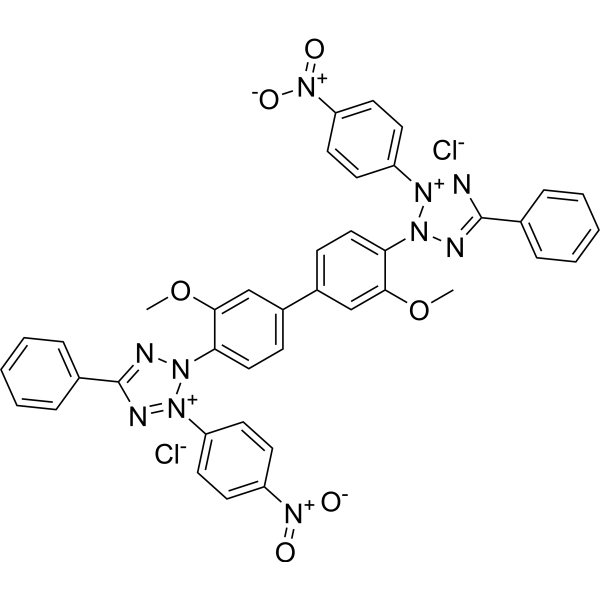
Physicochemical Properties
| Molecular Formula | C40H30CL2N10O6 |
| Molecular Weight | 817.6356 |
| Exact Mass | 816.172 |
| CAS # | 298-83-9 |
| PubChem CID | 9281 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.5521 (rough estimate) |
| Melting Point | 200ºC |
| Index of Refraction | 1.7350 (estimate) |
| LogP | 1.29 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 9 |
| Heavy Atom Count | 58 |
| Complexity | 1180 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | FSVCQIDHPKZJSO-UHFFFAOYSA-L |
| InChi Code | InChI=1S/C40H30N10O6.2ClH/c1-55-37-25-29(13-23-35(37)47-43-39(27-9-5-3-6-10-27)41-45(47)31-15-19-33(20-16-31)49(51)52)30-14-24-36(38(26-30)56-2)48-44-40(28-11-7-4-8-12-28)42-46(48)32-17-21-34(22-18-32)50(53)54;;/h3-26H,1-2H3;2*1H/q+2;;/p-2 |
| Chemical Name | 2-[2-methoxy-4-[3-methoxy-4-[3-(4-nitrophenyl)-5-phenyltetrazol-2-ium-2-yl]phenyl]phenyl]-3-(4-nitrophenyl)-5-phenyltetrazol-2-ium;dichloride |
| Synonyms | 298-83-9; Nitrotetrazolium Blue chloride; Nitroblue tetrazolium chloride; Nitro Blue tetrazolium chloride; nitro bt; Nitro Blue tetrazolium; NBT; Nitrotetrazolium Blue; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Fluorescent dye reagent; dehydrogenase |
| ln Vitro |
Staining protocol for NBT and BCIP 1. Materials Dimethylformamide (DMF). Nitro blue tetrazolium chloride (NBT) stock solution: Dissolve 0.5 g of NBT in 10 mL of 70% DMF (in water). BCIP stock solution: Dissolve 0.5 g of BCIP in 10 mL of 100% DMF. Substrate Buffer: 100 mM diethanolamine buffer (pH 9.5), containing 100 mM NaCl and 5 mM MgCl2. 2. Method 2.1 When preparing NBT/BCIP solution, add 66 µ L of NBT stock solution to 10 mL of Substrate Buffer and mix thoroughly. Add 33 μ L of BCIP stock solution to the solution. Use NBT/BCIP solution within 1 hour. 2.2 Place the imprint (or blot) in a suitable container and add NBT/BCIP solution. Take 10 mL of solution from a 15 × 15cm2 membrane. Stir at room temperature and incubate the imprint (or blot) until the band darkens appropriately. Usually, it takes about 30 minutes. 2.3 Remove the NBT/BCIP solution and rinse the imprint with water to terminate the reaction. |
| ln Vivo |
Materials and methods[1] Two imaging systems were used for the excitation and detection of the NBT/BCIP stain. The first, a Zeiss LSM 510 laser scanning confocal microscope with a 633-nm helium-neon (HeNe) laser and a 650-nm long pass emission filter, was used for detection of the NBT/BCIP signal. The long pass filter collects all emitted light above 650 nm. The second was a Zeiss Axio Imager Z1 system with an Hg arc lamp source, a 645-685 nm excitation band pass filter, and a 760-nm long pass emission filter coupled to an Ocean Optics HR2000 charged-couple device (CCD) spectrometer via a 200-µm core optical fiber mounted at the focal plane of the camera port that was used to determine the emission spectra. The optical fiber only collects the light falling on its core, which for the Zeiss EC Plan Neofluar® 40×/1.3 numerical aperture (NA) objective used in this work defines a 5-µm diameter area of the sample. Spatial correlation between the image and the spectra (fiber core) was established using an x-y translator in the optical fiber mount to align it to the eyepiece crosshair reticle.[1] The NBT/BCIP staining protocol was performed as follows: zebrafish and lamprey whole-mount embryos incubated with DIG-labeled riboprobes were immunolabeled with a sheep anti-DIG antibody conjugated to AP (1:3000). They were subsequently processed with NBT/BCIP solution according to the manufacturer's instructions to obtain the dark purple NBT/BCIP stain. Immunohistochemistry following NBT/BCIP staining was performed as follows: anti-green fluorescent protein (GFP) antibody (1:500) and anti--catenin (1:500) were incubated with embryos overnight at 4°C, followed by incubation with an anti-rabbit or anti-mouse Alexa-conjugated secondary (1:200). Stained embryos were embedded in 4% NuSieve® GTG low melting agarose and cut into 100-or 200-µm sections with a Vibratome® 1000 Sectioning System for imaging. [1] For a projected view of whole-mount samples (Figure 1F), embryos were embedded in 4% low melting agarose with the dorsal side of the embryo positioned parallel to the plane of imaging. Z-slices were imaged as described previously at 1.5-µm intervals. A subset of the Z-slices that contained the stained tissue was then projected along the y-axis using Zeiss LSM software.[1] NBT-DF was prepared by dissolving 20 mg NBT dichloride in 25 mL absolute ethanol. A solution of sodium borohydride was added with stirring (100 mg in 10 mL absolute ethanol). A deep purple precipitate formed and was allowed to stand overnight. The precipitate was filtered, washed with absolute ethanol, then washed with acetone. The acetone wash was continued until the wash was clear (approximately 30 mL) to remove the monoformazan. The resulting solid was bluish-black. The precipitate was soluble in nitrobenzene, and crystallization from nitrobenzene gave purple needles. The visible/near infrared (VIS/NIR) absorption spectra of the solution of this product in nitrobenzene is consistent with previously reported spectra (data not shown). |
