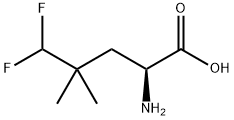
NV-5138 (NV 5138; NV5138) is a novel, selective, orally bioactive and brain/CNS-active mTORC1 (mammalian target of rapamycin complex 1) activator. Leucine analog used in research on antidepressants.Upregulation of brain-derived neurotrophic factor (BDNF) signaling is the mechanism through which NV-5138 exerts its antidepressant effects.
Physicochemical Properties
| Molecular Formula | C7H13F2NO2 |
| Molecular Weight | 181.1804292202 |
| Exact Mass | 181.09 |
| Elemental Analysis | C, 46.40; H, 7.23; F, 20.97; N, 7.73; O, 17.66 |
| CAS # | 2095886-80-7 |
| Related CAS # | NV-5138 hydrochloride;2639392-70-2 |
| PubChem CID | 129050791 |
| Appearance | White to off-white solid powder |
| LogP | -1 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 12 |
| Complexity | 171 |
| Defined Atom Stereocenter Count | 1 |
| SMILES | CC(C)(C[C@@H](C(=O)O)N)C(F)F |
| InChi Key | HRFIMCJTDKEPPV-BYPYZUCNSA-N |
| InChi Code | InChI=1S/C7H13F2NO2/c1-7(2,6(8)9)3-4(10)5(11)12/h4,6H,3,10H2,1-2H3,(H,11,12)/t4-/m0/s1 |
| Chemical Name | Pentanoic acid, 2-amino-5,5-difluoro-4,4-dimethyl-, (2S)- |
| Synonyms | NV 5138; NV-5138; 2095886-80-7; 4-(difluoromethyl)-L-leucine; (2S)-2-amino-5,5-difluoro-4,4-dimethylpentanoic acid; 06CA9QMG6Z; Pentanoic acid, 2-amino-5,5-difluoro-4,4-dimethyl-, (2S)-; ((S)-2-Amino-5,5-difluoro-4,4-dimethylpentanoic acid; NV5138 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
mTORC1 Target: NV-5138 selectively targets brain mammalian target of rapamycin complex 1 (mTORC1) and exerts effects by directly activating mTORC1; the EC50 for activating phosphorylation of mTORC1 downstream substrates in vitro is approximately 0.3 μM [1] - Target: NV-5138 acts as a Sestrin modulator, specifically activating brain mTORC1 by relieving Sestrin-mediated inhibition of mTORC1 [2] |
| ln Vitro |
NV-5138 modulates the Sestrin2-Gator2 interaction and activates mTORC1 in cells. [1] NV-5138 lacks proteinogenic capacity.[1] NV-5138 is not metabolized by Branched-Chain Amino Transaminase (BCAT).[1] NV-5138 is selective for Sestrin1/2.[1] NV-5138 produces ketamine-like antidepressant behavioral responses. [1] NV-5138 produces long-lasting antidepressant actions, similar to those of ketamine.[1] NV-5138–induced antidepressant actions require mTORC1 signaling.[1] NV-5138 increases synaptic number and function in mPFC pyramidal neurons. mTORC1 Activation Activity: NV-5138 activates mTORC1 in a dose-dependent manner. In primary cortical neurons, 1 μM concentration increases the phosphorylation level of mTORC1 downstream substrate S6 ribosomal protein (Ser235/236) by 2.8-fold and 4E-binding protein 1 (Thr37/46) by 2.1-fold [1] - Regulation of Sestrin-mTORC1 Interaction: In HEK293T cells overexpressing Sestrin2, NV-5138 (1 μM) disrupts the binding between Sestrin2 and GATOR2, relieves Sestrin2-mediated inhibition of mTORC1, and increases p-S6 levels by 3.2-fold compared with the control group [2] - Regulation of Synaptic Plasticity-Related Molecules: After primary hippocampal neurons are treated with NV-5138 (0.3-3 μM) for 24 hours, the protein expression levels of postsynaptic density protein 95 (PSD95) and Synapsin I are increased by 1.5-2.3-fold and 1.4-2.1-fold respectively, suggesting promotion of synaptogenesis [2] - Antidepressant-Related In Vitro Effects: In corticosterone-treated primary neurons, NV-5138 (1 μM) reverses corticosterone-induced reduction of p-S6 levels (recovered from 42% of the control group to 91%) and simultaneously upregulates brain-derived neurotrophic factor (BDNF) mRNA expression by 1.8-fold [2] |
| ln Vivo |
NV-5138 is found to be essentially 100% orally bioavailable with an elimination half-life in plasma of ~ 3 h determined following intravenous and oral dosing in rats[1].
