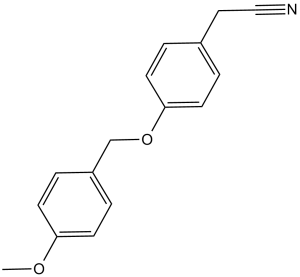
O4I1 (O4-I1; O4I-1; O4I 1) is a novel and potent inducer of Oct3/4 (POU5F1 and Oct4) with potential usefulness as a regenerative medicine. It can enhance Oct3/4 expression.
Physicochemical Properties
| Molecular Formula | C16H15NO2 | |
| Molecular Weight | 253.30 | |
| Exact Mass | 253.11 | |
| Elemental Analysis | C, 75.87; H, 5.97; N, 5.53; O, 12.63 | |
| CAS # | 175135-47-4 | |
| Related CAS # |
|
|
| PubChem CID | 2774690 | |
| Appearance | Light yellow solid powder | |
| Density | 1.1±0.1 g/cm3 | |
| Boiling Point | 424.5±30.0 °C at 760 mmHg | |
| Melting Point | 140ºC | |
| Flash Point | 154.8±17.8 °C | |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C | |
| Index of Refraction | 1.570 | |
| LogP | 2.93 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 19 | |
| Complexity | 293 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O(C1C([H])=C([H])C(C([H])([H])C#N)=C([H])C=1[H])C([H])([H])C1C([H])=C([H])C(=C([H])C=1[H])OC([H])([H])[H] |
|
| InChi Key | QJEJLARVLKHYFW-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C16H15NO2/c1-18-15-6-4-14(5-7-15)12-19-16-8-2-13(3-9-16)10-11-17/h2-9H,10,12H2,1H3 | |
| Chemical Name | 4-[(4-Methoxyphenyl)methoxy]benzeneacetonitrile | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Octamer-binding transcription factor 3/4 (Oct3/4) [1][2] |
||
| ln Vitro |
In vitro activity: O4I1 enhances Oct3/4 gene expression by approximately 2.5- and 4-fold at 10 μM and 20 μM, respectively, increases Oct3/4 protein levels, and promotes Oct3/4 mediated transcriptional activation. Cell Assay: When treated the synthesized cDNA from RNA isolated from the HEK293 cells with 10 µM and 20 µM O4I1 for 72 hrs, the Oct3/4 mRNA levels increased about 2.5- and 4-fold respectively. In human adult primary fibroblast (HF) and human neonatal foreskin fibroblast (HFF) cell lines, treatment with 10 µM O4I1 increased the gene and protein expression level of Oct3/4 after 72 hrs incubation in HF cells and 7 days in HFF cells. The derivatives of O4I1 such as compound 2, showed a time-dependent activation of Oct3/4 and markedly elevated the efficiency of Oct3/4 induction. In HEK293 cells transfected with Oct3/4 promoter-luciferase reporter plasmid, O4I1 (0.5-20 μM) dose-dependently activated Oct3/4 promoter activity, achieving a maximal 5.3-fold increase at 10 μM compared to vehicle control. This demonstrated potent induction of Oct3/4 transcriptional activity[1] - In P19 embryonic carcinoma cells and mouse embryonic stem cells (mESCs), O4I1 (1-15 μM) upregulated Oct3/4 mRNA expression by 4.0-6.2-fold (RT-PCR) and protein levels by 3.2-4.5-fold (Western blot) at 8 μM. It also significantly increased the expression of pluripotency-associated markers Sox2 (2.1-2.8-fold) and Nanog (1.8-2.5-fold)[1] - In retinoic acid-induced differentiated mESCs, O4I1 (8 μM) reversed Oct3/4 downregulation, restoring its mRNA and protein levels to ~80% of undifferentiated mESC levels. It also partially rescued the expression of Sox2 and Nanog, suggesting pluripotency-retaining potential[1] - In human somatic cells (fibroblasts), O4I1 (10 μM) combined with other reprogramming factors enhanced the efficiency of induced pluripotent stem cell (iPSC) formation by 3.0-fold compared to factors alone, as confirmed by alkaline phosphatase staining and Oct3/4 immunofluorescence[2] |
||
| ln Vivo |
|
||
| Cell Assay |
Oct3/4 promoter activation assay: HEK293 cells were seeded in 96-well plates and co-transfected with Oct3/4 promoter-luciferase plasmid and Renilla luciferase plasmid (internal control). After 24 hours, O4I1 (0.1 μM, 0.5 μM, 1 μM, 5 μM, 10 μM, 20 μM) was added, and cells were cultured for another 48 hours. Dual-luciferase assay was performed to measure relative luciferase activity (firefly/Renilla), reflecting Oct3/4 promoter activation[1] - Pluripotency marker expression assay: P19 cells and mESCs were seeded in 6-well plates and treated with O4I1 (1 μM, 4 μM, 8 μM, 15 μM) for 72 hours. Total RNA was extracted for RT-PCR analysis of Oct3/4, Sox2, and Nanog mRNA. Cell lysates were prepared for Western blot detection of corresponding proteins, with β-actin as the loading control[1] - Differentiated mESC rescue assay: mESCs were induced to differentiate with retinoic acid for 5 days, and O4I1 (8 μM) was added from day 2 to day 5. Oct3/4, Sox2, and Nanog mRNA/protein levels were detected by RT-PCR and Western blot, and compared to undifferentiated and vehicle-treated differentiated controls[1] - iPSC formation assay: Human fibroblasts were seeded in 6-well plates and transduced with reprogramming factor vectors. O4I1 (10 μM) was added to the culture medium and refreshed every 2 days. After 21 days, alkaline phosphatase staining was performed to count iPSC colonies, and immunofluorescence staining with Oct3/4 antibody was used to confirm pluripotency[2] |
||
| Animal Protocol |
|
||
| References |
[1]. Identification of 2-[4-[(4-Methoxyphenyl)methoxy]-phenyl]acetonitrile and Derivatives as Potent Oct3/4 Inducers. J Med Chem. 2015 Jun 25;58(12):4976-4983. [2]. Method for activating OCT4 for induction of pluripotent stem cell formation. From PCT Int. Appl. (2014), WO 2014000814 A1 20140103. |
||
| Additional Infomation |
O4I1 is a derivative of 2-[4-[(4-methoxyphenyl)methoxy]-phenyl]acetonitrile, identified as a potent inducer of the pluripotency transcription factor Oct3/4[1] - Its core mechanism involves binding to the Oct3/4 promoter region to enhance transcriptional activity, thereby upregulating Oct3/4 expression and promoting cross-activation of other pluripotency markers (Sox2, Nanog)[1] - It exhibits activity in multiple cell models (HEK293, P19, mESCs, human fibroblasts) and enhances somatic cell reprogramming efficiency, supporting its application as a tool compound in stem cell research[1][2] - The compound’s structure, featuring a methoxyphenylmethoxy-phenylacetonitrile scaffold, is critical for its Oct3/4-inducing activity, providing a template for optimizing pluripotency modulators[1] - It is covered in a patent for methods of inducing pluripotent stem cell formation by activating Oct3/4, highlighting its potential for regenerative medicine-related research[2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.9479 mL | 19.7394 mL | 39.4789 mL | |
| 5 mM | 0.7896 mL | 3.9479 mL | 7.8958 mL | |
| 10 mM | 0.3948 mL | 1.9739 mL | 3.9479 mL |