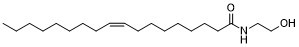
Oleoylethanolamide (AM-1301; AM1301) is an endogenous and potent PPAR-α agonist with the potential to be used in the treatment of obesity and arteriosclerosis.
Physicochemical Properties
| Molecular Formula | C20H39NO2 |
| Molecular Weight | 325.5291 |
| Exact Mass | 325.298 |
| CAS # | 111-58-0 |
| Related CAS # | Oleoylethanolamide-d4;946524-36-3;Oleoylethanolamide-d2;1245477-09-1 |
| PubChem CID | 5283454 |
| Appearance | White to off-white solid powder |
| Density | 0.9±0.1 g/cm3 |
| Boiling Point | 496.4±38.0 °C at 760 mmHg |
| Melting Point | 50-60ºC |
| Flash Point | 254.0±26.8 °C |
| Vapour Pressure | 0.0±2.9 mmHg at 25°C |
| Index of Refraction | 1.474 |
| LogP | 6.36 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 2 |
| Rotatable Bond Count | 17 |
| Heavy Atom Count | 23 |
| Complexity | 277 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CCCCCCCC/C=C\CCCCCCCC(NCCO)=O |
| InChi Key | BOWVQLFMWHZBEF-KTKRTIGZSA-N |
| InChi Code | InChI=1S/C20H39NO2/c1-2-3-4-5-6-7-8-9-10-11-12-13-14-15-16-17-20(23)21-18-19-22/h9-10,22H,2-8,11-19H2,1H3,(H,21,23)/b10-9- |
| Chemical Name | (Z)-N-(2-hydroxyethyl)octadec-9-enamide |
| Synonyms | N-(2-HydroxyethyloleamideAM-1301AM1301 OEA |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Hepatic stellate cells are the target of oléoylethanolamide (OEA), an endogenous PPAR-α ligand that attenuates liver fibrosis. Through PPAR-α, oleoylethanolamide inhibits the activation of hepatic stellate cells (HSCs) elicited by TGF-β1 in vitro. qPCR is used to measure the expression levels of Col1a and α-SMA in TGF-β1-stimulated HSCs in order to evaluate the effect of oleoylethanolamide on HSC activation. When TGF-β1 (5 ng/mL) is stimulated for 48 hours in the group of CFSC cells, the mRNA levels of α-SMA and Col1a are significantly increased; however, the mRNA levels are inhibited in a dose-dependent way when Oleoylethanolamide is applied. The results of immunofluorescence and western blot demonstrate that treatment with oleoylethanolamide dose-dependently suppresses the production of the HSC activation marker α-SMA in protein form. Oleoylethanolamide's inhibitory effects on HSC activation are totally prevented by the PPAR-α antagonist MK886 (10 μM). Furthermore, PPAR-α's mRNA and protein expression levels are down-regulated in response to TGF-β1 stimulation; however, these changes are dose-dependently restored by oleoylethanolamide therapy. Furthermore, it has been discovered that TGF-β1 stimulation causes an upregulation of Smad 2/3 phosphorylation, which is in line with the effects on HSC activation. On the other hand, phosphorylation of Smad 2/3 in CFSC mimicked with TGF-β1 is decreased by oleoylethanolamide (10 μM). |
| ln Vivo | In mice models of hepatic fibrosis, oleoylethanolamide (OEA) can highly reduce the pro-fibrotic cytokine TGF-β1 and adversely regulate genes in the TGF-β1 signaling pathway (α-SMA, collagen 1a, and collagen 3a). By preventing the activation of hepatic stellate cells (HSCs), treatment with oleoylethanolamide (5 mg/kg/day, intraperitoneal injection, ip) considerably slows the progression of liver fibrosis in both experimental animal models[1]. |
| References |
[1]. Oleoylethanolamide, an endogenous PPAR-α ligand, attenuates liver fibrosis targeting hepatic stellate cells. Oncotarget. 2015 Dec 15;6(40):42530-40. |
| Additional Infomation |
Oleoyl ethanolamide is an N-(long-chain-acyl)ethanolamine that is the ethanolamide of oleic acid. The monounsaturated analogue of the endocannabinoid anandamide. It has a role as a PPARalpha agonist, an EC 3.5.1.23 (ceramidase) inhibitor and a geroprotector. It is a N-(long-chain-acyl)ethanolamine, an endocannabinoid and a N-acylethanolamine 18:1. It is functionally related to an oleic acid. n-Oleoylethanolamine has been reported in Drosophila melanogaster, Apis cerana, and other organisms with data available. Mechanism of Action Oleoylethanolamide (OEA) is a major N-acylethanolamine and an endogenous ethanolamide fatty acid. Although it is an endocannabinoids-like compound, it does not bind to cannabinoid receptors. Instead, this lipid sensor is an agonist at peroxisome proliferator-activated receptor-α (PPAR-α) agonist while also being an inhibitor of ceramidase and thereby the sphingolipid signaling pathway. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~20.83 mg/mL (~63.99 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (6.39 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (6.39 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.0719 mL | 15.3596 mL | 30.7191 mL | |
| 5 mM | 0.6144 mL | 3.0719 mL | 6.1438 mL | |
| 10 mM | 0.3072 mL | 1.5360 mL | 3.0719 mL |