In synaptosomal preparations of PFC, NV-5138 (160 mg/kg, po, single dose) significantly elevates levels of phospho-mTOR as well as its downstream targets, phospho-p70S6K1 and phosphor-4EB-P1[2].
Additionally, NV-5138 (80 mg/kg, po, daily for a total of 7 days) exhibits antidepressant properties[2]. Repeated low-dose NV-5138 (80 mg/kg) also produces antidepressant effects.[2] The antidepressant effects of lower doses of NV-5138 (40 or 80 mg/kg, p.o.) administered daily for a total of 7 days (starting with day 0) were also tested (Figure 2F). Because the antidepressant actions of ketamine begin to reverse after 7 days (24), ketamine (10 mg/kg, i.p.) was administered every other day for 6 days as a positive control. The results demonstrate that 80 mg/kg of NV-5138 showed antidepressant effects by significantly reducing the immobility time and latency to feed in both the FST and NSFT, respectively,without alteration of locomotor activity or HCF (Figure 2, G–J). This was in contrast with the lack of efficacy at this dose when evaluated 24 hours following a single administration (Figure 1, B and D). Ketamine administration also produced significant effects on immobility time and latency to feed in the FST and NSFT, respectively (Figure 2, G and J). NV-5138 rapidly reverses the behavioral and synaptic deficits caused by chronic stress.[2] The development of anhedonia, a core symptom of depression, with chronic unpredictable stress (CUS) exposure and the requirement for chronic treatment of a typical antidepressant to reverse this effect makes CUS one of the most valid models of depression (25, 26). The CUS model also provides a rigorous test for fast-acting antidepressants, and a single dose of ketamine rapidly reverses CUS-induced anhedonia, determined in a sucrose-preference test (SPT) (8). The results of the current study demonstrate that repeated CUS exposure (21 days) decreases sucrose preference and that a single dose of NV-5138 rapidly reverses this effect (Figure 3, A and B). NV-5138 administration did not influence sucrose preference in nonstressed control rats (Figure 3B). There was no significant difference in the total amount of water or total volume of liquid consumed (Supplemental Figure 1, A and B; supplemental material available online with this article; https://doi.org/10.1172/JCI126859DS1). CUS exposure was continued for subsequent behavioral testing and tissue sampling on day 26. CUS exposure increased latency to feed in the NSFT (day 22), and NV-5138 rapidly reversed this effect (Figure 3C). There were no effects of CUS or NV-5138 on HCF conducted immediately following the NSFT (Figure 3D), indicating that the effects of NV-5138 were not due to general increases in feeding. CUS exposure significantly decreased body weight, a predicted outcome of chronic stress exposure (Figure 3E). Antidepressant actions of NV-5138 require BDNF.[2] Previous studies demonstrate that the antidepressant actions of ketamine and other rapid acting antidepressant including scopolamine and rapastinel require brain-derived neurotrophic factor (BDNF), determined by infusion of a BDNF neutralizing Ab (nAb) or in BDNF mutant mice (33–38). Here, we evaluated the role of BDNF in the antidepressant actions of NV-5138 using both of these approaches. Rats received intra-mPFC infusion of a BDNF (nAb) (0.5 μg/side) 30 minutes before NV-5138 (160 mg/kg p.o.) and behavioral testing started 24 hours later (Figure 5A). In control IgG-infused rats, NV-5138 produced significant reduction in immobility time and latency to feed in the FST and NSFT, respectively, but BDNF nAb infusion completely blocked these effects (Figure 5, B and D); there were no effects of BDNF nAb alone in the FST or NSFT or on locomotor activity or HCF (Figure 5, B–E). Brain mTORC1 Activation and Tissue Distribution: One hour after oral administration of NV-5138 (30 mg/kg) to C57BL/6 mice, p-S6 (Ser235/236) levels in the prefrontal cortex, hippocampus, and striatum are increased by 2.5-fold, 2.3-fold, and 1.9-fold respectively, with no obvious activation effect in peripheral tissues (liver, kidney, muscle) [1] - Antidepressant-Like