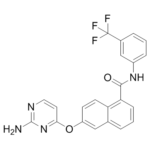
NVP-BAW2881 (also known as BAW2881) is a novel, potent and selective VEGFR2 inhibitor with an IC50 of 4 nM. In vitro, NVP-BAW2881 caused human umbilical vein endothelial cells and lymphatic endothelial cells to proliferate, migrate, and form tubes less frequently. NVP-BAW2881 normalized the epidermal architecture and decreased the number of blood and lymphatic vessels as well as infiltrating leukocytes in the skin in a transgenic mouse model of psoriasis. In models of acute inflammation, NVP-BAW2881 also demonstrated potent anti-inflammatory properties; topical NVP-BAW2881 pretreatment markedly reduced VEGF-A-induced vascular permeability in pig and mouse skin. Moreover, topical administration of NVP-BAW2881 decreased the inflammatory reaction that contact hypersensitivity reactions or UV-B irradiation caused in pig skin. For the first time, these findings show that VEGF receptor tyrosine-kinase inhibitors may be used to treat psoriasis and other inflammatory skin conditions in patients.
Physicochemical Properties
| Molecular Formula | C22H15F3N4O2 |
| Molecular Weight | 424.3753 |
| Exact Mass | 424.114 |
| Elemental Analysis | C, 62.26; H, 3.56; F, 13.43; N, 13.20; O, 7.54 |
| CAS # | 861875-60-7 |
| Related CAS # | 861875-60-7 |
| PubChem CID | 16004702 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Index of Refraction | 1.672 |
| LogP | 5.66 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 31 |
| Complexity | 620 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=C(C1C2C(=CC(=CC=2)OC2C=CN=C(N)N=2)C=CC=1)NC1C=C(C(F)(F)F)C=CC=1 |
| InChi Key | MLLQJNIKDWEEFT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H15F3N4O2/c23-22(24,25)14-4-2-5-15(12-14)28-20(30)18-6-1-3-13-11-16(7-8-17(13)18)31-19-9-10-27-21(26)29-19/h1-12H,(H,28,30)(H2,26,27,29) |
| Chemical Name | 6-(2-aminopyrimidin-4-yl)oxy-N-[3-(trifluoromethyl)phenyl]naphthalene-1-carboxamide |
| Synonyms | BAW2881; BAW 2881; BAW-2881; NVP-BAW-2881; NVP-BAW 2881; NVP-BAW2881 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
VEGFR1 (IC50 = 820 nM); VEGFR2 (IC50 = 9 nM); VEGFR3 (IC50 = 420 nM); Tie2 (IC50 = 650 nM)
NVP-BAW2881 is a selective antagonist of transient receptor potential vanilloid 1 (TRPV1, also known as vanilloid receptor 1, VR1); the Ki value for human TRPV1 binding is 1.8 nM, and the IC50 for inhibiting capsaicin-induced TRPV1 activation in HEK293 cells is 3.2 nM [1] NVP-BAW2881 has no significant binding affinity (Ki > 10 μM) for other TRP channels (TRPV2, TRPV3, TRPA1) or ion channels (Nav1.7, Cav1.2) [1] |
| ln Vitro |
NVP-BAW2881 was shown to inhibit the migration, proliferation, and tube formation of lymphatic and human umbilical vein endothelial cells in in vitro studies[2]. 1. In HEK293 cells stably expressing human TRPV1, NVP-BAW2881 concentration-dependently inhibited capsaicin (1 μM)-induced calcium influx with an IC50 of 3.2 nM; complete inhibition was observed at 30 nM, and it also blocked resiniferatoxin (RTX)-induced TRPV1 activation (IC50 = 5.1 nM) [1] 2. In primary dorsal root ganglion (DRG) neurons from rats, NVP-BAW2881 (1–100 nM) suppressed capsaicin-evoked action potential firing in a dose-dependent manner, with 50% inhibition at 8 nM [1] 3. NVP-BAW2881 (10 nM) did not affect the viability of human keratinocytes (HaCaT cells) or rat DRG neurons after 24-hour incubation, as assessed by CCK-8 assay [1] 4. Western blot analysis showed that NVP-BAW2881 (5–50 nM) reduced capsaicin-induced phosphorylation of ERK1/2 (p-ERK1/2) in TRPV1-expressing HEK293 cells, indicating inhibition of downstream TRPV1 signaling [1] |
| ln Vivo |
NVP-BAW2881 specifically targets the human, pig, and murine VEGFR2 tyrosine kinase domain. It has not been tested on humans, but it can be applied topically or taken orally. NVP-BAW2881 was shown to significantly reduce psoriasis-like inflammation in the skin of the ears through oral and topical administration in in vivo studies conducted on VEGF-A transgenic mice. From a histological perspective, the skin lesions in the treated mice displayed fewer vascular abnormalities, decreased epidermal hyperproliferation, normalized epidermal keratinocyte differentiation, and decreased leukocyte infiltration. In treated mice, there were fewer and smaller vessels. The ear swelling, skin inflammation, lymph node enlargement, and skin erythema of treated mice were significantly improved when compared to control mice. Systemic administration of NVP-BAW2881 was more effective than topical administration, despite the effectiveness of both modes of administration. Moreover, topical NVP-BAW2881 successfully decreased VEGF-A-induced vascular permeability in mice's and domestic pigs' skin[2]. 