Physicochemical Properties
| Molecular Formula | C21H28O6 |
| Molecular Weight | 376.4434 |
| Exact Mass | 376.188 |
| CAS # | 36062-07-4 |
| PubChem CID | 11068834 |
| Appearance | Light yellow to yellow ointment |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 623.5±55.0 °C at 760 mmHg |
| Flash Point | 330.9±31.5 °C |
| Vapour Pressure | 0.0±1.9 mmHg at 25°C |
| Index of Refraction | 1.592 |
| LogP | 1.73 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 27 |
| Complexity | 370 |
| Defined Atom Stereocenter Count | 0 |
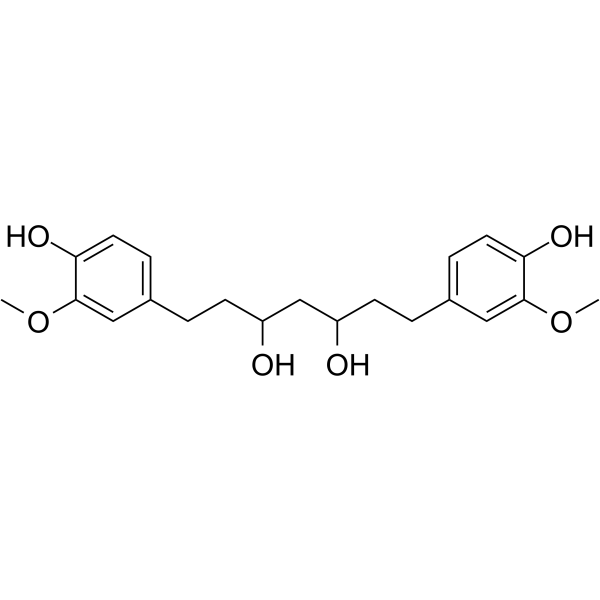
| SMILES | O([H])C([H])(C([H])([H])C([H])([H])C1C([H])=C([H])C(=C(C=1[H])OC([H])([H])[H])O[H])C([H])([H])C([H])(C([H])([H])C([H])([H])C1C([H])=C([H])C(=C(C=1[H])OC([H])([H])[H])O[H])O[H] |
| InChi Key | OELMAFBLFOKZJD-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C21H28O6/c1-26-20-11-14(5-9-18(20)24)3-7-16(22)13-17(23)8-4-15-6-10-19(25)21(12-15)27-2/h5-6,9-12,16-17,22-25H,3-4,7-8,13H2,1-2H3 |
| Chemical Name | 1,7-bis(4-hydroxy-3-methoxyphenyl)heptane-3,5-diol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Free radicals (DPPH, ABTS, superoxide anion): Octahydrocurcumin scavenges DPPH radical (IC₅₀ = 8.5 ± 0.3 μM), ABTS radical (IC₅₀ = 6.2 ± 0.2 μM), and inhibits superoxide anion generation (IC₅₀ = 22.4 ± 1.5 μM) [3] - Lipid peroxidation: Octahydrocurcumin inhibits Fe²⁺-induced lipid peroxidation in rat liver microsomes (IC₅₀ = 12.3 ± 0.8 μM) [3] - NF-κB signaling pathway (p65 subunit): Octahydrocurcumin inhibits LPS-induced NF-κB p65 nuclear translocation in RAW264.7 cells (inhibits translocation, not direct binding) [2] - Inflammatory cytokines (TNF-α, IL-6): Octahydrocurcumin inhibits LPS-induced TNF-α release (IC₅₀ = 15.2 ± 1.1 μM) and IL-6 release (IC₅₀ = 18.7 ± 1.3 μM) from RAW264.7 cells [2] |
| ln Vitro |
1. Antioxidant activity: - DPPH radical scavenging: Octahydrocurcumin (2-20 μM) scavenged DPPH radicals in a dose-dependent manner; IC₅₀ = 8.5 ± 0.3 μM (weaker than curcumin, IC₅₀ = 2.8 ± 0.1 μM) [3] - ABTS radical scavenging: At 10 μM, Octahydrocurcumin showed 72% ABTS radical scavenging rate; IC₅₀ = 6.2 ± 0.2 μM (curcumin IC₅₀ = 1.9 ± 0.1 μM) [3] - Superoxide anion inhibition: Octahydrocurcumin (10-50 μM) inhibited xanthine-xanthine oxidase-induced superoxide anion generation; 50 μM showed 68% inhibition rate, IC₅₀ = 22.4 ± 1.5 μM [3] - Lipid peroxidation inhibition: In rat liver microsomes, Octahydrocurcumin (5-30 μM) reduced Fe²⁺-induced malondialdehyde (MDA) production (MDA is a lipid peroxidation marker); IC₅₀ = 12.3 ± 0.8 μM [3] 2. Anti-inflammatory activity: - Inflammatory cytokine inhibition: RAW264.7 cells (murine macrophages) treated with Octahydrocurcumin (5-40 μM) + LPS (1 μg/ml) for 24 h; 20 μM Octahydrocurcumin reduced TNF-α release by 58% and IL-6 release by 52% (vs. LPS alone) [2] - NF-κB nuclear translocation inhibition: Western blot showed 20 μM Octahydrocurcumin reduced LPS-induced nuclear NF-κB p65 levels to 35% of LPS alone group; cytoplasmic p65 levels increased by 2.1-fold, confirming inhibited translocation [2] - No cytotoxicity: Octahydrocurcumin (up to 40 μM) showed no cytotoxicity to RAW264.7 cells (viability > 90%, MTT assay) [2] |