Behavioral Effects: In chronic unpredictable mild stress (CUMS)-induced depression model mice, oral administration of NV-5138 (10 mg/kg, once daily for 7 days) shortens the immobility time in the forced swim test from 218 seconds to 124 seconds, and the immobility time in the tail suspension test from 196 seconds to 112 seconds; a single oral dose of 30 mg/kg shows similar antidepressant effects 24 hours later, which lasts for 72 hours [2] - Improvement of Synaptic Plasticity: After CUMS model mice are treated with NV-5138 (30 mg/kg, oral administration for 14 consecutive days), the synaptic density in the hippocampal CA1 region is increased by 35% compared with the model group, and the protein expression levels of PSD95 and BDNF are increased by 42% and 38% respectively [2] - Blood-Brain Barrier Penetration: After oral administration of NV-5138 (30 mg/kg) to mice, the peak brain concentration (Cmax) is 1.2 μM at 1 hour post-dose, with a brain-plasma concentration ratio of 0.8 [1] |
| Enzyme Assay |
In vitro pharmacological screening[1] NV-5138 was tested in duplicate at a concentration of 300 µM for potential interaction with a panel of 107 adverse drug effect targets including 21 transporters, receptors, ion channels and enzymes expressed in the CNS. The threshold to exclude a significant interaction was set as a binding/response of >50%. BCAT enzymatic assay[1] The assay buffer conditions were as follows: alpha-ketoglutarate (5 mM), NADH (0.075 mM), pyridoxal 5′-phosphate (5 µM), leucine dehydrogenase (0.95 U), ammonium sulfate (50 mM), DTT (5 mM) and vehicle (water), L-leucine, arginine, and NV-5138 as indicated. The assay buffer was dispensed into a 96-well plate and brought up to 37 °C for 10 min before adding BCAT1 or BCAT2 (72 ng) in 0.1 M potassium phosphate buffer. Absorbance at 340 nm was then read every minute for 30 min at RT using an Envision plate reader. Source of each reagents used can be found in Supplementary Information. 14C-leucine protein incorporation assay[1] HeLa cells were washed once with PBS then incubated in starvation media (leucine and serum-free DMEM) containing CHX (10 ug/ml) or DMSO (1% final) for 2.5 h at 37 °C. After incubation, cells were washed once with PBS then incubated in labeling media containing 14C-leucine (2 µCi/ml) along with NV-5138 or non-radioactive leucine for 30 min at 37 °C. After incubation, cells were harvested and lysed in Tris/HCl 10 mM pH 7.4 plus 1% NP40. Lysate were mixed with water and 20% trichloroacetic acid (1:1:2) and incubated on ice for 2 h to precipitate the protein. Proteins were collected by centrifugation, solubilized in NaOH, placed in scintillation fluid and read on a Wallac Microbeta Trilux 2450 with a 2-min count. Scintillation values were normalized as the percentage of the counts from the DMSO-treated wells. Isothermal titration calorimetry (ITC)[1] Because NV-5138 is an analog of the natural ligand L-leucine, and because it was known that leucine appears to co-purify with Sestrin212, a procedure was developed to displace the endogenous leucine from the binding site and replace it with the weaker Sestrin2 ligand L-Methionine24. Methionine was later displaced by leucine and analogs for direct binding measurements. Details of NV-5138 synthesis and protein preparation for ITC can be found in Supplementary Information. mTORC1 Activity Detection Assay: Primary cortical neurons are seeded and cultured for 7 days, then treated with serial concentrations of NV-5138 (0.01-10 μM) for 24 hours. Total cellular protein is extracted, and Western blot is used to detect the expression of p-S6 (Ser235/236), p-4EBP1 (Thr37/46), total S6, and total 4EBP1. The p-S6/total S6 ratio is used as an indicator of mTORC1 activation, and EC50 is calculated by nonlinear regression analysis [1] - Sestrin-GATOR2 Binding Interference Assay: Expression vectors for Sestrin2 and GATOR2 subunit (WDR24) are constructed and co-transfected into HEK293T cells. After incubation with NV-5138 (0.1-3 μM) for 16 hours, co-immunoprecipitation assay is performed (precipitating complexes with Sestrin2 antibody), and Western blot is used to detect the content of WDR24 in the complexes to analyze the interference effect of NV-5138 on their binding [2] |