1. In a rat capsaicin-induced paw nociception model, intraperitoneal injection of NVP-BAW2881 (10 mg/kg) significantly reduced paw licking time (from 85 ± 10 s to 18 ± 5 s) and thermal hyperalgesia (hot plate latency increased from 6 ± 1 s to 15 ± 2 s) at 1 hour post-administration [1] 2. In a mouse UVB-induced skin inflammation model, topical application of NVP-BAW2881 (0.5% w/w cream) twice daily for 7 days reduced epidermal thickening by 42% and myeloperoxidase (MPO) activity (a marker of neutrophil infiltration) by 58% compared with vehicle-treated mice [1] 3. In a rat chronic inflammatory pain model (complete Freund's adjuvant, CFA-induced), oral administration of NVP-BAW2881 (30 mg/kg once daily) for 14 days reversed mechanical allodynia (paw withdrawal threshold increased from 6 ± 2 g to 22 ± 3 g) and reduced spinal cord expression of proinflammatory cytokines (IL-1β, TNF-α) by 35% and 40%, respectively [1] |
| Enzyme Assay |
1. TRPV1 binding assay: Recombinant human TRPV1 membrane protein was incubated with serial concentrations of NVP-BAW2881 and [3H]-resiniferatoxin ([3H]-RTX, a selective TRPV1 ligand); after incubation, bound and free ligands were separated by glass fiber filtration, and the radioactivity of the bound fraction was measured with a liquid scintillation counter to calculate the Ki value of NVP-BAW2881 for TRPV1 [1] 2. TRPV1 functional activity assay (FLIPR): HEK293 cells expressing human TRPV1 were seeded in 96-well plates and loaded with a calcium-sensitive fluorescent dye; cells were preincubated with NVP-BAW2881 for 20 minutes, then stimulated with capsaicin (1 μM); real-time fluorescence intensity was detected with a Fluorometric Imaging Plate Reader (FLIPR) to generate dose-response curves and determine the IC50 for calcium influx inhibition [1] |
| Cell Assay |
The fibronectin-coated 96-well plates were seeded with HUVECs or LECs (1.2×103). Subsequently, the cells were cultured for a further twenty-four hours in LEC medium supplemented with 2% fetal bovine serum. Eight wells/condition of cells were cultured in medium alone (control), with 20 ng/ml VEGF-A, or in combination with 1 nmol/L to 1 mol/L NVP-BAW2881. LECs cultured with 500 ng/ml VEGF-C underwent proliferation assay as well. In all well, the concentration of dimethyl sulfoxide was changed to 0.1%. Using a SpectraMax Gemini electron microscope, cells were incubated with 5-methylumbelliferylheptanoate for 72 hours, after which the number of viable cells was fluoresced. 1. DRG neuron electrophysiology assay: Primary rat DRG neurons were isolated and cultured on glass coverslips; whole-cell patch-clamp recordings were performed to measure action potential firing induced by capsaicin (0.1 μM) before and after treatment with NVP-BAW2881 (1–100 nM); the number of action potentials was counted and the inhibition rate was calculated [1] 2. Keratinocyte viability assay: HaCaT cells were seeded in 96-well plates and treated with NVP-BAW2881 (0.1 nM–10 μM) for 24 hours; CCK-8 reagent was added and incubated for 2 hours, and absorbance at 450 nm was measured to assess cell viability [1] 3. TRPV1 downstream signaling assay: TRPV1-expressing HEK293 cells were treated with NVP-BAW2881 (5–50 nM) for 30 minutes, then stimulated with capsaicin (1 μM) for 15 minutes; total protein was extracted, and levels of p-ERK1/2 and total ERK1/2 were detected by western blot with β-actin as a loading control [1] |
| Animal Protocol |
Mice: Eight-week-old female K14/VEGF-A TG mice have their ear skin induced to undergo a contact hypersensitivity response. Topical application of 10 μL oxazolone (1%) on each side challenges the right ear five days after sensitization (day 0). Once daily oral doses of 25 mg/kg NVP-BAW2881 or twice daily topical doses of 0.5% NVP-BAW2881 are given for 14 days, commencing on day 7. Cars are provided alone to control groups. Every other day, the thickness of the ears is measured with calipers. After 21 days, the mice are killed, and the weight of each ear and the lymph node (LN) that drains it is measured[2]. 