| Enzyme Assay |
1. Antioxidant activity assays: - DPPH radical scavenging assay: - Reaction mixture (1 ml) contained 50 μM DPPH ethanol solution and Octahydrocurcumin (2-20 μM, dissolved in ethanol) [3] - Incubated at room temperature in the dark for 30 minutes; absorbance measured at 517 nm [3] - Scavenging rate (%) = [(A₀ - A₁)/A₀] × 100 (A₀ = control absorbance, A₁ = sample absorbance); IC₅₀ calculated via dose-response curve [3] - ABTS radical scavenging assay: - ABTS radical cation was generated by reacting 7 mM ABTS with 2.45 mM potassium persulfate (incubated 16 h at room temperature) [3] - Diluted ABTS solution (absorbance 0.7 ± 0.05 at 734 nm) mixed with Octahydrocurcumin (2-20 μM); incubated 10 minutes [3] - Absorbance measured at 734 nm; scavenging rate and IC₅₀ calculated as above [3] - Lipid peroxidation inhibition assay: - Rat liver microsomes (0.5 mg protein/ml) mixed with 50 μM FeSO₄, 0.1 mM ascorbic acid, and Octahydrocurcumin (5-30 μM) in 50 mM Tris-HCl buffer (pH 7.4) [3] - Incubated at 37°C for 1 h; reaction terminated by adding 10% trichloroacetic acid (TCA) [3] - MDA concentration measured by thiobarbituric acid (TBA) reaction (absorbance 532 nm); inhibition rate = [(MDA₀ - MDA₁)/MDA₀] × 100 [3] 2. NF-κB transcriptional activity assay: - RAW264.7 cells were co-transfected with NF-κB luciferase reporter plasmid and Renilla luciferase plasmid (internal control) [2] - Transfected cells pre-treated with Octahydrocurcumin (5-40 μM) for 1 h, then stimulated with LPS (1 μg/ml) for 6 h [2] - Cells lysed; luciferase activity measured using dual-luciferase assay kit; NF-κB activity = firefly luciferase/Renilla luciferase ratio [2] - Result: 20 μM Octahydrocurcumin inhibited LPS-induced NF-κB activity by 62% (vs. LPS alone) [2] |
| Cell Assay |
1. RAW264.7 macrophage inflammatory response assay: - Cell culture: RAW264.7 cells were cultured in DMEM medium with 10% FBS, 100 U/ml penicillin, 100 μg/ml streptomycin (37°C, 5% CO₂) [2] - Treatment protocol: Cells (1×10⁶ cells/ml, 24-well plate) were pre-treated with Octahydrocurcumin (5-40 μM) for 1 h, then stimulated with LPS (1 μg/ml) for 24 h [2] - Inflammatory cytokine detection: Culture supernatant collected; TNF-α/IL-6 levels measured by sandwich ELISA (detection wavelength 450 nm); concentrations calculated via standard curves [2] - NF-κB p65 subcellular localization (Western blot): - Cytoplasmic and nuclear fractions extracted from treated cells using nuclear extraction kit [2] - 30 μg protein per fraction separated by SDS-PAGE, transferred to PVDF membrane, blocked with 5% non-fat milk (1 h) [2] - Membranes probed with anti-NF-κB p65 antibody (cytoplasmic/nuclear marker: α-tubulin/Lamin B1) at 4°C overnight, then secondary antibody (1 h, RT) [2] - Bands visualized with ECL; intensity quantified via densitometry [2] 2. Cell viability assay (文献[2]、[3]): - MTT assay for RAW264.7 cells: Cells (5×10³ cells/well, 96-well plate) treated with Octahydrocurcumin (5-40 μM) for 24 h; MTT (5 mg/ml, 20 μl/well) added for 4 h; formazan dissolved in DMSO; absorbance at 570 nm [2] |
