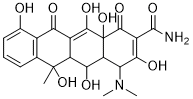
Oxytetracycline (Lenocycline; Oxyterracine; Oxyterracyne; Tarosin; Oxymykoin) is a naturally occurring tetracycline class of antibiotic extracted from the actinomycete STREPTOMYCES rimosus with a broad-spectrum of antibacterial effects. It has been used in a wide variety of clinical conditions for Gram-negative and Gram-positive bacteria. Oxytetracycline works by interfering with the ability of bacteria to produce essential proteins. Without these proteins, the bacteria cannot grow, multiply and increase in numbers. Oxytetracycline therefore stops the spread of the infection and the remaining bacteria are killed by the immune system or eventually die. Oxytetracycline is a broad-spectrum antibiotic, active against a wide variety of bacteria. However, some strains of bacteria have developed resistance to this antibiotic, which has reduced its effectiveness for treating some types of infections.
Physicochemical Properties
| Exact Mass | 460.148 |
| Elemental Analysis | C, 57.39; H, 5.25; N, 6.08; O, 31.27 |
| CAS # | 79-57-2 |
| Related CAS # | Oxytetracycline hydrochloride;2058-46-0;Oxytetracycline dihydrate;6153-64-6;Oxytetracycline calcium;7179-50-2 |
| PubChem CID | 54675779 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.7±0.1 g/cm3 |
| Boiling Point | 727.8±60.0 °C at 760 mmHg |
| Melting Point | 183 °C |
| Flash Point | 394.0±32.9 °C |
| Vapour Pressure | 0.0±2.5 mmHg at 25°C |
| Index of Refraction | 1.762 |
| LogP | 0.56 |
| Hydrogen Bond Donor Count | 7 |
| Hydrogen Bond Acceptor Count | 10 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 33 |
| Complexity | 1000 |
| Defined Atom Stereocenter Count | 6 |
| SMILES | O[C@@]12C(=O)C(C(=O)N)=C(O)[C@@H](N(C)C)[C@@H]1[C@H]([C@@H]1[C@@](O)(C)C3C=CC=C(C=3C(=O)C1=C2O)O)O |
| InChi Key | OWFJMIVZYSDULZ-PXOLEDIWSA-N |
| InChi Code | InChI=1S/C22H24N2O9/c1-21(32)7-5-4-6-8(25)9(7)15(26)10-12(21)17(28)13-14(24(2)3)16(27)11(20(23)31)19(30)22(13,33)18(10)29/h4-6,12-14,17,25-26,28,30,32-33H,1-3H3,(H2,23,31)/t12-,13-,14+,17+,21-,22+/m1/s1 |
| Chemical Name | (4S,4aR,5S,5aR,6S,12aR)-4-(dimethylamino)-1,5,6,10,11,12a-hexahydroxy-6-methyl-3,12-dioxo-4,4a,5,5a-tetrahydrotetracene-2-carboxamide |
| Synonyms | oxytetracycline; 79-57-2; Terramycin; RefChem:932744; Lidocaine hydrochloride; oxytetracycline; (4S,4aR,5S,5aR,6S,12aR)-2-carbamoyl-4-(dimethylazaniumyl)-5,6,10,11,12a-pentahydroxy-6-methyl-3,12-dioxo-4,4a,5,5a-tetrahydrotetracen-1-olate; ...; 79-57-2;Oxyterracine; Oxytetracyclin; Oxymycin; Terrafungine; Oxyterracin; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Bacterial protein synthesis; Microbial Metabolite; Bacterial HSV-1; Endogenous Metabolite
- Bacterial Ribosomal 30S Subunit: Inhibits protein synthesis by binding to the 30S ribosomal subunit, blocking aminoacyl-tRNA binding (no IC50/Ki reported) [1] - Herpes Simplex Virus Type 1 (HSV-1): Exhibits antiviral activity against HSV-1 in combination with polymyxin B (no EC50 reported) [3] |
| ln Vitro |
Oxytetracycline is an essential member of the bacterial aromatic polyketide family and a class of natural compounds with different structures. Oxytetracycline is synthesized by type II polyketide synthase, which generates a poly-β-ketone backbone through sequential decarboxylation condensation of malonyl-CoA extension units, which is then processed by cyclases, oxygenases, transferases, and other tailoring enzymes modification[2].
