Physicochemical Properties
| Molecular Formula | C₁₉H₃₃CL₂NS₂ |
| Molecular Weight | 410.51 |
| Exact Mass | 409.143 |
| Elemental Analysis | C, 55.59; H, 8.10; Cl, 17.27; N, 3.41; S, 15.62 |
| CAS # | 13116-77-3 |
| PubChem CID | 361263 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 15 |
| Heavy Atom Count | 24 |
| Complexity | 246 |
| Defined Atom Stereocenter Count | 0 |
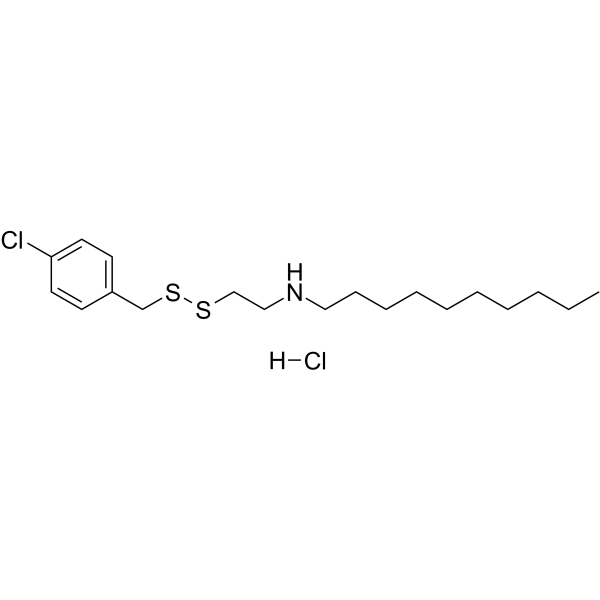
| SMILES | [Cl-].ClC1C=CC(C(S)(S)CC[NH2+]CCCCCCCCCC)=CC=1 |
| InChi Key | JQHUAYXPNPTQRO-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C19H32ClNS2.ClH/c1-2-3-4-5-6-7-8-9-14-21-15-16-22-23-17-18-10-12-19(20)13-11-18;/h10-13,21H,2-9,14-17H2,1H3;1H |
| Chemical Name | N-[2-[(4-chlorophenyl)methyldisulfanyl]ethyl]decan-1-amine;hydrochloride |
| Synonyms | NSC 624206; 13116-77-3; NSC624,206; NSC-624,206; NSC 624,206; N-(2-((p-Chlorobenzyl)dithio)ethyl)decylamine hydrochloride; 1-Decanamine, N-[2-[[(4-chlorophenyl)methyl]dithio]ethyl]-, hydrochloride (1:1); C19H33Cl2NS2; orb1709003; NSC-624206 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Ubiquitin-activating enzyme E1 (inhibits ubiquitin thioester formation without affecting ubiquitin adenylation; prevents p27 ubiquitination in vitro) [1] |
| ln Vitro |
- NSC624206 (1-10 μM) dose-dependently inhibited ubiquitin thioester formation in recombinant E1 enzyme assays, as measured by fluorescence resonance energy transfer (FRET)–based ubiquitin activation assays. It showed no significant effect on ubiquitin adenylation, indicating specificity for the thioesterification step [1] - In human cancer cell lines (e.g., HeLa, HCT116), NSC624206 (5-20 μM) increased levels of the tumor suppressor protein p27 (Western blot) by blocking its ubiquitination and proteasomal degradation. This led to G1 cell cycle arrest (flow cytometry) and induction of apoptosis (Annexin V/PI staining) [1] - The compound (10 μM) reduced cell proliferation (MTT assay) by 50-70% in p53-mutant colorectal cancer cells, with IC50 values ranging from 8-12 μM [1] In the HepG2 liver cancer cell line, NSC624206 promotes the accumulation of p27 and inhibits the in vitro ubiquitination of the cell cycle regulator p27 [1]. With an IC50 of 13 μM, NSC624206 considerably lowers ubiquitin-E1 thioester in a concentration-dependent manner [1]. In cells, NSC624206 (10 nM–10 μM; 24 hours) causes p27 to accumulate [1]. |
| ln Vivo |
- In nude mice bearing HCT116 xenografts, NSC624206 (20-50 mg/kg, intraperitoneal injection, daily for 14 days) significantly reduced tumor growth (tumor volume inhibition: 40-60%) compared to vehicle controls. Immunohistochemical analysis revealed increased p27 expression and decreased Ki-67 proliferation marker in tumor tissues [1] - Combination treatment with NSC624206 (30 mg/kg) and 5-fluorouracil (5-FU) in HCT116 xenografts showed synergistic antitumor effects, with tumor regression in 30% of mice and prolonged survival [1] |
| Enzyme Assay |