| Cell Assay |
Cell culture[1] All cell lines were cultured in DMEM supplemented with 10% FBS and maintained at 37 °C and 5% CO2. HEK-293T cells stably expressing Flag-WDR24 was generated as previously described24. For leucine starvation, cells were rinsed once with and incubated in leucine-free DMEM supplemented 10% dFBS for 50 min followed by treatment with NV-5138, leucine or vehicle for 10 min. Details of reagent used for cell culture can be found in Supplementary Information. Western blotting on immunoprecipitation (IP)[1] Cell lysates were prepared as previously described24. Lysates were cleared by centrifugation at 13,200 rpm at 4 °C for 8 min. For anti-FLAG IPs, the FLAG-M2 affinity gel was added to approximately 2 mg of lysates and incubated with rotation for 3 h at 4 °C. Following IP, the beads were washed one time with Triton wash buffer containing 500 mM NaCl. For lysate-based protein-protein interaction assay IPs were resuspended in cytosolic buffer containing vehicle, NV-5138 or leucine at the indicated doses for 10 min. After compound incubation, IPs were collected by centrifugation. Immunoprecipitated proteins were denatured and, resolved by SDS-PAGE. Membranes were imaged using the LI-COR imaging system. Reagents and antibodies used are listed in Supplementary Information. Cellular thermal shift assay (CETSA)[1] CETSA was performed as previously described23. Briefly, HEK293T cells were incubated for an hour in the leucine-free media, then NV-5138 (300 µM), leucine (300 µM) or vehicle (water) was added for an additional 30 min. Cells were then collected, centrifuged for 3 min at 300 g at room-temperature (RT), washed with PBS, centrifuged again, and finally resuspended in 1 ml of RT PBS containing EDTA-free protease-inhibitor tablets (Roche). 100 µl of each cell suspension was heated at the noted temperature for 3 min, samples were then placed on the bench at RT for 3 min before being flash-frozen in liquid nitrogen. To lyse the cells, samples were put through two cycles of freeze-thawing followed by centrifugation at 20,000 g for 20 min at 4 °C. 80 µl of the resulting supernatant was denatured and, resolved by SDS-PAGE. Membranes were imaged using the LI-COR imaging system. Antibodies used are listed in Supplementary Information. Primary Neuron Culture and mTORC1 Activation Detection: Cortical or hippocampal neurons are isolated from the brains of embryonic day 18 rats, seeded on coated culture plates, and cultured for 7-10 days. After treatment with NV-5138 for a specified time, protein is extracted for Western blot analysis, or cells are fixed for immunofluorescence staining (to detect the cellular localization of p-S6) [1] - Synaptic-Related Protein Expression Detection: Primary hippocampal neurons are treated with NV-5138 (0.3-3 μM) for 24 hours, total protein is extracted, and Western blot is used to detect the expression levels of PSD95 and Synapsin I, with β-actin as the internal reference. Band gray values are quantified using ImageJ software [2] - Corticosterone-Induced Neuronal Injury Model Experiment: Primary cortical neurons are cultured for 7 days, then treated with corticosterone (200 μM) to establish an injury model, and co-incubated with NV-5138 (0.1-3 μM) for 24 hours. RNA is extracted and reverse-transcribed into cDNA, qPCR is used to detect BDNF mRNA expression levels; protein is extracted to detect p-S6 and total S6 expression [2] |
| Animal Protocol |
