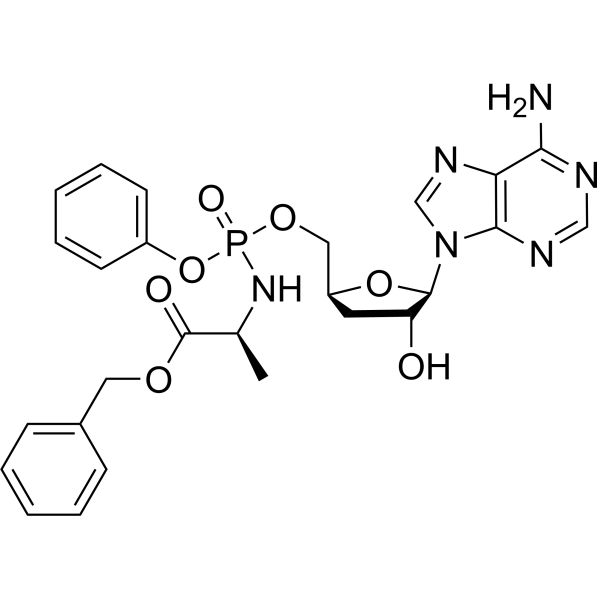
Physicochemical Properties
| Molecular Formula | C26H29N6O7P |
| Molecular Weight | 568.518226385117 |
| Exact Mass | 568.183 |
| CAS # | 2348493-39-8 |
| PubChem CID | 166177279 |
| Appearance | White to off-white solid powder |
| LogP | 1.8 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 12 |
| Rotatable Bond Count | 12 |
| Heavy Atom Count | 40 |
| Complexity | 877 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | C[C@@H](C(=O)OCC1=CC=CC=C1)NP(=O)(OC[C@@H]2C[C@H]([C@@H](O2)N3C=NC4=C(N=CN=C43)N)O)OC5=CC=CC=C5 |
| InChi Key | UDLWWGQHMQIYCV-LKKHFEEPSA-N |
| InChi Code | InChI=1S/C26H29N6O7P/c1-17(26(34)36-13-18-8-4-2-5-9-18)31-40(35,39-19-10-6-3-7-11-19)37-14-20-12-21(33)25(38-20)32-16-30-22-23(27)28-15-29-24(22)32/h2-11,15-17,20-21,25,33H,12-14H2,1H3,(H,31,35)(H2,27,28,29)/t17-,20-,21+,25+,40?/m0/s1 |
| Chemical Name | benzyl (2S)-2-[[[(2S,4R,5R)-5-(6-aminopurin-9-yl)-4-hydroxyoxolan-2-yl]methoxy-phenoxyphosphoryl]amino]propanoate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | NUC-7738 (compound 7a) exhibited cytotoxic and anticancer effects on leukocyte lineage cells with an LC50 value of less than 30 μM. It was tested against CCRF-CEM, HL-60, KG-1, MOLT-4, K562, MV4-11, THP-1, HEL92, NCI-H929, RPMI-8226, Jurkat, Z138, RL, HS445, HepG2, MCF-7, Bx-PC-3, HT29, MIA PaCa-2, and SW620 cells. NUC-7738 (1 μM) exhibits a t1/2 value of 48.1 min, indicating high stability in human hepatocytes[1]. With no change in plasma concentration for up to 4 hours, NUC-7738 is more stable in human plasma than the parent nucleoside [1]. |
| References | [1]. Serpi M, et, al. Synthesis and Characterization of NUC-7738, an Aryloxy Phosphoramidate of 3'-Deoxyadenosine, as a Potential Anticancer Agent. J Med Chem. 2022 Dec 8;65(23):15789-15804. |
| Additional Infomation | Fosdesdenosine Sipalabenamide is a phosphoramidate derivative of the monophosphate form of cordycepin (3'-deoxyadenosine; 3'-dA), an adenosine derivative first isolated from Cordyceps sinensis, with potential antineoplastic, antioxidant, and anti-inflammatory activities. Upon administration and cellular uptake of fosdesdenosine sipalabenamide by passive diffusion, cordycepin monophosphate (3'-dAMP) is converted into its active anti-cancer metabolite 3'-deoxyadenosine triphosphate (3'-dATP). 3'-dATP functions as a ribonucleoside analogue and competes with ATP during transcription. Therefore, this agent causes RNA synthesis inhibition, inhibits cellular proliferation, and induces apoptosis. Also, 3'-dAMP activates AMP-activated protein kinase (AMPK) and reduces mammalian target of rapamycin (mTOR) signaling. This prevents the hyperphosphorylation of the translation repressor protein 4E-BP1. This results in the induction of tumor cell apoptosis and a decrease in tumor cell proliferation. mTOR, a serine/threonine kinase belonging to the phosphatidylinositol 3-kinase (PI3K)-related kinase (PIKK) family, plays an important role in the PI3K/AKT/mTOR signaling pathway that regulates cell growth and proliferation, and its expression or activity is frequently dysregulated in human cancers. Compared to cordycepin alone, the addition of the phosphoramidate moiety may overcome cancer resistance and allow for greater cytotoxicity as fosdesdenosine sipalabenamide does not require a nucleoside transporter for cellular uptake, is independent of enzymatic activation by adenosine kinase (AK) and is not susceptible to enzymatic degradation by adenosine deaminase (ADA). Altogether, this may help overcome cancer resistance to cordycepin. |
Solubility Data
| Solubility (In Vitro) | DMSO: 100 mg/mL (175.90 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (4.40 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (4.40 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7590 mL | 8.7948 mL | 17.5895 mL | |
| 5 mM | 0.3518 mL | 1.7590 mL | 3.5179 mL | |
| 10 mM | 0.1759 mL | 0.8795 mL | 1.7590 mL |