Physicochemical Properties
| Molecular Formula | C8H8O4 |
| Molecular Weight | 168.1467 |
| Exact Mass | 168.042 |
| CAS # | 480-64-8 |
| PubChem CID | 68072 |
| Appearance | Off-white to light yellow solid powder |
| Density | 1.5±0.1 g/cm3 |
| Boiling Point | 391.4±22.0 °C at 760 mmHg |
| Melting Point | 173-174ºC |
| Flash Point | 204.7±18.8 °C |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.646 |
| Source | Phomopsis velata, Penicillium cyclopium, and other organisms |
| LogP | 2.06 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 12 |
| Complexity | 180 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | AMKYESDOVDKZKV-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H8O4/c1-4-2-5(9)3-6(10)7(4)8(11)12/h2-3,9-10H,1H3,(H,11,12) |
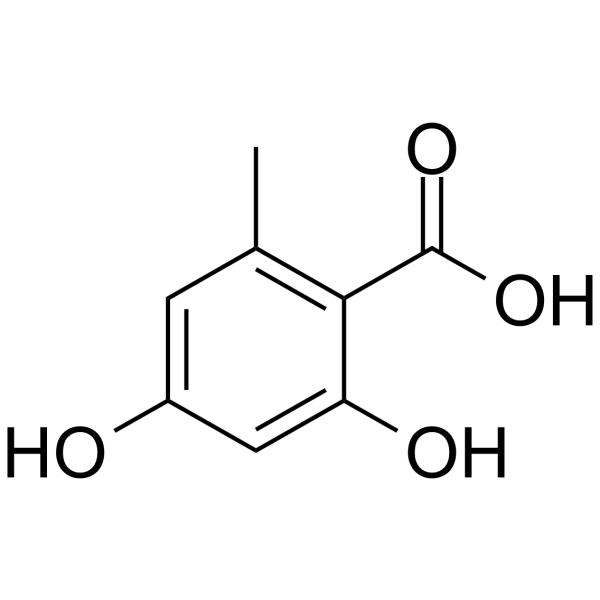
| Chemical Name | 2,4-dihydroxy-6-methylbenzoic acid |
| Synonyms | Orsellinic acid; 2,4-Dihydroxy-6-methylbenzoic acid; 480-64-8; o-Orsellinic acid; Orsellic acid; Orcinolcarboxylic acid; 2,4-dihydroxy-6-methyl-benzoic acid; 4,6-Dihydroxy-o-toluic acid; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural lichen depside |
| ln Vitro | Lichens are an important source of phenolic compounds and have been intensively investigated for their biological and pharmacological activities. Lecanoric acid (1), a lichen depside, was isolated from a Parmotrema tinctorum specimen and treated with alcohols to produce orsellinic acid (2) and orsellinates (3) to (9) (2,4-dihydroxy-6-n-methyl benzoates). Free radical scavenging activity of methyl (3), ethyl (4), n-propyl (5), n-butyl (6), iso-propyl (7), sec-butyl (8), tert-butyl (9) orsellinates was evaluated using 2,2'-diphenyl-1-picrylhydrazyl (DPPH) method. Results showed that chain elongation of methyl (3) to n-butyl (6) causes a rise in the antioxidant activity. However, iso-propyl (7) and tert-butyl (9) were more active than the correspondent linear compounds, although sec-butyl (8) was less active among the chain ramified compounds. All the orsellinates were less active than lecanoric acid (1) and orsellinic acid (2). Orcinol (10) and resorcinol (11) were also determined for comparison with activities of orsellinates. Gallic acid (12) was used as control.[1] |
| References |
[1]. Radical-scavenging activity of orsellinates. Chem Pharm Bull (Tokyo). 2008;56(11):1551-1554. |
| Additional Infomation |
O-orsellinic acid is a dihydroxybenzoic acid that is 2,4-dihydroxybenzoic acid in which the hydrogen at position 6 is replaced by a methyl group. It has a role as a metabolite, a marine metabolite and a fungal metabolite. It is a dihydroxybenzoic acid and a member of resorcinols. It is a conjugate acid of an o-orsellinate. 2,4-Dihydroxy-6-methylbenzoic acid has been reported in Phomopsis velata, Penicillium cyclopium, and other organisms with data available. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~1486.77 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (12.37 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (12.37 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (12.37 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.9471 mL | 29.7354 mL | 59.4707 mL | |
| 5 mM | 1.1894 mL | 5.9471 mL | 11.8941 mL | |
| 10 mM | 0.5947 mL | 2.9735 mL | 5.9471 mL |