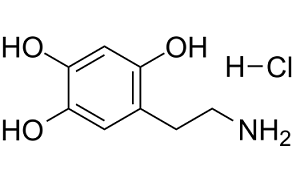
Physicochemical Properties
| Molecular Formula | C₈H₁₂CLNO₃ |
| Molecular Weight | 205.64 |
| Exact Mass | 205.051 |
| Elemental Analysis | C, 46.73; H, 5.88; Cl, 17.24; N, 6.81; O, 23.34 |
| CAS # | 28094-15-7 |
| Related CAS # | 1199-18-4; 28094-15-7 (HCl); 636-00-0 (HBr) |
| PubChem CID | 160157 |
| Appearance | White to gray solid |
| Boiling Point | 406ºC at 760 mmHg |
| Melting Point | 232-233ºC (dec.)(lit.) |
| Flash Point | 199.3ºC |
| Vapour Pressure | 3.58E-07mmHg at 25°C |
| LogP | 1.806 |
| Hydrogen Bond Donor Count | 5 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 13 |
| Complexity | 142 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | OC1=CC(CCN)=C(O)C=C1O.[H]Cl |
| InChi Key | QLMRJHFAGVFUAC-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H11NO3.ClH/c9-2-1-5-3-7(11)8(12)4-6(5)10;/h3-4,10-12H,1-2,9H2;1H |
| Chemical Name | 2,4,5-Trihydroxyphenethylamine hydrochloride |
| Synonyms | 6-Hydroxydopamine hydrochloride; 28094-15-7; 6-Hydroxydopamine hydrochloride; 5-(2-aminoethyl)benzene-1,2,4-triol hydrochloride; 6-Hydroxydopamine chloride; Oxidopamine (hydrochloride); 2,4,5-Trihydroxyphenethylamine hydrochloride; 6-HYDROXYDOPAMINE HCL; 6-OHDA hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture.(2). This product is not stable in solution, please use freshly prepared working solution for optimal results. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
COX-2; IL-1;βCaspase-3; Caspase-8; Caspase-9 - Dopaminergic neurons (selective toxicity) [1][2][3][4] - Cyclooxygenase-2 (COX-2)/prostaglandin E2 (PGE2)/EP2 receptor signaling pathway [1] - Akt survival pathway (inhibition) [3] - Reactive oxygen species (ROS) generation system [4] - Apoptosis-related molecules (caspase-3, Bax/Bcl-2) [3] |
| ln Vitro |
Hydrochloric acid oxidation of dopamine (0-500 μM, 24 h) lowers the viability of Neuro-2a cells and SH-SY5Y cells in a concentration-dependent manner [1]. Hydrochloric acid oxidizes dopamine (75-150 μM, 0-24 h) to promote COX-2 expression. Oxydopamine hydrochloride (75-150 μM, 0-24 h) induces PGE 2 biosynthesis and the generation of pro-inflammatory cytokine IL-1β. [1]. Oxystopamine hydrochloride (0-150 μM, 12 h) and nuclear translocation [1]. ) promotes mitochondrial membrane delocalization and mitochondrial membrane delocalization in alkaline electrophoresis cell carcinoma PC12 cells [3]. Oxidopamine hydrochloride (75 μM, 0-12 h) promotes p38 phosphorylation [3]. [1] - Oxidopamine hydrochloride (6-OHDA) exhibited selective neurotoxicity to dopaminergic cells. In PC12 cells, 50 μM induced 58±4% cell death after 24 hours; in primary cortical neurons, 100 μM reduced viability by 65±5% [3][4] - It activated the COX-2/EP2 pathway: 100 μM increased COX-2 mRNA and protein levels by 2.8±0.3 and 3.2±0.2-fold, and PGE2 secretion by 2.5±0.2-fold in neurons [1] - It induced apoptosis via inhibiting the Akt pathway: 50 μM decreased p-Akt (Ser473) by 62±4%, increased cleaved caspase-3 by 2.3±0.2-fold, and raised the Bax/Bcl-2 ratio by 3.1±0.3-fold in PC12 cells [3] - It generated excessive ROS through autoxidation: 100 μM increased ROS levels by 3.8±0.3-fold in neurons, which was mitigated by antioxidants (e.g., resveratrol) [4] |
| ln Vivo |
Dopaminergic neurons in the substantia nigra degenerate when oxytopamine hydrochloride (5 μg/2 μL) is unilaterally injected into the right striatum [2]. - In rat Parkinson's disease (PD) model: Stereotaxic injection of Oxidopamine hydrochloride (10 μg/μL, 4 μL unilateral striatal injection) induced dopaminergic neuron loss. Substantia nigra pars compacta (SNpc) tyrosine hydroxylase (TH)-positive neurons decreased by 72±6% after 21 days [2] - It caused motor dysfunction: Rats showed 45±4% reduction in rotarod latency and 38±3% decrease in open-field locomotor activity compared to sham group [2] - It activated neuroinflammation: Striatal TNF-α and IL-1β levels increased by 2.1±0.2 and 1.8±0.1-fold, respectively, 7 days post-injection [1] |
| Enzyme Assay |
