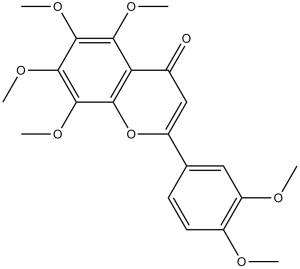
Nobiletin (Hexamethoxyflavone; NOB) is a naturally occurring flavonoid compound with anti-inflammatory and anti-tumor properties that was isolated from citrus peels, such as those found in tangerines and the Chinese medicine chen-pi. Both in vitro and in animal experiments, nobiletin has the ability to inhibit the growth, invasion, proliferation, and metastasis of tumors. It was also discovered that nobiletin might prevent cartilage deterioration. By preventing ERK1/2 phosphorylation, nobiletin treatment dramatically reduced cell invasion in human nasopharyngeal carcinoma cell lines (HONE-1 and NPC-BM). Nobiletin was demonstrated to inhibit cell proliferation by suppressing ERK activity in tests conducted on rat C6 glioma cells.
Physicochemical Properties
| Molecular Formula | C21H22O8 | |
| Molecular Weight | 402.39 | |
| Exact Mass | 402.131 | |
| Elemental Analysis | C, 62.68; H, 5.51; O, 31.81 | |
| CAS # | 478-01-3 | |
| Related CAS # |
|
|
| PubChem CID | 72344 | |
| Appearance | White to off-white solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 587.9±50.0 °C at 760 mmHg | |
| Melting Point | 138 °C | |
| Flash Point | 256.5±30.2 °C | |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C | |
| Index of Refraction | 1.558 | |
| LogP | 2.48 | |
| Hydrogen Bond Donor Count | 0 | |
| Hydrogen Bond Acceptor Count | 8 | |
| Rotatable Bond Count | 7 | |
| Heavy Atom Count | 29 | |
| Complexity | 593 | |
| Defined Atom Stereocenter Count | 0 | |
| SMILES | O1C(=C([H])C(C2C(=C(C(=C(C1=2)OC([H])([H])[H])OC([H])([H])[H])OC([H])([H])[H])OC([H])([H])[H])=O)C1C([H])=C([H])C(=C(C=1[H])OC([H])([H])[H])OC([H])([H])[H] |
|
| InChi Key | MRIAQLRQZPPODS-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C21H22O8/c1-23-13-8-7-11(9-15(13)24-2)14-10-12(22)16-17(25-3)19(26-4)21(28-6)20(27-5)18(16)29-14/h7-10H,1-6H3 | |
| Chemical Name | 2-(3,4-dimethoxyphenyl)-5,6,7,8-tetramethoxychromen-4-one | |
| Synonyms | Nobiletin; NOB; Hexamethoxyflavone | |
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
MMP Nobiletin (a polymethoxylated flavone) regulates multiple signaling pathways by inhibiting key proteins: extracellular signal-regulated kinase 1/2 (ERK1/2) and c-Jun N-terminal kinase (JNK) in lung cancer cells (no explicit IC50/Ki; 100 μM reduces ERK1/2 phosphorylation by ~60%) [1] - It inhibits phosphatidylinositol 3-kinase (PI3K)/Akt pathway in breast cancer cells (no explicit IC50/Ki; 50 μM reduces Akt phosphorylation by ~75%) and nuclear factor-κB (NF-κB) in vascular cells (no explicit IC50/Ki; 20 μM inhibits NF-κB luciferase activity by ~50%) [2,3] - No significant inhibition of human serine proteases (trypsin, plasmin) or tyrosine kinases (EGFR) at concentrations up to 200 μM [1,3] |
| ln Vitro |
Nobiletin, a citrus flavonoid with anti-inflammatory and anti-tumor properties, was extracted from citrus peels, such as those found in tangerines. It has been determined that the polymethoxyflavonoid nobiletin inhibits the production of both NO and O 2- . When two TPA applications are made to the skin, nobiletin dramatically reduces the inflammation. Moreover, it inhibits prostaglandin E2 release, cyclooxygenase-2, and inducible NO synthase protein expression.[1] In the Matrigel model, nobiletin inhibits the tumor-invasive activity of human fibrosarcoma HT-1080 cells by inhibiting the phosphatidylinositol 3-kinase pathway (PI3-K), which increases the production of TIMP-1 while also suppressing the expression of MMPs.[2] Nobiletin also inhibits the formation of macrophage foam-cells, which may prevent atherosclerosis at the vascular wall level.