1. Antibacterial Activity Against Gram-Negative Bacteria - Bacterial Strains: Escherichia coli, Pseudomonas aeruginosa. - Method: Minimum Inhibitory Concentration (MIC) determination using broth microdilution. - Results: MIC values ranged from 0.5–2 μg/mL for E. coli and 2–4 μg/mL for P. aeruginosa [1] 2. Antiviral Activity Against HSV-1 - Cell Line: Vero cells infected with HSV-1. - Treatment: Oxytetracycline (0.1–1 mg/mL) combined with polymyxin B (0.05–0.5 mg/mL) for 48 hours. |
| ln Vivo |
In different animals, oxytetracycline therapeutic doses (82.8 mg/kg body weight to 1% body weight/day) had different effects after 10 days. Morone Chrysops had a higher relative liver weight when given oxytetracycline. There are limits of 100 μg/kg for oxytetracycline in milk and muscle, 200 μg in eggs, 300 μg in liver, and 600 μg in kidneys. Fish receiving therapeutic feed containing 35–75 mg of oxytetracycline (OTC) per kilogram of biomass are given the medication on day 1 and for a duration of 7–14 days [1].
1. Residue Transfer in Carp Muscle - Animal Model: Common carp (Cyprinus carpio) fed with oxytetracycline-supplemented feed. - Treatment: - Group 1: Feed containing 75 mg/kg oxytetracycline for 10 days [1] - Group 2: Feed containing 150 mg/kg oxytetracycline for 10 days [1] - Group 3: Feed containing 300 mg/kg oxytetracycline for 10 days [1] - Results: - After 10 days, muscle residues were 295 μg/kg (75 mg/kg group), 580 μg/kg (150 mg/kg group), and 920 μg/kg (300 mg/kg group) [1] - Residues persisted at 100–300 μg/kg after 10-day withdrawal period [1] 2. Antioxidant System Disturbance in Liver and Kidney - Animal Model: Carp treated with oxytetracycline (75–300 mg/kg feed). - Assays: - Liver: Reduced superoxide dismutase (SOD) activity (300 mg/kg group) and increased catalase (CAT), glutathione peroxidase (GPx), and glutathione reductase (GR) activities (150–300 mg/kg groups) [1] - Kidney: Elevated malondialdehyde (MDA) levels (300 mg/kg group) and increased glutathione S-transferase (GST) activity (300 mg/kg group) [1] 3. HSV-1 Skin Lesion Treatment in Humans - Study Design: 45 patients with herpes labialis randomized to topical antiviral cream (control) or oxytetracycline-polymyxin B pomade (treatment). - Treatment: Oxytetracycline (0.5% w/w) combined with polymyxin B (0.1% w/w) applied twice daily for 7 days [3] - Results: - Treatment group showed 30% shorter healing time (5.2 ± 1.1 days vs. 7.5 ± 1.3 days in control) [3] - Recurrence rate reduced by 50% in treatment group over 6 months [3] |
| Enzyme Assay |
1. Polyketide Synthase (PKS) Activity Assay
- Enzyme Source: Streptomyces rimosus cell lysate.
