Physicochemical Properties
| Molecular Formula | C20H25FN4O |
| Molecular Weight | 356.44 |
| Exact Mass | 356.201 |
| CAS # | 27367-90-4 |
| Related CAS # | 34426-53-4 (tri-hydrochloride) |
| PubChem CID | 71919 |
| Appearance | White to off-white solid powder |
| LogP | 3.129 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 26 |
| Complexity | 434 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | RSKQGBFMNPDPLR-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H25FN4O/c1-16(23-20(26)17-3-2-9-22-15-17)8-10-24-11-13-25(14-12-24)19-6-4-18(21)5-7-19/h2-7,9,15-16H,8,10-14H2,1H3,(H,23,26) |
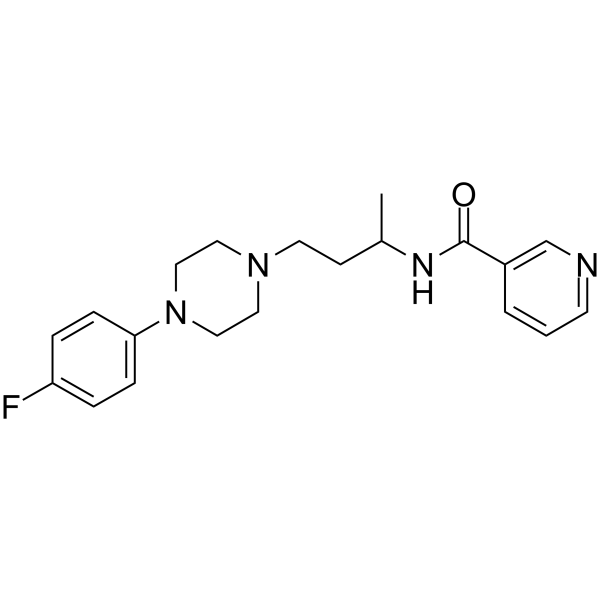
| Chemical Name | N-[4-[4-(4-fluorophenyl)piperazin-1-yl]butan-2-yl]pyridine-3-carboxamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Niaprazine has minimal affinity for D2, α2, β, H1, and mAch receptors as well as the vesicular monoamine transporter. 5-HT1A and 5-HT1B binding sites do not recognize niaprazine well, while α1 (Ki = 77 nM) and 5-HT2 (Ki = 25 nM) binding sites do[2]. Niaprazine, in particular, the (+)stereoisomer, has a higher affinity for these sites. |
| ln Vivo | Rat brain 5-hydroxyindole acetic acid (5-HIAA) concentrations rise 30 minutes after niaprazine (60 mg/kg; ip; once) administration and fall 3–8 hours later. Moreover, niaprazine causes a transient reduction in dopamine (DA) and noradrenaline (NA) in rat brains[3]. |
| Animal Protocol |
Animal/Disease Models: Male SD (Sprague-Dawley) rats (150-200 g)[3] Doses: 60 mg/kg Route of Administration: intraperitoneal (ip) injection; once Experimental Results: Increased rat brain 5-hydroxyindole acetic acid (5-HIAA) concentrations 30 min after treatment, and decreased them at 3-8 hr after treatment. |
| References |
[1]. Molecular pharmacology of niaprazine. Prog Neuropsychopharmacol Biol Psychiatry. 1988;12(6):989-1001. [2]. Niaprazine in the treatment of autistic disorder. J Child Neurol. 1999 Aug;14(8):547-50. [3]. The effect of niaprazine on the turnover of 5-hydroxytryptamine in the rat brain. Neuropharmacology. 1982 Feb;21(2):163-9. |
| Additional Infomation |
Niaprazine is a member of piperazines. Niaprazine is a selective brain catecholamine depletor. |
Solubility Data
| Solubility (In Vitro) | DMSO: 100 mg/mL (280.55 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 0.83 mg/mL (2.33 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 8.3 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 0.83 mg/mL (2.33 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), suspension solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 8.3 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 0.83 mg/mL (2.33 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 8.3 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8055 mL | 14.0276 mL | 28.0552 mL | |
| 5 mM | 0.5611 mL | 2.8055 mL | 5.6110 mL | |
| 10 mM | 0.2806 mL | 1.4028 mL | 2.8055 mL |