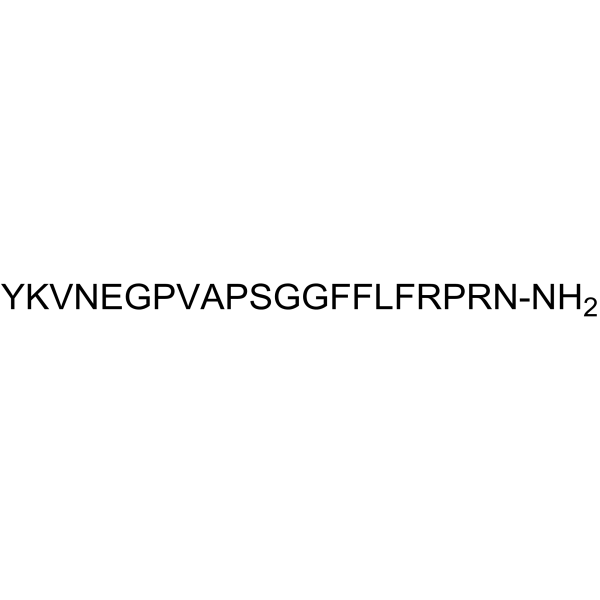Physicochemical Properties
| Molecular Formula | C124H180N34O31 |
| Molecular Weight | 2642.97000 |
| Exact Mass | 2641.36 |
| CAS # | 117505-80-3 |
| Related CAS # | Neuromedin U, rat TFA |
| PubChem CID | 71311710 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 4.233 |
| Hydrogen Bond Donor Count | 35 |
| Hydrogen Bond Acceptor Count | 35 |
| Rotatable Bond Count | 81 |
| Heavy Atom Count | 189 |
| Complexity | 6040 |
| Defined Atom Stereocenter Count | 20 |
| SMILES | NCCCC[C@@H](C(N[C@H](C(N[C@H](C(N[C@H](C(N[C@H](C(N[C@H](C(NCC(N1CCC[C@H]1C(N[C@H](C(N[C@H](C(N1CCC[C@H]1C(N[C@H](C(NCC(NCC(N[C@H](C(N[C@H](C(N[C@H](C(N[C@H](C(N[C@H](C(N1CCC[C@H]1C(N[C@H](C(N[C@H](C(=O)N)CC(=O)N)=O)CCCNC(=N)N)=O)=O)CCCNC(=N)N)=O)CC1=CC=CC=C1)=O)CC(C)C)=O)CC1=CC=CC=C1)=O)CC1=CC=CC=C1)=O)=O)=O)CO)=O)=O)C)=O)C(C)C)=O)=O)=O)CCC(=O)N)=O)CC1=CC=C(O)C=C1)=O)CCC(=O)O)=O)CC(=O)N)=O)C(C)C)=O)NC([C@H](CC1=CC=C(O)C=C1)N)=O |
| InChi Key | YYDRKWCPVLDJJD-XOXVDEAXSA-N |
| InChi Code | InChI=1S/C124H180N34O31/c1-66(2)54-84(110(177)150-86(57-71-26-13-9-14-27-71)113(180)146-82(32-20-50-136-124(133)134)122(189)158-53-23-35-93(158)116(183)145-79(31-19-49-135-123(131)132)107(174)147-83(103(130)170)60-95(128)163)148-114(181)87(58-72-28-15-10-16-29-72)151-111(178)85(56-70-24-11-8-12-25-70)141-98(166)63-137-97(165)62-138-106(173)90(65-159)153-117(184)92-34-22-52-157(92)121(188)69(7)140-119(186)101(67(3)4)155-118(185)91-33-21-51-156(91)99(167)64-139-105(172)80(44-46-94(127)162)143-112(179)88(59-74-38-42-76(161)43-39-74)149-108(175)81(45-47-100(168)169)144-115(182)89(61-96(129)164)152-120(187)102(68(5)6)154-109(176)78(30-17-18-48-125)142-104(171)77(126)55-73-36-40-75(160)41-37-73/h8-16,24-29,36-43,66-69,77-93,101-102,159-161H,17-23,30-35,44-65,125-126H2,1-7H3,(H2,127,162)(H2,128,163)(H2,129,164)(H2,130,170)(H,137,165)(H,138,173)(H,139,172)(H,140,186)(H,141,166)(H,142,171)(H,143,179)(H,144,182)(H,145,183)(H,146,180)(H,147,174)(H,148,181)(H,149,175)(H,150,177)(H,151,178)(H,152,187)(H,153,184)(H,154,176)(H,155,185)(H,168,169)(H4,131,132,135)(H4,133,134,136)/t69-,77-,78-,79-,80-,81-,82-,83-,84-,85-,86-,87-,88-,89-,90-,91-,92-,93-,101-,102-/m0/s1 |
| Chemical Name | (4S)-4-[[(2S)-4-amino-2-[[(2S)-2-[[(2S)-6-amino-2-[[(2S)-2-amino-3-(4-hydroxyphenyl)propanoyl]amino]hexanoyl]amino]-3-methylbutanoyl]amino]-4-oxobutanoyl]amino]-5-[[(2S)-1-[[(2S)-5-amino-1-[[2-[(2S)-2-[[(2S)-1-[[(2S)-1-[(2S)-2-[[(2S)-1-[[2-[[2-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-1-[[(2S)-5-carbamimidamido-1-[(2S)-2-[[(2S)-5-carbamimidamido-1-[[(2S)-1,4-diamino-1,4-dioxobutan-2-yl]amino]-1-oxopentan-2-yl]carbamoyl]pyrrolidin-1-yl]-1-oxopentan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-4-methyl-1-oxopentan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-1-oxo-3-phenylpropan-2-yl]amino]-2-oxoethyl]amino]-2-oxoethyl]amino]-3-hydroxy-1-oxopropan-2-yl]carbamoyl]pyrrolidin-1-yl]-1-oxopropan-2-yl]amino]-3-methyl-1-oxobutan-2-yl]carbamoyl]pyrrolidin-1-yl]-2-oxoethyl]amino]-1,5-dioxopentan-2-yl]amino]-3-(4-hydroxyphenyl)-1-oxopropan-2-yl]amino]-5-oxopentanoic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Neuromedin U (rat) is an endogenous agonist for the Neuromedin U receptors NMUR1 and NMUR2. In competition binding assays using human NMUR2, the Ki value for unlabeled NMU-23 was determined to be 4.7 nM.[1] |
| ln Vitro |
