Physicochemical Properties
| Molecular Formula | C29H28NO4F |
| Molecular Weight | 473.53532 |
| Exact Mass | 473.2 |
| CAS # | 200803-37-8 |
| PubChem CID | 9934547 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 667.3±55.0 °C at 760 mmHg |
| Flash Point | 357.4±31.5 °C |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.607 |
| LogP | 5.9 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 35 |
| Complexity | 776 |
| Defined Atom Stereocenter Count | 0 |
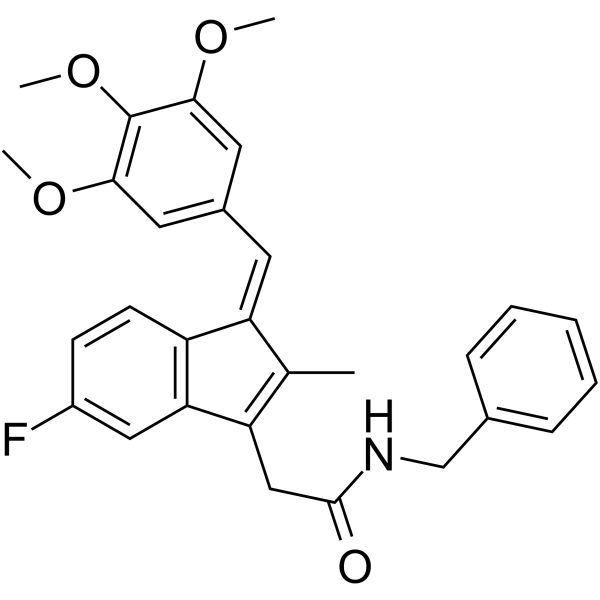
| SMILES | CC\1=C(C2=C(/C1=C\C3=CC(=C(C(=C3)OC)OC)OC)C=CC(=C2)F)CC(=O)NCC4=CC=CC=C4 |
| InChi Key | KATLSKIJHJJAEB-FMCGGJTJSA-N |
| InChi Code | InChI=1S/C29H28FNO4/c1-18-23(12-20-13-26(33-2)29(35-4)27(14-20)34-3)22-11-10-21(30)15-25(22)24(18)16-28(32)31-17-19-8-6-5-7-9-19/h5-15H,16-17H2,1-4H3,(H,31,32)/b23-12- |
| Chemical Name | N-benzyl-2-[(3Z)-6-fluoro-2-methyl-3-[(3,4,5-trimethoxyphenyl)methylidene]inden-1-yl]acetamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Microtubules |
| ln Vitro |
At 0.1 μM, OSIP486823 causes apoptosis and suppresses the development of human colon cancer SW480 cells. In mitotic cells, OSIP-486823 (OSIP486823) prevents spindle formation, produces multinucleated cells, and promotes microtubule depolymerization in interphase cells. Targeting cyclic guanosine 3',5'-monophosphate phosphodiesterase (cGMP-PDE), exisulind and its powerful synthetic derivative OSIP-486823 belong to a novel family of medications known as selective apoptotic antineoplastic medicines. Additionally, OSIP-486823 impairs cell mitosis in human glioma cells, interferes with mitotic spindle function, and disrupts microtubule polymerization [1]. OSIP-486823 is a sulindac derivative with potent antiproliferative activity against human colon cancer cell lines. [1] It exhibits concentration-dependent inhibition of cell viability in HCT116, SW480, and HT29 colon cancer cells, with IC₅₀ values of 0.3 μM, 0.5 μM, and 0.7 μM, respectively, after 72 hours of treatment (MTT assay). [1] The compound arrests colon cancer cells in the mitotic phase (M phase) of the cell cycle: Flow cytometric analysis shows that treatment with OSIP-486823 (0.5 μM for 24 hours) increases the proportion of HCT116 cells in M phase from ~5% (control) to ~38%. [1] It induces microtubule depolymerization: Immunofluorescence staining of α-tubulin reveals that OSIP-486823 (0.3-1.0 μM for 16 hours) disrupts the normal microtubule network, leading to the formation of fragmented microtubule structures, which is consistent with its mechanism of mitotic arrest. [1] OSIP-486823 triggers apoptosis in colon cancer cells: Western blot analysis confirms that treatment with 0.5 μM OSIP-486823 for 48 hours induces cleavage of PARP (a marker of apoptosis) and activation of caspase-3 in HCT116 cells. [1] It inhibits clonogenic potential of colon cancer cells: Colony formation assay shows that OSIP-486823 (0.1-0.5 μM) reduces the number of colonies formed by HCT116 cells by 45%-82% compared to the control group, indicating long-term suppression of cell proliferation. [1] The antiproliferative and mitotic arrest effects of OSIP-486823 are reversible at low concentrations (<0.3 μM); washing out the compound after 24 hours of treatment allows cells to resume normal cell cycle progression. [1] |
| Cell Assay |