| Enzyme Assay | In situ hybridization techniques typically employ chromogenic staining by enzymatic amplification to detect domains of gene expression. We demonstrate the previously unreported near infrared (NIR) fluorescence of the dark purple stain formed from the commonly used chromogens, nitro blue tetrazolium (NBT) and 5-bromo-4-chloro-3-indolyl phosphate (BCIP). The solid reaction product has significant fluorescence that enables the use of confocal microscopy to generate high-resolution three-dimensional (3-D) imaging of gene expression[1]. |
| Cell Assay |
For fluorescent in situ hybridization (FISH) with NBT/BCIP chromogenic stain, cells were first fixed with an appropriate fixative and permeabilized to enhance probe penetration. The specific nucleic acid probe was hybridized with the target sequence in the cells under optimized temperature and time conditions. After hybridization, unbound probes were removed by stringency washing. The cells were then incubated with a conjugate of alkaline phosphatase (AP) and secondary reagent, which binds to the hybridized probe. Subsequently, the chromogenic substrate mixture containing Nitro blue tetrazolium chloride and 5-bromo-4-chloro-3-indolyl phosphate (BCIP) was added to the cells. The reaction was incubated in the dark at room temperature for a specified time to allow the AP-catalyzed conversion of Nitro blue tetrazolium chloride/BCIP into an insoluble purple-blue precipitate at the target sequence site. Finally, the cells were rinsed to stop the reaction and observed under a light microscope to visualize the hybridized signals [1] |
| References |
[1]. Fluorescent in situ hybridization employing the conventional NBT/BCIP chromogenic stain. Biotechniques. 2007 Jun;42(6):756-9. |
| Additional Infomation |
Nitro blue tetrazolium dichloride is an organic chloride salt having nitro blue tetrazolium(2+) as the counterion. It contains a nitro blue tetrazolium(2+). Colorless to yellow dye that is reducible to blue or black formazan crystals by certain cells; formerly used to distinguish between nonbacterial and bacterial diseases, the latter causing neutrophils to reduce the dye; used to confirm diagnosis of chronic granulomatous disease. Nitro blue tetrazolium chloride (NBT) is used to distinguish between nonbacterial and bacterial diseases, the latter causing neutrophils to reduce the dye; used to confirm diagnosis of chronic granulomatous disease.It is a microscopic stain used to detect dehydrogenases and alkaline phosphatase. In summary, we report the heretofore undescribed fluorescence of the NBT-DF/BCI reaction product derived from NBT/BCIP chromogenic staining protocols. We have demonstrated that it can be employed as a NIR fluorescence label that permits cellular resolution in whole-mount in situ hybridization studies. Although both components of the reaction product exhibit fluorescence. NBT-DF is the major NIR fluorophore in the specimens. The red excitation and NIR emission are ideal for use in whole-mounted specimens, as longer wavelengths are less scattered by tissue and autofluorescence is much lower in this wavelength range in most specimens; furthermore, the emission is easy to separate spectrally from the autofluorescence of the matrix tissue and from most fluorescent labels. Thus, the NIR emission will facilitate multi-label NBT/BCIP staining protocols in combination with visible fluorophores. We show here that current NBT/BCIP in situ hybridization protocols coupled to optical sectioning techniques result in single cell resolution without the development of new protocols for fluorescent in situ hybridization methods.[1] Nitro blue tetrazolium chloride is a chromogenic substrate commonly used in immunohistochemical and in situ hybridization assays, paired with BCIP [1] It acts as an electron acceptor in the reaction catalyzed by alkaline phosphatase: the enzyme hydrolyzes BCIP, generating a product that reduces Nitro blue tetrazolium chloride to form an insoluble purple-blue formazan precipitate, which enables the visualization of target molecules or sequences [1] In the reported FISH assay, it is used to detect the hybridization signal, providing a clear and stable chromogenic outcome for the identification of target nucleic acid sequences in cells [1] |
Solubility Data
| Solubility (In Vitro) |
70%DMF : 100 mg/mL (~122.30 mM) DMSO : ~33.33 mg/mL (~40.76 mM) H2O : ~8 mg/mL (~9.78 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (3.06 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (3.06 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (3.06 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.2230 mL | 6.1152 mL | 12.2303 mL | |
| 5 mM | 0.2446 mL | 1.2230 mL | 2.4461 mL | |
| 10 mM | 0.1223 mL | 0.6115 mL | 1.2230 mL |