The peptide Neuromedin U (NMU-23) was N-terminally tagged with Ru(bpy)2+3 and allowed to attach to human NMUR2 receptors in fixed cell membranes. attach to the electrodes on the bottom of each test plate in order to create an electrochemiluminescence (ECL) binding assay for NMUR2 receptors. The Ru(bpy)2+3-ligand attached to the receptor experiences a redox cycle with the application of an electric current, and in the presence of the co-reactant tripropylamine, it emits light. Ru(bpy)2+3 label only generates a signal when it is close to the electrode, allowing for the separation of bound from unbound label and a uniform assay format that doesn't need to be washed. We employed the ECL-based NMUR2 binding assay to screen the compound collection of our organization. The ability of over 670,000 different-structured compounds to prevent 0.5 nM Ru(bpy)2+3-NMU-23 from binding to the human NMUR2 receptor at 10 μM was investigated. According to competitive binding studies, NMU-23's Ki value was found to be 4.7 nM[1]. In HEK293S cells stably expressing NMUR2, Neuromedin U (rat, NMU-23) stimulates phosphoinositide (PI) turnover and intracellular calcium mobilization through the Gαq/11 signaling pathway.[1] In a radioactive competition binding assay using 125I-NMU-25 as the labeled ligand, unlabeled NMU-23 competed for binding to human NMUR2 membranes.[1] |
| ln Vivo |
Intrathecal administration of Neuromedin U (rat, NMU-23; 5 nmol) in decerebrate-spinalized rats increased nociceptive spinal reflex responses to pinch and touch stimuli applied to the hind paw.[1] |
| Enzyme Assay |
An electrochemiluminescent (ECL) ligand binding assay was developed. In this assay, unlabeled NMU-23 competed with Ru(bpy)2(5+)-NMU-23 for binding to human NMUR2 receptors immobilized on plates. The Ki value for NMU-23 was determined from the competition curve.[1] A radioactive ligand competition binding assay was also performed. Cell membranes containing NMU receptors were incubated with wheat germ agglutinin-coated SPA beads and 125I-NMU-25 in the presence of increasing concentrations of unlabeled NMU-23. Bound radioactivity was measured to determine binding affinity.[1] |
| Cell Assay |
For intracellular calcium mobilization assay, HEK293S cells stably expressing NMU receptors were plated in 384-well plates and loaded with a calcium-sensitive fluorescent dye. After incubation, cells were stimulated with Neuromedin U (rat, NMU-23), and the fluorescence response (indicative of calcium flux) was recorded using a fluorometric imaging plate reader (FLIPR).[1] For phosphoinositide (PI) turnover assay, HEK293S cells expressing NMUR2 were plated in 24-well plates and grown in medium containing myo-[3H]inositol for 2 days. After preincubation with LiCl-containing buffer, cells were stimulated with Neuromedin U (rat, NMU-23). The accumulated [3H]inositol phosphate was measured by ion-exchange column chromatography.[1] |
| Animal Protocol |
In vivo studies on nociceptive responses were conducted on adult male Sprague-Dawley rats. For testing the effects of exogenous Neuromedin U (rat, NMU-23), the peptide was delivered via an intrathecal cannula in a volume of 10 µL to decerebrate-spinalized rats. Nociceptive reflex responses to mechanical (pinch, touch) and thermal (52°C water) stimuli applied to the hind paw were recorded electrophysiologically.[1] |
| References | [1]. Liu JJ, et al. Discovery and pharmacological characterization of a small-molecule antagonist at neuromedin U receptor NMUR2. J Pharmacol Exp Ther. 2009 Jul;330(1):268-75. |
| Additional Infomation |
Neuromedin U (rat), specifically the 23-amino acid form (NMU-23), is a brain-gut peptide. It regulates several physiological functions including energy balance, stress response, and nociception. Its biological activity is dependent on a conserved C-terminal seven-amino acid segment.[1] In the central nervous system, NMU and its receptor NMUR2 are abundantly expressed in nociceptive sensory pathways, including the spinal dorsal horn, dorsal root ganglia, thalamus, and brain stem.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 0.3784 mL | 1.8918 mL | 3.7836 mL | |
| 5 mM | 0.0757 mL | 0.3784 mL | 0.7567 mL | |
| 10 mM | 0.0378 mL | 0.1892 mL | 0.3784 mL |