Cyclooxygenase-2 (COX-2) triggers pro-inflammatory processes that can aggravate neuronal degeneration and functional impairments in many neurological conditions, mainly via producing prostaglandin E2 (PGE2) that activates four membrane receptors, EP1-EP4. However, which EP receptor is the culprit of COX-2/PGE2-mediated neuronal inflammation and degeneration remains largely unclear and presumably depends on the insult types and responding components. Herein, we demonstrated that COX-2 was induced and showed nuclear translocation in two neuronal cell lines - mouse Neuro-2a and human SH-SY5Y - after treatment with neurotoxin 6-hydroxydopamine (6-OHDA), leading to the biosynthesis of PGE2 and upregulation of pro-inflammatory cytokine interleukin-1β. Inhibiting COX-2 or microsomal prostaglandin E synthase-1 suppressed the 6-OHDA-triggered PGE2 production in these cells. Treatment with PGE2 or EP2 selective agonist butaprost, but not EP4 agonist CAY10598, increased cAMP response in both cell lines. PGE2-initiated cAMP production in these cells was blocked by our recently developed novel selective EP2 antagonists - TG4-155 and TG6-10-1, but not by EP4 selective antagonist GW627368X. The 6-OHDA-promoted cytotoxicity was largely blocked by TG4-155, TG6-10-1 or COX-2 selective inhibitor celecoxib, but not by GW627368X. Our results suggest that PGE2 receptor EP2 is a key mediator of COX-2 activity-initiated cAMP signaling in Neuro-2a and SH-SY5Y cells following 6-OHDA treatment, and contributes to oxidopamine-mediated neurotoxicity [1]. - COX-2 activity assay: Neurons were treated with Oxidopamine hydrochloride (50, 100 μM) for 24 hours. Culture supernatants were collected to measure PGE2 concentration by enzyme immunoassay, reflecting COX-2 activity [1] - ROS detection assay: Cells were loaded with ROS-specific fluorescent probe after 6-OHDA treatment (25, 50, 100 μM) for 12 hours. Fluorescence intensity was measured at 488/525 nm to quantify ROS levels [4] - Caspase-3 activity assay: PC12 cell lysates were incubated with caspase-3 specific fluorogenic substrate. Fluorescence at 400/505 nm was detected to evaluate apoptotic enzyme activity [3] |
| Cell Assay |
Cell Viability Assay[1] Cell Types: Neuro-2a cells and SH-SY5Y cells Tested Concentrations: 0-500 µM Incubation Duration: 24 or 48 hrs (hours) Experimental Results: Induction of neurotoxicity in Neuro-2a cells and SH-SY5Y cells in a concentration-dependent manner Cytotoxicity. In Neuro-2a cells, EC50=111 µM (incubation for 24 hrs (hours)) and 109 µM (incubation for 48 hrs (hours)); in SH-SY5Y cells, EC50=118 µM for 24 hrs (hours) and EC50=107 µM for 48 hrs (hours). RT-PCR[1] Cell Types: Neuro-2a cells and SH-SY5Y cells Tested Concentrations: 75 or 150 µM Incubation Duration: 0, 6 or 24 hrs (hours) Experimental Results: Rapid and robust induction of COX-2 in a time-dependent manner. Induces COX-2 activation, characterized by induction of expression and nuclear translocation. PGE2 in the culture medium increased Dramatically by nearly 5-fold in Neuro-2a cells (75 µM) and 3-fold in SH-SY5Y cells (150 µM). The pro-inflammatory cytokine interleukin 1β (IL-1β) was Dramatically upregulated in Neuro-2a cells and SH-SY5Y cells. Apoptosis analysis [3] Cell Types: PC12 Cell conce - Dopaminergic cell toxicity assay: PC12 cells or primary dopaminergic neurons were seeded in 96-well plates (5×10³ cells/well), treated with Oxidopamine hydrochloride (10–200 μM) for 24–48 hours. Cell viability was measured by MTT assay [3][4] - Apoptosis detection assay: PC12 cells were treated with 50 μM 6-OHDA for 24 hours, stained with Annexin V-FITC/PI, and analyzed by flow cytometry. Bax/Bcl-2 and cleaved caspase-3 levels were detected by Western blot [3] - COX-2/EP2 pathway assay: Neurons were treated with 6-OHDA (50, 100 μM) for 24 hours. COX-2 and EP2 mRNA levels were detected by RT-PCR; protein levels by Western blot [1] |
| Animal Protocol |