Male Sprague-Dawley rats weighing 250-260 g[2]. 40, 80, 160 mg/kg. PO, single dose (160 mg/kg) or daily for a total of 7 days (40, 80 mg/kg). Animals and drug administration.[2] Male Sprague-Dawley rats weighing 250–260 g were used for most studies, with the exception of the CUS experiment, which used rats weighing 120–140 g because of the longer duration of the study. In addition, mutant BDNF Val66Met knockin mice (Val/Val WT, heterozygous Val/Met, and homozygous Met/Met mice at 8–12 weeks) generated as previously described were used to test the role of BDNF. Animals were singly housed and maintained in standard conditions with a 12-hour light/12-hour dark cycle and ad libitum access to food and water. Rats received a single oral administration of vehicle (0.5% methylcellulose and 0.1% Tween 80), NV-5138 (40, 80, or 160 mg/kg, p.o., Navitor Pharmaceuticals Inc.), ketamine (10 mg/kg, i.p.), DMSO (0.5%; 1 ml/kg, i.p.), or NBQX (10 mg/kg, i.p., Tocris Bioscience). For the repeated dose study, NV-5138 (40 or 80 mg/kg, p.o.) was administered daily for a total of 7 days and ketamine was injected (10 mg/kg, i.p.) every other day for 6 days. Pharmacokinetic analysis of NV-5138 in rats[1] To determine oral bioavailability, male SD rats were dosed I.V at 1 mg/kg and PO at 5 mg/kg with NV-5138 in 0.5% MC/0. 1% Tween80 (n = 3 per time point per group). Rats were fasted overnight for PO group and were given free access to food and water for IV group. After dosing, tail-vein blood was collected at the indicated time points into K2EDTA tubes and centrifuged at 2,000 g for 5 min to collect plasma. To measure levels of NV-5138 in the brain and plasma, rats were dosed PO at 160 mg/kg (n = 5), sacrificed 1 h after dosing via decapitation and trunk blood and one hemisphere of the brain was collected. Plasma was combined with acetonitrile containing pregabalin as an internal standard and centrifuged, brain and heart samples were homogenized directly into acetonitrile. Levels of NV-5138 were quantified via LC-MS/MS. WinNonlin V 6.2 statistics software was used to generate pharmacokinetics parameters using non-compartmental model. Western Blotting analysis of mTORC1 signaling and synaptic proteins[1] SD rats were dosed via oral gavage with NV-5138, leucine or vehicle (50 mg/ml in 0.5% MC and 0.1% Tween80 in water) at indicated doses and time points. After compound administration, animals were sacrificed by decapitation using a guillotine, immediately Specific regions of the brain were quickly microdissected, tissues collected, and flash frozen in liquid nitrogen. Peripheral tissues and all brain were homogenized using MP homogenizer in Lysis Buffer (Cell lysis buffer: 1% Triton X-100, 50 mM HEPES pH 7.4, 100 mM NaCl, 2 mM EDTA, 10 mM Beta-glycerophosphate, 10 mM Sodium pyrophosphate, and 1 protease inhibitor) at 4 °C. Synaptic proteins and mTORC1 analyses in each brain region were conducted on crude synaptoneurosome preparations as previously described16. Equal amounts of total protein from each sample were denatured and, resolved by SDS-PAGE. Membranes were imaged using the LI-COR imaging system. Proteins levels are normalized to tubulin or GAPDH levels and further normalized to vehicle treated rats for each tissue. Antibodies used are listed in Supplementary Information. Brain mTORC1 Activation and Pharmacokinetic Distribution Experiment: 8-week-old C57BL/6 mice are randomly grouped and orally administered NV-5138 (10, 30 mg/kg) or vehicle (0.5% methylcellulose + 0.1% Tween 80). Mice are sacrificed at 0.5, 1, 2, 4, and 8 hours post-dose, plasma and brain tissues (prefrontal cortex, hippocampus, striatum, liver, kidney) are collected. Brain tissues are homogenized, and drug concentrations are detected by LC-MS/MS; meanwhile, brain tissue protein is extracted, and p-S6 levels are detected by Western blot [1] - Chronic Depression Model (CUMS) Efficacy Experiment: 8-week-old C57BL/6 mice are subjected to CUMS treatment (including food deprivation, water deprivation, cage tilting, day-night reversal, etc., one stimulus per day for 4 consecutive weeks). After modeling, NV-5138 (10, 30 mg/kg) or positive drug is orally administered once daily for 7 days. Behavioral tests are performed during administration: forced swim test (recording immobility time within 6 minutes), tail suspension test (recording immobility time within 6 minutes), novelty-suppressed feeding test (recording first feeding latency) [2] - Single-Dose Antidepressant Effect Experiment: Normal C57BL/6 mice or CUMS model mice are given a single oral dose of NV-5138 (30 mg/kg) or vehicle. Forced swim and tail suspension tests are performed at 24, 48, and 72 hours post-dose to evaluate the duration of antidepressant effects [2] - Synaptic Plasticity Detection Animal Experiment: After CUMS model mice are treated with NV-5138 (30 mg/kg, oral administration for 14 consecutive days), mice are sacrificed to collect hippocampal tissues, frozen sections are prepared, and immunofluorescence staining is used to detect the fluorescence intensity of PSD95 (postsynaptic marker) and Synapsin I (presynaptic marker). Synaptic density is analyzed by ImageJ software [2] |
