NS11394 is a potent, selective and novel positive allosteric modulator of GABA(A) receptor. NS11394 possesses a functional selectivity profile at GABA(A) receptors of alpha(5) > alpha(3) > alpha(2) > alpha(1) based on oocyte electrophysiology with human GABA(A) receptors. Compared with other subtype-selective ligands, NS11394 is unique in having superior efficacy at GABA(A)-alpha(3) receptors while maintaining low efficacy at GABA(A)-alpha(1) receptors. NS11394 has an excellent pharmacokinetic profile, which correlates with pharmacodynamic endpoints (CNS receptor occupancy), yielding a high level of confidence in deriving in vivo conclusions anchored to an in vitro selectivity profile and allowing for translation to higher species.
Physicochemical Properties
| Molecular Formula | C23H19N3O | |
| Molecular Weight | 353.42 | |
| Exact Mass | 353.152 | |
| CAS # | 951650-22-9 | |
| Related CAS # |
|
|
| PubChem CID | 16747643 | |
| Appearance | White to yellow solid powder | |
| Density | 1.2±0.1 g/cm3 | |
| Boiling Point | 623.7±65.0 °C at 760 mmHg | |
| Flash Point | 331.0±34.3 °C | |
| Vapour Pressure | 0.0±1.9 mmHg at 25°C | |
| Index of Refraction | 1.637 | |
| LogP | 4.16 | |
| Hydrogen Bond Donor Count | 1 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 3 | |
| Heavy Atom Count | 27 | |
| Complexity | 567 | |
| Defined Atom Stereocenter Count | 0 | |
| InChi Key | HLKYSQGBIIIQJN-UHFFFAOYSA-N | |
| InChi Code | InChI=1S/C23H19N3O/c1-23(2,27)18-10-11-22-21(13-18)25-15-26(22)19-8-5-7-16(12-19)20-9-4-3-6-17(20)14-24/h3-13,15,27H,1-2H3 | |
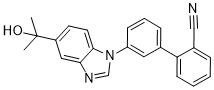
| Chemical Name | 2-[3-[5-(2-hydroxypropan-2-yl)benzimidazol-1-yl]phenyl]benzonitrile | |
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
NS-11394 targets γ-aminobutyric acid type A (GABAA) receptors with subtype selectivity: EC50 = 0.12 μM (α2β3γ2), EC50 = 0.15 μM (α3β3γ2), EC50 = 0.8 μM (α5β3γ2); no significant activity (EC50 > 10 μM) at α1β3γ2, α4β3γ2, α6β3γ2 subtypes [1] NS-11394 acts as a positive allosteric modulator (PAM) of GABAA receptors, enhancing GABA-induced chloride currents without direct agonist activity [1][2][3] |
| ln Vitro |
In vitro activity: NS11394 is unique in having superior efficacy at GABA(A)-alpha(3) receptors while maintaining low efficacy at GABA(A)-alpha(1) receptors. NS11394 has an excellent pharmacokinetic profile, which correlates with pharmacodynamic endpoints (CNS receptor occupancy), yielding a high level of confidence in deriving in vivo conclusions anchored to an in vitro selectivity profile and allowing for translation to higher species Kinase Assay: NS11394 is a potent and subtype-selective GABA(A) receptor-positive modulator; possesses a functional efficacy selectivity profile of alpha(5) > alpha(3) > alpha(2) > alpha(1) at GABA(A) alpha subunit-containing receptors. Cell Assay: In HEK293 cells stably expressing human α2β3γ2 GABAA receptors, NS-11394 dose-dependently enhanced GABA (1 μM)-induced chloride currents with an EC50 of 0.12 μM, achieving a maximal enhancement of ~280% at 10 μM [1] - In α3β3γ2 GABAA receptor-expressing HEK293 cells, NS-11394 (0.01-10 μM) enhanced GABA-induced currents with an EC50 of 0.15 μM and maximal enhancement of ~250% [1] - For α5β3γ2 GABAA receptors, NS-11394 exhibited weaker activity (EC50 = 0.8 μM) with maximal enhancement of ~180% at 10 μM [1] - No significant enhancement of GABA-induced currents was observed at α1β3γ2, α4β3γ2, or α6β3γ2 receptors (enhancement < 20% at 10 μM) [1] - NS-11394 (up to 10 μM) did not exhibit direct agonist activity at any GABAA subtype (no current induction in the absence of GABA) [1] - In primary rat hippocampal neurons, NS-11394 (0.3 μM) enhanced spontaneous inhibitory postsynaptic currents (sIPSCs) amplitude by ~45% without altering frequency, confirming modulation of native GABAA receptors [1] |