The present study was undertaken to investigate the neuroprotective effects of resveratrol on 6-hydroxydopamine (6-OHDA)-induced Parkinson's disease in rats. 6-OHDA-induced Parkinson's disease rat model involves chronic inflammation, mitochondrial dysfunction, and oxidative stress, and the loss of the dopaminergic neurons in the substantia nigra is the predominant lesion. Resveratrol has been shown to have anti-inflammatory actions, and thus was tested for its beneficial effects using 6-OHDA-induced Parkinson's disease rat model. Adult Sprague-Dawley (SD) rats were unilaterally injected with 6-OHDA (5 microg/2 microl) into the right striatum, and the striatum damage was assessed by rotational test, ultrahistopathology, and molecular alterations. Resveratrol (10, 20 and 40 mg/kg) was then given orally to Parkinson's disease rats, daily for 10 weeks to examine the protective effects. Rotational test (turns of rats) showed that resveratrol significantly attenuated apomorphine-induced turns of rats in 6-OHDA-injuried Parkinson's disease rat model as early as two weeks of administration. Ultrastructural analysis showed that resveratrol alleviated 6-OHDA-induced chromatin condensation, mitochondrial tumefaction and vacuolization of dopaminergic neurons in rat substantia nigra. Furthermore, resveratrol treatment also significantly decreased the levels of COX-2 and TNF-alpha mRNA in the substantia nigra as detected by real-time RT-PCR. COX-2 protein expression in the substantia nigra was also decreased as evidenced by Western blotting. These results demonstrate that resveratrol exerts a neuroprotective effect on 6-OHDA-induced Parkinson's disease rat model, and this protection is related to the reduced inflammatory reaction.[2]. - Rat PD model establishment: Male Sprague-Dawley rats (250–300 g) were anesthetized. Oxidopamine hydrochloride was dissolved in 0.9% saline containing 0.02% ascorbic acid (to prevent autoxidation) and injected unilaterally into the striatum (coordinates: AP +0.2 mm, ML -3.0 mm, DV -5.0 mm relative to bregma) at 10 μg/μL, 4 μL total volume [2] - Behavioral tests: Rotarod test (latency to fall) and open-field test (locomotor distance) were performed 7, 14, 21 days post-injection [2] - Histological analysis: Rats were sacrificed 21 days post-injection. Brain tissues were sectioned, and TH-positive neurons in SNpc were detected by immunohistochemistry [2] |
| Toxicity/Toxicokinetics |
- Selective neurotoxicity: Targets dopaminergic neurons; no significant cytotoxicity to non-dopaminergic cells (e.g., astrocytes) at 100 μM [1][4] - In vitro IC50: 42.5 ± 3.1 μM for PC12 cell viability inhibition (24-hour incubation) [3] - No obvious systemic toxicity: Rats showed no significant changes in liver/kidney function or body weight post-injection [2] |
| References |
[1]. Cyclooxygenase-2 contributes to oxidopamine-mediated neuronal inflammation and injury via the prostaglandin E2 receptor EP2 subtype. Sci Rep. 2017 Aug 25;7(1):9459. [2]. Neuroprotective effect of resveratrol on 6-OHDA-induced Parkinson's disease in rats. Eur J Pharmacol. 2008 Dec 14;600(1-3):78-82. [3]. Cell-permeable cAMP analog suppresses 6-hydroxydopamine-induced apoptosis in PC12 cells through the activation of the Akt pathway. Brain Res. 2006 Oct 3;1113(1):10-23. [4]. Autoxidation and neurotoxicity of 6-hydroxydopamine in the presence of some antioxidants: potential implication in relation to the pathogenesis of Parkinson's disease. J Neurochem. 2000 Apr;74(4):1605-12. |
| Additional Infomation |
6-hydroxydopamine hydrochloride is a beige solid. (NTP, 1992) A neurotransmitter analogue that depletes noradrenergic stores in nerve endings and induces a reduction of dopamine levels in the brain. Its mechanism of action is related to the production of cytolytic free-radicals. - Oxidopamine hydrochloride (6-OHDA) is a synthetic dopamine analog widely used to induce PD models in vitro and in vivo [1][2][3][4] - Its neurotoxic mechanism involves: 1) Autoxidation to generate ROS and quinones, causing oxidative stress; 2) Activating COX-2/EP2-mediated neuroinflammation; 3) Inhibiting the Akt survival pathway to induce apoptosis [1][3][4] - Antioxidants (e.g., resveratrol), COX-2 inhibitors, or cAMP analogs can mitigate its neurotoxicity [2][3][4] - It is unstable in aqueous solution and requires fresh preparation with antioxidant (ascorbic acid) to prevent pre-autoxidation [4] |
Solubility Data
| Solubility (In Vitro) |
H2O : ~100 mg/mL (~486.29 mM) DMSO : ~83.33 mg/mL (~405.22 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (10.11 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (10.11 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: 100 mg/mL (486.29 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.8629 mL | 24.3143 mL | 48.6287 mL | |
| 5 mM | 0.9726 mL | 4.8629 mL | 9.7257 mL | |
| 10 mM | 0.4863 mL | 2.4314 mL | 4.8629 mL |