| ADME/Pharmacokinetics |
Pharmacokinetics (PK) and Pharmacodynamics (PD) Following Oral Administration of NV-5138[1] NV-5138 was found to be essentially 100% orally bioavailable with an elimination half-life in plasma of ~ 3 h determined following intravenous and oral dosing in rats (Supplementary Table 3 and Fig. 4a). Given the favorable PK properties of NV-5138, we wished to determine whether oral administration of NV-5138 could activate mTORC1 in the brain and other organs of ad libitum fed rats using the mTORC1 downstream substrate phosphorylated S6 (S240/244pS6) as a surrogate PD parameter for the activation status of the complex. As shown in Fig. 3a and Supplementary Fig. 5a, there was a dose-dependent increase in S240/244pS6 in synaptoneurosomes isolated from the brain pre-frontal cortex (PFC) 1 h following dosing with a significant increase of ~ 2-fold versus vehicle observed at 160 mg/kg and a trending increase at 80 mg/kg. Based on this result we decided to perform subsequent experiments using the 160 mg/kg oral dose which resulted in a similar PK profile in plasma and whole brain extracts suggesting a high level of brain exposure (Supplementary Fig. 4b). One hour following a single oral administration of NV-5138, significant activation of mTORC1 across multiple brain regions in addition to the pre-frontal cortex including the striatum, hippocampus, neocortex but not the cerebellum was observed (Fig. 3b and Supplementary Fig. 5b). Oral Bioavailability and Plasma Metabolism: The oral bioavailability of NV-5138 (30 mg/kg) in mice is 42%, with a plasma peak time (Tmax) of 1 hour, Cmax of 1.5 μM, terminal half-life (t1/2) of 3.8 hours, and AUC0-∞ of 8.7 μM·h [1] - Tissue Distribution and Blood-Brain Barrier Penetration: At 1 hour post-dose, the concentration of NV-5138 in various brain regions (prefrontal cortex, hippocampus, striatum) is 0.9-1.2 μM, with a brain-plasma concentration ratio of 0.8; concentrations in the liver and kidney are 2.3 μM and 1.8 μM respectively, and the muscle concentration is only 0.3 μM, showing brain targeting [1] - Metabolism and Excretion: Human liver microsome experiments show that NV-5138 is mainly metabolized by CYP3A4 to generate 2 major oxidative metabolites; within 48 hours after administration in mice, urinary excretion accounts for 32% of the administered dose, and fecal excretion accounts for 58%, of which the parent drug accounts for 12% and 25% of urinary and fecal excretion respectively [1] |
| Toxicity/Toxicokinetics |
Acute Toxicity: A single oral dose of NV-5138 up to 200 mg/kg in mice causes no death or obvious toxic symptoms (normal body weight, behavior, and food intake) within 14 days, with a median lethal dose (LD50) >200 mg/kg [1] - Repeat-Dose Toxicity: Rats are orally administered NV-5138 (30, 100 mg/kg) once daily for 28 consecutive days, showing normal weight gain, and blood routine, liver and kidney function (ALT, AST, creatinine, urea nitrogen) and electrolyte indicators are within the normal range; pathological sections show no obvious pathological damage to major organs such as heart, liver, spleen, lung, kidney, and brain [1] - Plasma Protein Binding Rate: In vitro experiments determine that the human plasma protein binding rate of NV-5138 is 82%-86%, and the mouse plasma protein binding rate is 78%-83% [1] - Safety Characteristics: At antidepressant effective doses (10-30 mg/kg), there is no obvious sedation, motor coordination disorder, or cognitive function impairment, and no abnormalities in blood glucose or blood lipids are observed [2] |
| References |
