Nrf2 activator-4 is a novel potent and orally bioactive Nrf2 activator (EC50 = 0.63 µM) with anti-AD effects. It suppresses reactive oxygen species against oxidative stress in microglia. It has the potential for teating learning and memory impairment in a scopolamine-induced mouse model. Alzheimer's disease is a common neurodegenerative disease characterized by progressive degeneration and neuronal cell death, resulting in neural network dysfunction. As the underlying mechanisms, oxidative damage and neuroinflammation have been reported to contribute to the onset and deterioration of Alzheimer's disease. The nuclear factor E2-related factor 2-antioxidant responsive element signaling pathway is a pivotal cellular defense mechanism against oxidative stress. Nrf2, a transcription factor, regulates the cellular redox balance and is primarily involved in anti-inflammatory responses.
Physicochemical Properties
| Molecular Formula | C23H24CLF3N2O3 |
| Molecular Weight | 468.896475791931 |
| Exact Mass | 468.142 |
| CAS # | 2383016-68-8 |
| PubChem CID | 155527528 |
| Appearance | Light yellow to yellow solid powder |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 32 |
| Complexity | 637 |
| Defined Atom Stereocenter Count | 0 |
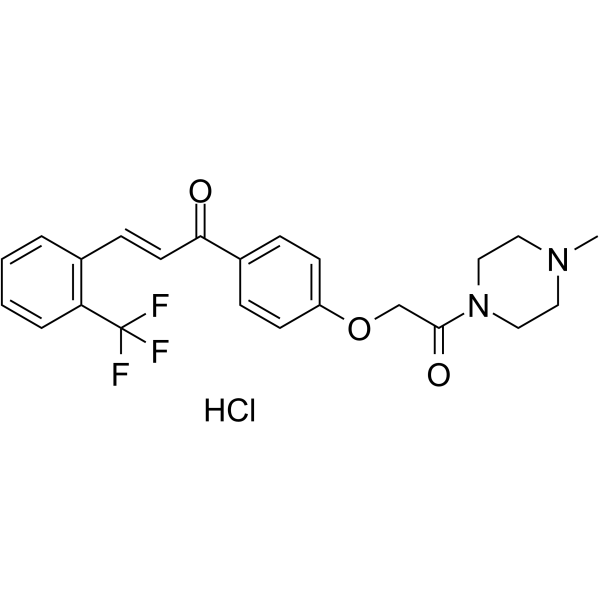
| SMILES | C(/C1C=CC=CC=1C(F)(F)F)=C\C(C1C=CC(OCC(N2CCN(C)CC2)=O)=CC=1)=O.Cl |
| InChi Key | MLBFDNAGDZEBHA-YGCVIUNWSA-N |
| InChi Code | InChI=1S/C23H23F3N2O3.ClH/c1-27-12-14-28(15-13-27)22(30)16-31-19-9-6-18(7-10-19)21(29)11-8-17-4-2-3-5-20(17)23(24,25)26;/h2-11H,12-16H2,1H3;1H/b11-8+; |
| Chemical Name | (E)-1-[4-[2-(4-methylpiperazin-1-yl)-2-oxoethoxy]phenyl]-3-[2-(trifluoromethyl)phenyl]prop-2-en-1-one;hydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| References |
[1]. A novel chalcone derivative as Nrf2 activator attenuates learning and memory impairment in a scopolamine-induced mouse model. Eur J Med Chem. 2020 Jan 1;185:111777. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~213.27 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.33 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.33 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.33 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1327 mL | 10.6633 mL | 21.3265 mL | |
| 5 mM | 0.4265 mL | 2.1327 mL | 4.2653 mL | |
| 10 mM | 0.2133 mL | 1.0663 mL | 2.1327 mL |