| References |
[1]. Different curcuminoids inhibit T-lymphocyte proliferation independently of their radical scavenging activities. Pharm Res. 2008 Aug;25(8):1822-7. [2]. Curcumin and its major metabolites inhibit the inflammatory response induced by lipopolysaccharide: Translocation of nuclear factor-κB as potential target. Mol Med Rep. 2015 Apr;11(4):3087-93. [3]. Comparative antioxidant activities of curcumin and its demethoxy and hydrogenated derivatives. Biol Pharm Bull. 2007 Jan;30(1):74-8. |
| Additional Infomation |
Octahydrocurcumin has been reported in Zingiber officinale with data available. 1. Chemical background: - Octahydrocurcumin is a hydrogenated derivative of curcumin (the major active component of turmeric); its structure differs from curcumin by full saturation of the conjugated double bonds in the central heptadienone chain, increasing its chemical stability and aqueous solubility [2][3] - It is a major in vivo metabolite of curcumin, formed via reduction of curcumin’s double bonds by hepatic enzymes (e.g., aldo-keto reductases) [2] 2. Mechanism of action: - Antioxidant mechanism: Octahydrocurcumin scavenges free radicals (DPPH, ABTS, superoxide anion) via its phenolic hydroxyl groups (electron donation) and inhibits lipid peroxidation by chelating Fe²⁺ (a pro-oxidant metal ion) [3] - Anti-inflammatory mechanism: Octahydrocurcumin suppresses LPS-induced inflammation by inhibiting NF-κB activation—blocking p65 subunit nuclear translocation, thereby reducing transcription of pro-inflammatory cytokines (TNF-α, IL-6) [2] 3. Activity comparison: - Antioxidant activity: Octahydrocurcumin has weaker free radical scavenging activity than curcumin (e.g., DPPH IC₅₀ 8.5 μM vs. curcumin 2.8 μM) but stronger lipid peroxidation inhibition (curcumin IC₅₀ 18.5 μM vs. Octahydrocurcumin 12.3 μM) [3] - Anti-inflammatory activity: Octahydrocurcumin shows ~80% of curcumin’s anti-inflammatory potency (e.g., TNF-α IC₅₀ 15.2 μM vs. curcumin 12.8 μM) but better stability in biological systems [2] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~265.65 mM) Ethanol : ~10 mg/mL (~26.56 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 3.75 mg/mL (9.96 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 37.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 3.75 mg/mL (9.96 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 37.5 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 3.75 mg/mL (9.96 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 37.5 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.6565 mL | 13.2823 mL | 26.5647 mL | |
| 5 mM | 0.5313 mL | 2.6565 mL | 5.3129 mL | |
| 10 mM | 0.2656 mL | 1.3282 mL | 2.6565 mL |