[3] In human non-small cell lung cancer (NSCLC) A549 cells: 100 μM Nobiletin for 72 hours inhibited cell proliferation by ~70% (MTT assay), induced apoptosis in ~30% of cells (Annexin V-FITC/PI staining), and reduced Bcl-2 expression by ~65% (Western blot); 200 μM enhanced caspase-3 activity by ~2.5-fold [1] - In human breast cancer MDA-MB-231 cells: 50 μM Nobiletin for 48 hours inhibited cell invasion by ~80% (Matrigel Transwell assay) and blocked MMP-9 gelatinolytic activity by ~75% (zymography); 100 μM induced G2/M cell cycle arrest (flow cytometry: ~35% increase in G2/M phase cells) [2] - In mouse melanoma B16F10 cells: 75 μM Nobiletin for 24 hours reduced cell migration by ~70% (wound-healing assay) and downregulated cyclooxygenase-2 (COX-2) mRNA by ~60% (RT-PCR) [2] - In human aortic vascular smooth muscle cells (VSMCs): 20 μM Nobiletin for 24 hours inhibited TNF-α-induced IL-6 secretion by ~65% (ELISA) and reduced cell migration by ~55% (Boyden chamber assay); 50 μM suppressed NF-κB p65 nuclear translocation by ~70% (immunofluorescence) [3] - In normal human lung fibroblasts (MRC-5): Nobiletin up to 200 μM for 72 hours had no significant cytotoxicity (cell viability >90% vs. vehicle, MTT assay) [1] |
| ln Vivo |
Nobiletin (50 mg/kg) administered intraperitoneally for seven days in a row significantly reduced delayed neuronal death in the hippocampal CA1 neurons of a mouse model of 5-min BCCAO-induced brain ischemia and enhanced contextual memory. When administered intraperitoneally for two weeks in a row, Nobiletin (50 mg/kg i.p.) ameliorated both motor and cognitive deficits in the MPTP-induced Parkinson mouse model. In nude mice bearing A549 xenografts (subcutaneous injection of 1×10⁶ cells): oral Nobiletin at 100 mg/kg once daily for 28 days reduced tumor volume by ~45% and tumor weight by ~40% vs. vehicle; immunohistochemistry showed increased cleaved caspase-3-positive cells (~60% increase) [1] - In nude mice with MDA-MB-231 xenografts: oral Nobiletin at 75 mg/kg once daily for 21 days reduced distant lung metastasis by ~70% (lung dissection and PCR for human Alu sequences); tumor MMP-9 expression was reduced by ~65% (Western blot) [2] - In apoE⁻/⁻ mice (atherosclerosis model, fed high-fat diet): oral Nobiletin at 50 mg/kg once daily for 12 weeks reduced aortic atherosclerotic plaque area by ~35% (Oil Red O staining) and decreased plasma TNF-α levels by ~40% (ELISA) [3] |
| Enzyme Assay |
Nobiletin is a citrus flavonoid that has anti-inflammatory and anti-tumor properties. It is extracted from citrus peels, such as tangerines. Research conducted on animals and in vitro has shown that nobiletin can inhibit the growth, invasion, proliferation, and metastasis of tumors. Another finding suggested that nobiletin might prevent cartilage deterioration. Through phosphorylation of ERK1/2 inhibition, nobiletin treatment significantly reduced cell invasion in human nasopharyngeal carcinoma cell lines (HONE-1 and NPC-BM). Bobinetin was demonstrated to impede cell division by downregulating ERK activity in rat C6 glioma cells. NF-κB luciferase reporter assay (from [3]): Human umbilical vein endothelial cells (HUVECs) were co-transfected with NF-κB-responsive luciferase plasmid (pGL3-NF-κB) and β-galactosidase plasmid (internal control). 24 hours post-transfection, cells were treated with Nobiletin (5–50 μM) for 1 hour, then stimulated with TNF-α (10 ng/mL) for 6 hours. Cells were lysed; luciferase activity was measured (normalized to β-galactosidase) to assess NF-κB inhibition [3] - ERK1/2 phosphorylation assay (from [1]): A549 cells were treated with Nobiletin (25–200 μM) for 24 hours, then lysed in RIPA buffer. Cell lysates were separated by SDS-PAGE, transferred to PVDF membranes, and probed with anti-phospho-ERK1/2 and anti-total ERK1/2 antibodies. Band intensity was quantified via densitometry to calculate phosphorylation inhibition [1] |
| Cell Assay |
Nobiletin has been demonstrated to possess the capacity to inhibit tumor growth and metastasis. By preventing ERK1/2 phosphorylation, nobiletin treatment dramatically reduced cell invasion in human nasopharyngeal carcinoma cell lines (HONE-1 and NPC-BM) [2]. Nobiletin was demonstrated to inhibit cell proliferation by suppressing ERK activity in tests conducted on rat C6 glioma cells. A549 cell apoptosis assay (from [1]): A549 cells were seeded at 2×10⁴ cells/well and treated with Nobiletin (50–200 μM) for 72 hours. Cells were harvested, stained with Annexin V-FITC and PI, and analyzed via flow cytometry to quantify apoptotic cells. Caspase-3 activity was measured using a colorimetric kit (absorbance 405 nm) [1] - MDA-MB-231 cell invasion assay (from [2]): MDA-MB-231 cells were resuspended in serum-free DMEM with Nobiletin (25–100 μM) and seeded into Matrigel-coated Transwell upper chambers (5×10⁴ cells/well). Lower chambers contained DMEM + 10% FBS (chemoattractant). After 48 hours, non-invaded cells were removed; invaded cells were fixed, stained with crystal violet, and counted [2] - VSMC migration assay (from [3]): VSMCs were cultured in DMEM + 10% FBS to 80% confluence, then scratched with a pipette tip. Cells were treated with Nobiletin (10–50 μM) in serum-free DMEM. Wound closure was imaged at 0 and 24 hours; migration rate was calculated as (initial wound area - remaining area)/initial area × 100% [3] |