| ln Vivo |
In the formalin test, NS11394 (1-120 mg/kg) selectively reduces injury-induced nociceptive behaviors[2]. In CFA rats, NS11394 (1–10 mg/kg) significantly reduces the hindpaw weight bearing deficit [F(4,61) = 7.569, p < 0.001][2]. Anxiolytic activity: In Wistar rats subjected to the elevated plus maze (EPM) test, oral administration of NS-11394 (1 mg/kg, 3 mg/kg, 10 mg/kg) dose-dependently increased open-arm exploration: 3 mg/kg treatment increased open-arm time from 22 ± 3% (vehicle) to 48 ± 5% and open-arm entries from 18 ± 2 to 35 ± 4 [1][2] - In the light-dark box (LDB) test, oral NS-11394 (3 mg/kg) increased time spent in the light compartment from 35 ± 4 seconds (vehicle) to 82 ± 7 seconds in rats [1] - Compared to diazepam (1 mg/kg, i.p.), NS-11394 (3 mg/kg, p.o.) exhibited comparable anxiolytic efficacy but no sedative effects (no reduction in locomotor activity) [2] - Analgesic activity: In C57BL/6 mice subjected to the formalin test, intraperitoneal NS-11394 (3 mg/kg, 10 mg/kg) dose-dependently reduced nociceptive responses: 10 mg/kg inhibited phase II (inflammatory pain) licking time by ~55% (vehicle: 210 ± 25 seconds vs. drug: 95 ± 18 seconds) [3] - In the hot plate test, NS-11394 (10 mg/kg, i.p.) increased paw withdrawal latency from 12 ± 1.5 seconds (vehicle) to 22 ± 2.3 seconds in mice, indicating antinociceptive effects [3] - Memory and motor function: In the Morris water maze test, NS-11394 (3 mg/kg, 10 mg/kg, p.o.) did not impair spatial memory (escape latency and target quadrant time comparable to vehicle) in rats, whereas diazepam (1 mg/kg, i.p.) significantly increased escape latency [2][3] - In the rotarod test, NS-11394 (up to 10 mg/kg, p.o.) did not affect motor coordination (rotarod latency > 180 seconds, same as vehicle) in rats, while diazepam (1 mg/kg, i.p.) reduced latency to ~80 seconds [2][3] |
| Enzyme Assay |
GABAA subtype-specific current enhancement assay: HEK293 cells were transfected with cDNAs encoding human GABAA receptor subunits (α2β3γ2, α3β3γ2, α5β3γ2, etc.). Whole-cell patch clamp recordings were performed using intracellular and extracellular solutions optimized for chloride current detection. Cells were pre-incubated with serial dilutions of NS-11394 (0.01-10 μM) for 5 minutes, followed by application of GABA (1 μM, submaximal concentration) to induce currents. Current amplitude was recorded, and EC50 values were calculated from dose-response curves of current enhancement [1] - Direct agonist activity assay: The same patch clamp protocol was used without GABA application. NS-11394 (0.01-10 μM) was applied alone to transfected HEK293 cells to assess direct receptor activation (current induction) [1] - Native neuron sIPSC assay: Primary rat hippocampal neurons were cultured on glass coverslips. Whole-cell patch clamp recordings were performed to measure sIPSCs before and after application of NS-11394 (0.3 μM). Amplitude and frequency of sIPSCs were quantified to evaluate modulation of native GABAA receptors [1] |
| Cell Assay |