- Ubiquitin E1 activation assay: Recombinant human E1 enzyme (10 nM) was incubated with NSC624206 (0.1-10 μM) in reaction buffer containing ATP and ubiquitin. Thioester formation was detected using a FRET-based ubiquitin conjugate assay, where inhibition was calculated by comparing fluorescence intensity changes to vehicle controls [1] - Ubiquitin adenylation assay: E1 enzyme (10 nM) was incubated with NSC624206 (1-100 μM) and [α-³²P]ATP. Adenylated E1-ubiquitin complexes were resolved by SDS-PAGE and visualized by autoradiography. NSC624206 showed no significant inhibition of adenylation at concentrations up to 100 μM [1] |
| Cell Assay |
Western Blot Analysis[1] Cell Types: Kip16 Cell Tested Concentrations: 10 nM-10 μM Incubation Duration: 24 hrs (hours) Experimental Results: Induction of p27 accumulation. - p27 stabilization assay: HeLa cells were treated with NSC624206 (5-20 μM) for 24 hours, lysed, and subjected to Western blot analysis. Levels of p27 and β-actin (loading control) were quantified. NSC624206 increased p27 protein levels in a dose-dependent manner [1] - Cell cycle analysis: HCT116 cells treated with NSC624206 (10 μM) for 48 hours were fixed, stained with propidium iodide, and analyzed by flow cytometry. The percentage of cells in G1 phase increased from 40% (control) to 65-70%, with a corresponding decrease in S phase [1] |
| Animal Protocol |
- Xenograft model: Nude mice (6-8 weeks old) were subcutaneously implanted with HCT116 cells (5×10⁶). When tumors reached 100-200 mm³, NSC624206 (20-50 mg/kg) was administered via intraperitoneal injection daily for 14 days. Tumor volume was measured twice weekly using calipers, and tissues were harvested for immunohistochemistry [1] - Combination therapy: Mice with HCT116 xenografts received NSC624206 (30 mg/kg) daily and 5-FU (20 mg/kg, intraperitoneal) twice weekly for 14 days. Tumor growth and survival were monitored [1] |
| ADME/Pharmacokinetics |
- In mice, NSC624206 (50 mg/kg, intraperitoneal) showed rapid absorption, with peak plasma concentration (Cmax) of 2.8 μg/mL at 1 hour. Plasma half-life (t1/2) was 3.5 hours, and bioavailability after oral administration was approximately 25% [1] - The compound was primarily distributed to tumor tissues, with a tumor/plasma concentration ratio of 2.1 at 4 hours post-dose [1] |
| Toxicity/Toxicokinetics |
- In acute toxicity studies, NSC624206 had an LD50 > 200 mg/kg in mice. No significant changes in liver (ALT/AST) or kidney (creatinine) function markers were observed at therapeutic doses [1] - Common adverse effects in mice included transient diarrhea and weight loss at doses ≥50 mg/kg, which resolved within 24 hours [1] |
| References |
[1]. Identification and Mechanistic Studies of a Novel Ubiquitin E1 Inhibitor. J Biomol Screen . 2012 Apr;17(4):421-34. |
| Additional Infomation |
- NSC624206 is a small-molecule inhibitor that selectively targets the thioesterification step of ubiquitin E1 activation, disrupting the ubiquitin-proteasome pathway and stabilizing tumor suppressor proteins like p27 [1] - Preclinical studies suggest potential efficacy in cancers with p27 degradation defects, such as colorectal and breast cancers [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4360 mL | 12.1800 mL | 24.3599 mL | |
| 5 mM | 0.4872 mL | 2.4360 mL | 4.8720 mL | |
| 10 mM | 0.2436 mL | 1.2180 mL | 2.4360 mL |