[1]. Discovery of NV-5138, the first selective Brain mTORC1 activator. Sci Rep. 2019 Mar 11;9(1):4107. [2]. Sestrin modulator NV-5138 produces rapid antidepressant effects via direct mTORC1 activation. J Clin Invest. 2019 Apr 16;129(6):2542-2554. |
| Additional Infomation |
The mechanistic target of rapamycin complex 1 (mTORC1) has been linked to several important chronic medical conditions many of which are associated with advancing age. A variety of inputs including the amino acid leucine are required for full mTORC1 activation. The cytoplasmic proteins Sestrin1 and Sestrin2 specifically bind to the multiprotein complex GATOR2 and communicate leucine sufficiency to the mTORC1 pathway activation complex. Herein, we report NV-5138, a novel orally bioavailable compound that binds to Sestrin2 and activates mTORC1 both in vitro and in vivo. NV-5138 like leucine transiently activates mTORC1 in several peripheral tissues, but in contrast to leucine uniquely activates this complex in the brain due lack of metabolism and utilization in protein synthesis. As such, NV-5138 will permit the exploration in areas of unmet medical need including neuropsychiatric conditions and cognition which have been linked to the activation status of mTORC1.[1] Preclinical studies demonstrate that rapid acting antidepressants, including ketamine require stimulation of mTORC1 signaling. This pathway is regulated by neuronal activity, endocrine and metabolic signals, notably the amino acid leucine, which activates mTORC1 signaling via binding to the upstream regulator sestrin. Here, we examined the antidepressant actions of NV-5138, a novel highly selective small molecule modulator of sestrin that penetrates the blood brain barrier. The results demonstrate that a single dose of NV-5138 produced rapid and long-lasting antidepressant effects, and rapidly reversed anhedonia caused by chronic stress exposure. The antidepressant actions of NV-5138 required BDNF release as the behavioral responses are blocked by infusion of a BDNF neutralizing antibody into the medial prefrontal cortex (mPFC) or in mice with a knock-in of a BDNF polymorphism that blocks activity dependent BDNF release. NV-5138 administration also rapidly increased synapse number and function in the mPFC, and reversed the synaptic deficits caused by chronic stress. Together, the results demonstrate that NV-5138 produced rapid synaptic and antidepressant behavioral responses via activation of the mTORC1 pathway and BDNF signaling, indicating that pharmacological modulation of sestrin is a novel approach for development of rapid acting antidepressants.[2] Drug Classification and R&D Background: NV-5138 is the first discovered selective brain mTORC1 activator, a small-molecule compound with a molecular weight of 412.5 Da. After structural optimization, it has good brain penetration and oral bioavailability [1] - Mechanism of Action: NV-5138 activates brain mTORC1 through two pathways: one is directly binding to the regulatory domain of mTORC1 to enhance its kinase activity; the other is acting as a Sestrin modulator to interfere with the interaction between Sestrin and GATOR2, relieving Sestrin-mediated inhibition of mTORC1, and ultimately promoting synaptic plasticity and neurotrophic factor expression to exert antidepressant effects [2] - Indication-Related Potential: Based on animal experimental results, NV-5138 has the potential for rapid-acting antidepressant effects. Its antidepressant-like effects are evident 24 hours after a single dose, without the delayed onset problem of traditional antidepressants, providing a new strategy for the treatment of depression [2] - Selectivity Characteristics: NV-5138 has no activating or inhibitory effect on mTORC2, no obvious impact on other kinases (such as PI3K, Akt, ERK), and weak mTORC1 activation effect in peripheral tissues, reducing the risk of systemic side effects [1] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~10 mg/mL (~55.2 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.5194 mL | 27.5969 mL | 55.1937 mL | |
| 5 mM | 1.1039 mL | 5.5194 mL | 11.0387 mL | |
| 10 mM | 0.5519 mL | 2.7597 mL | 5.5194 mL |