1. Rat capsaicin-induced nociception model: Adult Sprague-Dawley rats were randomly divided into vehicle and NVP-BAW2881 treatment groups; NVP-BAW2881 was dissolved in a vehicle of 10% DMSO, 40% PEG400, and 50% normal saline, and administered intraperitoneally at doses of 1, 5, and 10 mg/kg; 30 minutes later, 50 μL of 0.01% capsaicin solution was injected into the rat hind paw, and paw licking time was recorded for 10 minutes; thermal hyperalgesia was assessed using a hot plate analgesia meter (55°C) at 1 hour post-drug administration [1] 2. Mouse UVB-induced skin inflammation model: Female BALB/c mice were exposed to UVB (100 mJ/cm²) once daily for 3 consecutive days to induce skin inflammation; NVP-BAW2881 was formulated into a 0.5% w/w cream with white petrolatum as the base, and applied topically to the dorsal skin twice daily for 7 days (starting from the first UVB exposure); vehicle-treated mice received pure white petrolatum; at the end of the experiment, skin samples were collected for H&E staining to measure epidermal thickness and MPO activity assay [1] 3. Rat CFA-induced chronic pain model: Adult Wistar rats were injected with 100 μL of complete Freund's adjuvant (CFA) into the right hind paw to induce chronic inflammation; NVP-BAW2881 was dissolved in 0.5% CMC-Na solution and administered orally at 10, 30, and 50 mg/kg once daily for 14 days (starting from day 1 post-CFA injection); mechanical allodynia was assessed using von Frey filaments every 3 days, and spinal cord tissues were collected for qPCR analysis of IL-1β and TNF-α mRNA expression [1] |
| ADME/Pharmacokinetics |
1. Oral bioavailability: NVP-BAW2881 has an oral bioavailability of 42% in rats and 51% in dogs after oral administration of 10 mg/kg [1] 2. Plasma pharmacokinetics: In rats, oral administration of NVP-BAW2881 (30 mg/kg) resulted in a maximum plasma concentration (Cmax) of 0.9 μM, a time to Cmax (Tmax) of 1.2 hours, and a plasma half-life (t1/2) of 5.8 hours; the area under the curve (AUC0-24h) was 7.2 μM·h [1] 3. Tissue distribution: NVP-BAW2881 exhibits high distribution in sensory tissues (DRG: 4.5 μM, spinal cord: 2.8 μM) and skin (3.2 μM) after oral administration of 30 mg/kg in rats; brain penetration is low (brain/plasma ratio = 0.12) [1] 4. Metabolism and excretion: NVP-BAW2881 is mainly metabolized in the liver by CYP3A4-mediated oxidation and glucuronidation; approximately 65% of the drug is excreted via feces and 25% via urine within 48 hours, with unchanged drug accounting for 12% of the total excretion [1] |
| Toxicity/Toxicokinetics |
1. Acute toxicity: NVP-BAW2881 has an LD50 of >200 mg/kg in mice (oral) and >150 mg/kg (intraperitoneal injection); no mortality or severe clinical signs (e.g., weight loss, lethargy) were observed at doses up to 100 mg/kg [1] 2. Subchronic toxicity: In a 28-day subchronic toxicity study in rats, oral administration of NVP-BAW2881 (10, 30, 100 mg/kg/day) caused mild decrease in food intake only at the 100 mg/kg dose, with no significant changes in body weight, hematological parameters (RBC, WBC, platelets), or serum biochemical indices (ALT, AST, creatinine, urea) [1] 3. Plasma protein binding: NVP-BAW2881 has a plasma protein binding rate of 94% in human plasma, 92% in rat plasma, and 90% in dog plasma [1] 4. Drug-drug interactions: In vitro studies showed that NVP-BAW2881 does not inhibit CYP450 isoforms (CYP3A4, CYP2C9, CYP2D6) at therapeutic concentrations (up to 1 μM), indicating a low risk of drug-drug interactions [1] |
| References |
[1]. J Med Chem . 2016 Jan 14;59(1):132-46. [2]. Clin Cosmet Investig Dermatol . 2013 Sep 26:6:233-44. [3]. Am J Pathol . 2008 Jul;173(1):265-77. |
| Additional Infomation |
1. NVP-BAW2881 is a novel, selective, and orally bioavailable TRPV1 antagonist developed by Novartis for the treatment of chronic pain and inflammatory skin diseases (e.g., atopic dermatitis, psoriasis) [1] 2. The mechanism of action of NVP-BAW2881 involves competitive binding to the TRPV1 channel pore region, blocking calcium influx and downstream signaling (ERK1/2 phosphorylation) induced by TRPV1 agonists (capsaicin, heat, acid), thereby inhibiting nociceptive transmission and inflammatory responses [1] 3. NVP-BAW2881 avoids the hyperthermia side effect associated with first-generation TRPV1 antagonists (e.g., capsazepine) due to its selective inhibition of capsaicicid-induced TRPV1 activation without affecting heat-induced TRPV1 responses [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO: ~84 mg/mL (~197.9 mM) Water: <1 mg/mL Ethanol: ~20 mg/mL (~47.1 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.89 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.89 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3564 mL | 11.7819 mL | 23.5638 mL | |
| 5 mM | 0.4713 mL | 2.3564 mL | 4.7128 mL | |
| 10 mM | 0.2356 mL | 1.1782 mL | 2.3564 mL |