GABAA receptor-expressing HEK293 cell culture and transfection: HEK293 cells were seeded in 6-well plates or on glass coverslips and transfected with GABAA receptor subunit cDNAs using transfection reagents. After 48-hour incubation, cells were used for patch clamp recordings. Cells were maintained in serum-containing medium at 37°C with 5% CO2 [1] - Primary hippocampal neuron culture: Rat embryonic hippocampi were dissected, enzymatically dissociated, and plated on poly-L-lysine-coated coverslips. Neurons were cultured in neurobasal medium supplemented with growth factors for 14-21 days before patch clamp experiments. NS-11394 was added to the culture medium at specified concentrations, and sIPSCs were recorded [1] |
| Animal Protocol |
Animal/Disease Models: Adult male SD (Sprague-Dawley) rats[2]. Doses: 1-120 mg/kg. Route of Administration: Orally. Experimental Results: Dramatically attenuated motor function compared with corresponding vehicle responses. Dramatically decreased flinching behavior during interphase [F(3,30) = 4.139, p < 0.05] and the second phase [F(3,30) = 11.033, p < 0.001] of the formalin test compared with vehicle treatment indicative of a selective effect on injury-induced nociceptive transmission. Elevated Plus Maze (EPM) anxiolytic assay: Male Wistar rats (200-250 g) were randomly divided into vehicle control and NS-11394 (1 mg/kg, 3 mg/kg, 10 mg/kg) groups (n=8 per group). The drug was dissolved in 10% DMSO + 90% physiological saline and administered by oral gavage 60 minutes before testing. Rats were placed in the center of the EPM (4 arms: 2 open, 2 closed), and behavior was recorded for 5 minutes. Open-arm time, open-arm entries, and total locomotor activity were quantified [1][2] - Light-Dark Box (LDB) assay: Rats were treated with NS-11394 (3 mg/kg, p.o.) or vehicle 60 minutes before testing. The LDB consisted of a light compartment (200 lux) and dark compartment (0 lux) connected by an opening. Rats were placed in the dark compartment, and time spent in the light compartment, number of transitions, and locomotor activity were recorded for 10 minutes [1] - Formalin test for analgesia: Male C57BL/6 mice (20-25 g) were divided into vehicle and NS-11394 (3 mg/kg, 10 mg/kg) groups (n=7 per group). The drug was administered by intraperitoneal injection 30 minutes before subcutaneous injection of 5% formalin (20 μL) into the hind paw. Licking/biting time of the injected paw was recorded in two phases (phase I: 0-5 minutes; phase II: 15-30 minutes) [3] - Hot plate test: Mice were treated with NS-11394 (10 mg/kg, i.p.) or vehicle 30 minutes before placement on a hot plate (55 ± 0.5°C). Paw withdrawal latency was recorded (cut-off time: 30 seconds) to avoid tissue damage [3] - Morris Water Maze (MWM) memory assay: Rats were trained in the MWM (1.2 m diameter pool, hidden platform) for 5 days (4 trials/day). On day 6, NS-11394 (3 mg/kg, 10 mg/kg, p.o.) or diazepam (1 mg/kg, i.p.) was administered 60 minutes before the probe trial. Escape latency, time spent in the target quadrant, and number of platform crossings were recorded [2][3] - Rotarod motor coordination assay: Rats were trained on a rotarod (10 rpm acceleration) for 3 days (3 trials/day). On day 4, NS-11394 (up to 10 mg/kg, p.o.) or diazepam (1 mg/kg, i.p.) was administered 60 minutes before testing. Latency to fall from the rotarod was recorded (cut-off time: 300 seconds) [2][3] |
| ADME/Pharmacokinetics |
Oral bioavailability: In rats, oral administration of NS-11394 (10 mg/kg) resulted in an oral bioavailability of ~70% [1] - Plasma half-life (t1/2): In rats, t1/2 = 3.5 ± 0.4 hours (oral 10 mg/kg); in mice, t1/2 = 2.8 ± 0.3 hours (intraperitoneal 10 mg/kg) [1] - Peak plasma concentration (Cmax): In rats, oral 10 mg/kg achieved Cmax = 1.2 ± 0.15 μg/mL at 1.0 ± 0.2 hours post-dosing [1] - Area under the plasma concentration-time curve (AUC0-∞): In rats, AUC0-∞ = 4.8 ± 0.6 μg·h/mL (oral 10 mg/kg) [1] - Volume of distribution (Vd/F): In rats, Vd/F = 8.3 ± 1.1 L/kg (oral 10 mg/kg) [1] - Clearance (CL/F): In rats, CL/F = 5.2 ± 0.7 mL/min/kg (oral 10 mg/kg) [1] - Metabolism: NS-11394 is primarily metabolized in the liver via glucuronidation; no significant metabolism by CYP450 isoforms was observed [1] - Excretion: Within 72 hours of oral administration in rats, ~65% of the dose was excreted in feces (mainly as metabolites) and ~20% in urine (glucuronide conjugates) [1] |