MTT antiproliferative assay: HCT116, SW480, and HT29 colon cancer cells are seeded in 96-well plates (5×10³ cells/well) and cultured overnight. OSIP-486823 is added at concentrations ranging from 0.01 μM to 5 μM, and cells are incubated for 72 hours. MTT reagent is added, and after 4 hours of incubation, the absorbance at 570 nm is measured to calculate cell viability and IC₅₀ values. [1] Cell cycle analysis by flow cytometry: HCT116 cells are seeded in 6-well plates and treated with OSIP-486823 (0.1-1.0 μM) for 24 hours. Cells are harvested, fixed with 70% ethanol, stained with propidium iodide (PI) containing RNase, and analyzed by flow cytometry to determine the distribution of cells in G₀/G₁, S, and M phases. [1] Immunofluorescence staining for microtubules: HCT116 cells are seeded on coverslips and treated with OSIP-486823 (0.3-1.0 μM) for 16 hours. Cells are fixed, permeabilized, and incubated with anti-α-tubulin primary antibody and fluorescent secondary antibody. Nuclei are stained with DAPI, and the microtubule network is observed under a confocal microscope to assess microtubule integrity. [1] Western blot assay for apoptotic markers: HCT116 cells are treated with OSIP-486823 (0.25-1.0 μM) for 48 hours. Cell lysates are prepared, proteins (PARP, cleaved PARP, caspase-3, cleaved caspase-3, α-tubulin) are separated by SDS-PAGE, transferred to membranes, and probed with specific antibodies. α-tubulin is used as an internal reference to quantify protein expression. [1] Colony formation assay: HCT116 cells are seeded in 6-well plates (1×10³ cells/well) and cultured for 24 hours. OSIP-486823 (0.1-0.5 μM) is added, and cells are cultured for 14 days. Colonies are fixed, stained with crystal violet, and counted to evaluate the clonogenic potential. [1] Reversibility assay: HCT116 cells are treated with OSIP-486823 (0.1-0.5 μM) for 24 hours, then the medium containing the compound is replaced with fresh medium. Cells are cultured for an additional 48 hours, and cell cycle distribution and viability are analyzed to assess reversibility of the drug effect. [1] |
| References |
[1]. The sulindac derivatives OSI-461, OSIP486823, and OSIP487703 arrest colon cancer cells in mitosis by causing microtubule depolymerization. Mol Cancer Ther. 2006 Jan;5(1):60-7. |
| Additional Infomation |
OSIP-486823 is a synthetic derivative of sulindac (a non-steroidal anti-inflammatory drug), modified to enhance antiproliferative activity against cancer cells. [1] Its mechanism of action involves targeting microtubules and inducing microtubule depolymerization, which disrupts the formation of the mitotic spindle, leading to mitotic arrest at the M phase and subsequent activation of the apoptotic pathway in colon cancer cells. [1] Unlike the parent compound sulindac (which exerts anticancer effects primarily through inhibiting cyclooxygenase enzymes), OSIP-486823 acts via a microtubule-targeting mechanism, representing a novel mode of action for sulindac-derived anticancer agents. [1] OSIP-486823 exhibits higher potency against colon cancer cells compared to sulindac, making it a potential lead compound for the development of targeted therapies for colorectal cancer. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1118 mL | 10.5588 mL | 21.1175 mL | |
| 5 mM | 0.4224 mL | 2.1118 mL | 4.2235 mL | |
| 10 mM | 0.2112 mL | 1.0559 mL | 2.1118 mL |