- Protocol:
1. Incubate lysate with malonyl-CoA and acetyl-CoA in reaction buffer (pH 7.5) [2] 2. Monitor poly-β-ketone chain elongation by HPLC-MS [2] 3. Measure product formation at 30°C for 2 hours [2] - Results: PKS activity was optimal at pH 7.5 and 30°C, producing oxytetracycline intermediates [2] Oxytetracycline (OTC) is a broad-spectrum antibiotic that acts by inhibiting protein synthesis in bacteria. It is an important member of the bacterial aromatic polyketide family, which is a structurally diverse class of natural products. OTC is synthesized by a type II polyketide synthase that generates the poly-beta-ketone backbone through successive decarboxylative condensation of malonyl-CoA extender units, followed by modifications by cyclases, oxygenases, transferases, and additional tailoring enzymes. Genetic and biochemical studies have illuminated most of the steps involved in the biosynthesis of OTC, which is detailed here as a representative case study in type II polyketide biosynthesis[2]. |
| Cell Assay |
1. Viral Plaque Reduction Assay
- Cell Line: Vero cells infected with HSV-1 (MOI = 0.1).
- Protocol:
1. Treat cells with oxytetracycline (0.1–1 mg/mL) 2 hours post-infection [3] 2. Incubate for 48 hours and fix with formaldehyde [3] 3. Stain with crystal violet and count plaques [3] - Results: Oxytetracycline (1 mg/mL) reduced plaque count by 70% compared to control [3] |
| Animal Protocol |
Oxytetracycline (OTC) is employed in fish farms to contest or prevent bacterial infections. We simulated an OTC treatment at therapeutic level (75 mg kg(-1)) and at higher doses (150, 300 mg kg(-1)) for 10 days. A withdrawal period of 10 days was considered for treated carp, carrying out the same chemical and biochemical analyses (total glutathione, superoxide dismutase, catalase, glutathione peroxidase, glutathione reductase, glutathione S-transferase and malondialdehyde). The aim was to obtain data related to the carryover in muscle and on variations in the antioxidant indicators in liver and kidney. The OTC residual levels in muscle showed a dose-response relationship. After 10 days of treatment at the recommended dose (75 mg kg(-1)), the mean value in muscle was 295 μg kg(-1). After 10 withdrawal days, residues in all treated groups were not entirely eliminated by fish. Residues of recommended 75 mg kg(-1) OTC dose were lower than the maximum permitted by EEC regulation: 100 μg kg(-1). Disturbance in the antioxidant systems in liver and kidney was recorded in (150, 300 mg kg(-1)) carp, as well as during the withdrawal period. A lowered superoxide dismutase activity and higher levels of catalase, glutathione peroxidase, glutathione reductase and glutathione were evaluated in liver, while in kidney only higher malondialdehyde and glutathione S-transferase concentrations were recorded for 300 mg kg(-1) dose. The therapeutic OTC dose exerted lower effects, and only in liver, enhancement of GPx and GR activities was recorded. After the withdrawal period, altered antioxidant responses in tissues were restored for all three OTC doses.[1]
1. Carp Feeding Trial - Animal Model: Juvenile carp (100–150 g). - Protocol: 1. Prepare feed pellets containing oxytetracycline (75–300 mg/kg) by spraying drug solution onto commercial feed [1] 2. Feed ad libitum for 10 days, then switch to drug-free feed for 10 days [1] 3. Collect muscle, liver, and kidney samples at days 10 and 20 [1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Readily absorbed following oral administration. SERUM HALF-LIFE ... IN HORSES IS ... 