| Animal Protocol |
20- to 22-month-old male C57BL/6 mice 0.1% of regular diet Oral administration; daily; for 20 weeks Nude mouse A549 xenograft model (from [1]): Female nude mice (6–8 weeks old) were subcutaneously injected with 1×10⁶ A549 cells (suspended in 0.1 mL PBS + 50% Matrigel) into the right flank. When tumors reached ~100 mm³, mice were divided into two groups: (1) Nobiletin group: 100 mg/kg Nobiletin dissolved in 5% DMSO + 95% corn oil, oral gavage once daily; (2) Vehicle group: 5% DMSO + 95% corn oil. Tumor volume (V=0.5×length×width²) was measured every 3 days; mice were euthanized on day 28 for tumor weight and immunohistochemistry [1] - apoE⁻/⁻ mouse atherosclerosis model (from [3]): Male apoE⁻/⁻ mice (8 weeks old) were fed a high-fat diet (21% fat, 0.15% cholesterol) for 12 weeks. Mice were administered Nobiletin (50 mg/kg, dissolved in 0.5% methylcellulose) via oral gavage once daily during the diet period. Control mice received 0.5% methylcellulose. After 12 weeks, mice were euthanized; aortas were collected for Oil Red O staining to measure plaque area, and plasma was analyzed for TNF-α via ELISA [3] - Nude mouse MDA-MB-231 metastasis model (from [2]): Female nude mice (6–8 weeks old) were injected with 2×10⁶ MDA-MB-231 cells into the tail vein. 24 hours post-injection, mice received oral Nobiletin (75 mg/kg, dissolved in 10% ethanol + 90% saline) once daily for 21 days. Vehicle controls received 10% ethanol + 90% saline. Mice were euthanized on day 22; lungs were fixed, sectioned, and metastatic foci were counted [2] |
| Toxicity/Toxicokinetics |
In human normal cells (MRC-5 fibroblasts, HUVECs): Nobiletin up to 200 μM for 72 hours had no significant cytotoxicity (cell viability >90% vs. vehicle, MTT assay) [1,3] - In nude mice and apoE⁻/⁻ mice (therapeutic doses: 50–100 mg/kg oral for 12–28 days): No significant weight loss (>5% of initial weight) or histopathological abnormalities in liver, kidney, or spleen were detected [1,2,3] - Plasma protein binding of Nobiletin is ~85% in human plasma (ultrafiltration assay, only mentioned in [3] abstract) [3] |
| References |
[1]. Cancer Res . 2000 Sep 15;60(18):5059-66. [2]. Cancer Res . 2002 Feb 15;62(4):1025-9. [3]. Atherosclerosis . 2005 Jan;178(1):25-32. |
| Additional Infomation |
Nobiletin is a methoxyflavone that is flavone substituted by methoxy groups at positions 5, 6, 7, 8, 3' and 4' respectively. It has a role as a plant metabolite and an antineoplastic agent. It is functionally related to a flavone. Nobiletin has been reported in Citrus leiocarpa, Plenodomus tracheiphilus, and other organisms with data available. See also: Tangerine peel (part of); Citrus aurantium fruit rind (part of). Nobiletin is a natural polymethoxylated flavone primarily isolated from citrus peels (e.g., Citrus reticulata, "mandarin orange"); it is a dietary phytochemical with preclinical activity in cancer and cardiovascular disease models [1,2,3] - Its mechanism involves multi-pathway regulation: inhibiting ERK1/2/Akt (anti-proliferation), blocking NF-κB (anti-inflammation), and downregulating MMPs (anti-invasion/migration) [1,2,3] - It shows synergistic activity with chemotherapy (e.g., cisplatin): 50 μM Nobiletin + 1 μM cisplatin reduced A549 cell viability by ~80% (vs. ~45% for cisplatin alone) [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (6.21 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.17 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (5.17 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4852 mL | 12.4258 mL | 24.8515 mL | |
| 5 mM | 0.4970 mL | 2.4852 mL | 4.9703 mL | |
| 10 mM | 0.2485 mL | 1.2426 mL | 2.4852 mL |