DEA Controlled Substances. Nemorexant (Daridorexant; ACT-541468; Quviviq; ACT541468) is a novel and potent orexin receptors antagonist that has been approved in January 2022 for the treatment of Insomnia Disorder in Adult Patients. It inhibits Ox1 receptor and Ox2 receptors with IC50s of 2 nM and 3 nM, respectively. By specific binding to both orexin receptors, daridorexant inhibits the actions of the wake-promoting orexin (also called hypocretin) neuropeptides. This mechanism avoids a more widespread inhibition of neuronal pathways and associated side effects that are intrinsic to positive allosteric GABA-A receptor modulators.
Physicochemical Properties
| Molecular Formula | C23H23CLN6O2 |
| Molecular Weight | 450.92 |
| Exact Mass | 450.157 |
| Elemental Analysis | C, 61.26; H, 5.14; Cl, 7.86; N, 18.64; O, 7.10 |
| CAS # | 1505484-82-1 |
| Related CAS # | 1792993-84-0 (HCl);1505484-82-1;Daridorexant mesylate; |
| PubChem CID | 91801202 |
| Appearance | Solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 747.6±70.0 °C at 760 mmHg |
| Flash Point | 405.9±35.7 °C |
| Vapour Pressure | 0.0±2.5 mmHg at 25°C |
| Index of Refraction | 1.704 |
| LogP | 3.38 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 32 |
| Complexity | 693 |
| Defined Atom Stereocenter Count | 1 |
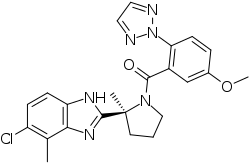
| SMILES | ClC1C=CC2=C(C=1C)N=C([C@]1(C)CCCN1C(C1=CC(=CC=C1N1N=CC=N1)OC)=O)N2 |
| InChi Key | NBGABHGMJVIVBW-QHCPKHFHSA-N |
| InChi Code | InChI=1S/C23H23ClN6O2/c1-14-17(24)6-7-18-20(14)28-22(27-18)23(2)9-4-12-29(23)21(31)16-13-15(32-3)5-8-19(16)30-25-10-11-26-30/h5-8,10-11,13H,4,9,12H2,1-3H3,(H,27,28)/t23-/m0/s1 |
| Chemical Name | [(2S)-2-(5-chloro-4-methyl-1H-benzimidazol-2-yl)-2-methylpyrrolidin-1-yl]-[5-methoxy-2-(triazol-2-yl)phenyl]methanone |
| Synonyms | Nemorexant; Daridorexant; ACT-541468; Quviviq; ACT54146 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Orexin receptors |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Daridorexant reaches peak plasma concentrations within one to two hours. Daridorexant has an absolute bioavailability of 62%. While a high-fat and high-calorie meal delayed the Tmax by 1.3 hours and decreased the Cmax by 16% in healthy subjects, the total exposure (AUC) was not affected. The primary route of excretion is via feces, accounting for approximately 57% of drug excretion. About 28% of the drug is excreted via urine primarily in the form of metabolites. Trace amounts of the parent drug were found in feces and urine. Daridorexant has a volume of distribution of 31 L. The blood to plasma ratio is 0.64. It effectively passes the blood-brain barrier. There is limited information on clearance. Metabolism / Metabolites Daridorexant undergoes extensive metabolism primarily mediated by CYP3A4 (89%), mostly via oxidative transformations. Other CYP enzymes individually contribute to less than 3% of metabolic clearance of daridorexant. Biological Half-Life The terminal half-life is approximately 8 hours. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Amounts of daridorexant in milk are very low. If daridorexant is required by the mother, it is not a reason to discontinue breastfeeding. Until more data in breastfed infants are available, monitor the infant for sedation, poor feeding and poor weight gain. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding Daridorexant is 99.7% bound to plasma proteins. |
| References | USE OF BENZIMIDAZOLE-PROLINE DERIVATIVES. WO2015083094A1. |
| Additional Infomation |
Pharmacodynamics Daridorexant binds to and antagonizes the orexin receptors OX1R and OX2R (Ki = 0.47 and 0.93 nM, respectively) equipotentally. In clinical trials, daridorexant improved sleep onset and sleep maintenance, and patient-reported total sleep time. Patient-reported daytime sleepiness was also reportedly reduced. At a dose four times the maximum recommended dose, daridorexant does not prolong the QTc interval to any clinically relevant extent. Daridorexant is currently being assessed for a controlled substance schedule in the US. In a human abuse potential study, daridorexant showed some abuse potential at doses higher than the recommended dose (100-150 mg), indicated by similar “drug liking” ratings to zolpidem (30 mg) and suvorexant in recreational sedative drug users. However, at clinically relevant concentrations, daridorexant does not bind to abuse-associated CNS targets. In animal studies and clinical trials evaluating physical dependence, chronic administration of daridorexant did not produce withdrawal signs or symptoms upon drug discontinuation, indicating that the drug does not produce physical dependence. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2177 mL | 11.0884 mL | 22.1769 mL | |
| 5 mM | 0.4435 mL | 2.2177 mL | 4.4354 mL | |
| 10 mM | 0.2218 mL | 1.1088 mL | 2.2177 mL |