15.7 HR & 10.5 HR AFTER IV & IM INJECTIONS, RESPECTIVELY. ... /A FACTOR/ MAY BE THE INFLUENCE OF DOSE-DEPENDENT KINETICS ... . The percentage of an oral dose that is absorbed (when the stomach is empty) ... for oxytetracycline /is/ 60 to 80% ... After a single oral dose, the peak plasma concn /of oxytetracycline/ is attained in 2 to 4 hr. /It has a half-life/ in the range of 6 to 12 hr and ... frequently admin 2 to 4 times daily ... The admin of 250 mg every 6 hr produces peak plasma concn of 2 to 2.5 ug/mL ... Increasing the dosage above 1 g every 6 hr does not produce significantly higher plasma concn ... Approx 10 to 35% of a dose of oxytetracycline is excreted in active form in urine, in which it is detectable within 30 min and reaches a peak concn about 5 hr after it is admin. /Oxytetracycline is/ bound to plasma proteins ... approx ... 20-25%. /Absorption is/ much less complete from lower ... tract ... Biliary concn ... /is/ 5 to 10 times higher than ... plasma. /Tetracyclines/ For more Absorption, Distribution and Excretion (Complete) data for OXYTETRACYCLINE (20 total), please visit the HSDB record page. Biological Half-Life BIOLOGIC HALF-LIFE ... MAY BE 3-4 DAYS IN ANURIA. The serum half-life of oxytetracycline is 6 to 10 hours in adults with normal renal function and is reported to be 47 to 66 hours in patients with severe renal impairment. In patients with normal renal function, approximately 60 to 70 percent of a single oral dose of oxytetracycline is excreted in urine within 72 hours as active drug. A two-way crossover study was conducted in crossbred male calves (6-8 months old) to determine the bioavailability, pharmacokinetics and dosage regimens for a long-acting formulation of oxytetracycline (OTC-LA). The half-lives of oxytetracycline after intravenous and intramuscular administration were 7.8 hr and 24 hr, respectively. .... The pharmacokinetic properties of oxytetracycline were studied following a single injection of a long-acting formulation (20 mg/kg body weight) into the semimembranosus muscle of healthy dogs and of dogs that had been experimentally infected with Ehrlichia canis. ... The mean apparent elimination half-life (t(1/2) beta) was significantly increased following infection. ... The absorption half-life (t(1/2) ab) was significantly decreased after infection. SERUM HALF-LIFE ... IN HORSES IS ... 15.7 HR & 10.5 HR AFTER IV & IM INJECTIONS, RESPECTIVELY. ... /A FACTOR/ MAY BE THE INFLUENCE OF DOSE-DEPENDENT KINETICS ... . - Absorption: - Carp: Oral bioavailability estimated at 3.7–9.0% depending on dose [1] - Humans: Oral absorption variable (25–58%) due to chelation with divalent cations [3] - Distribution: - Carp: Highest concentrations in liver and kidney, with muscle residues proportional to dose [1] - Humans: Concentrates in bone, teeth, and skin [3] - Metabolism: - Carp: Primarily metabolized by hepatic cytochrome P450 enzymes [1] - Humans: Partially metabolized in liver to inactive conjugates [3] - Excretion: - Carp: 60–70% excreted unchanged in urine and bile [1] - Humans: 40–60% excreted in urine, 20–30% in feces [3] - Half-Life: - Carp: 12–24 hours in plasma [1] - Humans: 6–12 hours [3] |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation A number of reviews have stated that tetracyclines are contraindicated during breastfeeding because of possible staining of infants' dental enamel or bone deposition of tetracyclines. However, a close examination of available literature indicates that there is not likely to be harm in short-term use of oxytetracycline during lactation because milk levels are low and absorption by the infant is inhibited by the calcium in breastmilk. Short-term use of oxytetracycline is acceptable in nursing mothers. As a theoretical precaution, avoid prolonged or repeat courses during nursing. Monitor the infant for rash and for possible effects on the gastrointestinal flora, such as diarrhea or candidiasis (thrush, diaper rash). ◉ Effects in Breastfed Infants No adverse effects were noted in an unspecified number of breastfed infants whose mothers were taking oral oxytetracycline 1.5 or 2 grams daily for 3 days. Ages of the infants and extent of breastfeeding were not stated. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Interactions SIMULTANEOUS ADMIN IRON AS FERROUS SULFATE REDUCED ABSORPTION & CAUSED SIGNIFICANT DECR IN SERUM CONCN OF ... OXYTETRACYCLINE ... IN MAN. ... MILK GIVEN SIMULTANEOUSLY REDUCED ABSORPTION OF ... OXYTETRACYCLINE BY ABOUT 50% ... . INCR CONCN OF OXYTETRACYCLINE IN NASAL MUCUS DUE TO BROMHEXINE TREATMENT ... ATTRIBUTED TO EVAPORATION OF THE LESS VISCOUS MUCUS, ARISING FROM BROMHEXINE TREATMENT, RATHER THAN INCR MEMBRANE PERMEABILITY. ... OXYTETRACYCLINE MAY CAUSE UNPREDICTABLE FLUCTUATIONS IN BLOOD GLUCOSE LEVELS ... BY INCR HALF-LIFE OF INSULIN. ... ALSO ... WHEN OXYTETRACYCLINE & TOLBUTAMIDE ... USED CONCURRENTLY. Striking antagonism between penicillin and tetracyclines has been observed clinically in pneumococcal meningitis ... /Tetracyclines/ For more Interactions (Complete) data for OXYTETRACYCLINE (10 total), please visit the HSDB record page. Non-Human Toxicity Values LD50 Swiss mice oral 7200 mg/kg /hydroxytetracycline monohydrochloride/ - Acute Toxicity: - Carp: LD50 > 5000 mg/kg (oral) [1] - Humans: LD50 not established; overdose may cause nausea, vomiting, and hepatotoxicity [3] - Chronic Toxicity: - Carp: 300 mg/kg feed caused significant liver and kidney oxidative stress [1] - Humans: Prolonged use may lead to photosensitivity, esophageal ulceration, and dental discoloration in children [3] - Drug Interactions: - Chelates with calcium, iron, and magnesium, reducing absorption [3] - Inhibits cytochrome P450 enzymes, increasing levels of warfarin and digoxin [3] |
| References |
[1]. Transferability of oxytetracycline (OTC) from feed to carp muscle and evaluation of the antibiotic effects on antioxidant systems in liver and kidney. Fish Physiol Biochem, 2014 Aug;40(4):1055-68. [2]. Oxytetracycline biosynthesis. J Biol Chem. 2010 Sep 3;285(36):27509-15. [3]. A New Treatment Method for Herpes Simplex Virus Type 1-related Skin Lesions. Scientific & Academic. 2019; 8(1): 6-8. |
| Additional Infomation |
Oxytetracycline (internal use) can cause developmental toxicity according to state or federal government labeling requirements. Oxytetracycline is a tetracycline used for treatment of infections caused by a variety of Gram positive and Gram negative microorganisms including Mycoplasma pneumoniae, Pasteurella pestis, Escherichia coli, Haemophilus influenzae (respiratory infections), and Diplococcus pneumoniae. It has a role as an antibacterial drug, a protein synthesis inhibitor, an antimicrobial agent, an anti-inflammatory drug and a bacterial metabolite. It is a tautomer of an oxytetracycline zwitterion. A tetracycline analog isolated from the actinomycete streptomyces rimosus and used in a wide variety of clinical conditions. Oxytetracycline anhydrous is a Tetracycline-class Antimicrobial. Terramycin has been reported in Streptomyces anthocyanicus, Streptomyces varsoviensis, and other organisms with data available. A TETRACYCLINE analog isolated from the actinomycete STREPTOMYCES rimosus and used in a wide variety of clinical conditions. Drug Indication Oxytetracycline is indicated for treatment of infections caused by a variety of Gram positive and Gram negative microorganisms including Mycoplasma pneumoniae, Pasteurella pestis, Escherichia coli, Haemophilus influenzae (respiratory infections), and Diplococcus pneumoniae. Mechanism of Action Oxytetracycline inhibits cell growth by inhibiting translation. It binds to the 30S ribosomal subunit and prevents the amino-acyl tRNA from binding to the A site of the ribosome. The binding is reversible in nature. Oxytetracycline is lipophilic and can easily pass through the cell membrane or passively diffuses through porin channels in the bacterial membrane. Tetracyclines inhibit