| Toxicity/Toxicokinetics |
In vitro cytotoxicity: NS-11394 (up to 100 μM) did not affect the viability of HEK293 cells or primary hippocampal neurons (MTT assay, viability > 90% vs. vehicle) [1] - Acute toxicity in rats: Single oral administration of NS-11394 up to 200 mg/kg did not cause mortality or overt toxicity (lethargy, ataxia, weight loss) [1][2] - Chronic toxicity in rats: Repeated oral administration of NS-11394 (10 mg/kg/day for 14 days) did not induce significant changes in hematological parameters (RBC, WBC, platelets) or serum biochemical markers (ALT, AST, creatinine, BUN) [1] - Plasma protein binding: NS-11394 exhibited plasma protein binding of 92 ± 2% in rat plasma and 90 ± 3% in human plasma (equilibrium dialysis) [1] - No sedative, motor-impairing, or memory-impairing effects were observed at therapeutic doses (1-10 mg/kg), distinguishing it from non-selective GABAA PAMs (e.g., diazepam) [2][3] |
| References |
[1]. NS11394 [3 -[5-(1-Hydroxy-1-methyl-ethyl)-benzoimidazol-1-yl]-biphenyl-2-carbonitrile] a Unique Subtype-Selective GABAA Receptor Positive Allosteric Modulator: In Vitro Actions, Pharmacokinetic Properties and in Vivo Anxiolytic Effica. [2]. Comparison of the Novel Subtype-Selective GABAA Receptor-Positive Allosteric Modulator NS11394 [3′-[5-(1-Hydroxy-1-methyl-ethyl)-benzoimidazol-1-yl]-biphenyl-2-carbonitrile] with Diazepam, Zolpidem, Bretazenil, and Gaboxadol in Rat. [3]. Assessment of the effects of NS11394 and L-838417, a2/3 subunit-selective GABAA receptor-positive allosteric modulators, in tests for pain, anxiety, memory and motor function. Behavioural Pharmacology 2012, 23:790–801. |
| Additional Infomation |
NS-11394 (chemical name: 3-[5-(1-hydroxy-1-methyl-ethyl)-benzoimidazol-1-yl]-biphenyl-2-carbonitrile) is a novel, subtype-selective positive allosteric modulator (PAM) of GABAA receptors [1][2][3] - The therapeutic mechanism of NS-11394 involves selective enhancement of GABAA receptors containing α2, α3, and α5 subunits (predominantly expressed in anxiety-related brain regions and pain pathways), while sparing α1-containing receptors (linked to sedation and memory impairment) [1][2][3] - NS-11394 was developed for the treatment of anxiety disorders and pain, with the advantage of lacking sedative, motor-coordination-impairing, or memory-impairing side effects associated with non-selective GABAA modulators (e.g., diazepam, zolpidem) [1][2][3] - Preclinical data demonstrate potent anxiolytic and analgesic efficacy in rodent models, favorable pharmacokinetic profiles (good oral bioavailability, moderate half-life), and high safety margins [1][2][3] - NS-11394 exhibits comparable anxiolytic efficacy to diazepam but with superior safety profiles, and significant analgesic effects in inflammatory and thermal pain models [2][3] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.07 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (7.07 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (7.07 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8295 mL | 14.1475 mL | 28.2949 mL | |
| 5 mM | 0.5659 mL | 2.8295 mL | 5.6590 mL | |
| 10 mM | 0.2829 mL | 1.4147 mL | 2.8295 mL |