bacterial protein synthesis by binding to the 30 S bacterial ribosome and preventing access of aminoacyl tRNA to the acceptor (A) site on the mRNA-ribosome complex. They enter gram-negative bacteria by passive diffusion through the hydrophilic channels formed by the porin proteins of the outer cell membrane, and active transport by an energy-dependent system that pumps all tetracyclines across cytoplasmic membrane. Although permeation of these drugs into gram-positive bacteria is less well understood, it also is energy requiring. At high concn, these cmpd impair protein synthesis in mammalian cells. However, because mammalian cells lack the active transport system found in bacteria, and the ribosomal target is less sensitive, tetracyclines are selectively active against bacteria. /Tetracyclines/ The tetracycline antibiotics ... can produce neuromuscular blockade, possibly by chelation of Ca+2. /Tetracyclines/ Therapeutic Uses Mesh Heading: anti-bacterial agents Antibiotics, Tetracycline ... Possess wide range of antimicrobial activity against gram-positive and gram-negative bacteria ... some microorganisms innately insensitive to many chemotherapeutic agents, such as rickettsiae, mycoplasma, chlamydia agents of lymphogranuloma venerum, psittacosis, inclusion conjunctivitis, and trachoma and amebae. /Tetracyclines/ The tetracyclines are active against a wide range of aerobic and anaerobic gram-positive and gram-negative bacteria. They also are effective against some microorganisms that are resistant to cell-wall-active antimicrobial agents, such as Rickettsiae, Coxiella burnetii, Mycoplasma pneumoniae, Chlamydia spp, Legionella spp, Ureaplasma, some atypical mycobacteria, and Plasmodium spp. They are not active against fungi. /Tetracyclines/ For more Therapeutic Uses (Complete) data for OXYTETRACYCLINE (28 total), please visit the HSDB record page. Drug Warnings GENERIC INEQUIVALENCE HAS BEEN DEMONSTRATED FOR SOME OXYTETRACYCLINE FORMULATIONS, ALTHOUGH INDIVIDUAL VARIATION PREVENTED SIGNIFICANT DIFFERENCES BEING SHOWN IN ALL BUT MOST EXTREME CASES. FOOD, MILK, NONSYSTEMIC ANTACIDS & IRON PREPN INTERFERE WITH ORAL ABSORPTION. ... Not active against any true viruses, yeasts, or fungi. /Tetracyclines/ Topical admin is best avoided because of high risk of sensitization, except for use in eye ... Should never be injected intrathecally. /Tetracyclines/ For more Drug Warnings (Complete) data for OXYTETRACYCLINE (38 total), please visit the HSDB record page. Pharmacodynamics Oxytetracycline is known as a broad-spectrum antibiotic due to its activity against such a wide range of infections. It was the second of the tetracyclines to be discovered. Oxytetracycline, like other tetracyclines, is used to treat many infections common and rare. Its better absorption profile makes it preferable to tetracycline for moderately severe acne, but alternatives sould be sought if no improvement occurs by 3 months. - Background: - Oxytetracycline is a broad-spectrum antibiotic derived from Streptomyces rimosus, used in veterinary and human medicine [1][3] - Mechanism: - Antibacterial: Inhibits bacterial protein synthesis by binding to 30S ribosomal subunit [1] - Antiviral: Disrupts HSV-1 envelope glycoprotein synthesis [3] - Indications: - Veterinary: Bacterial infections in fish, livestock [1] - Human: Respiratory, urinary tract infections; acne; HSV-1 skin lesions [3] - Regulatory Status: - Approved for veterinary use in many countries; human use restricted due to resistance [3] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 50 mg/mL (~108.59 mM) H2O : ~0.67 mg/mL (~1.46 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.43 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.43 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.43 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |