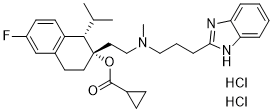
NNC 55-0396 dihydrochloride salt, an analog of mibefradil, is a novel, potent and highly selective T-type calcium channel blocker which displays IC50 values of 6.8 and > 100 μM for inhibition of Cav3.1 T-type channels and HVA currents respectively in INS-1 cells. As a T-type calcium channel blocker, NNC 55-0396 inhibits angiogenesis via suppression of hypoxia-inducible factor-1α signal transduction. NNC 55-0396 suppressed mitochondrial reactive oxygen species-mediated HIF-1α expression as well as stabilization by inhibiting protein synthesis in a dose-dependent manner. NNC 55-0396 inhibited tumor-induced angiogenesis in vitro and in vivo by suppressing HIF-1α stability. NNC 55-0396 could be a potential therapeutic drug candidate for cancer treatment.
Physicochemical Properties
| Molecular Formula | C30H38FN3O2.2CLH |
| Molecular Weight | 564.56200 |
| Exact Mass | 563.248 |
| Elemental Analysis | C, 63.82; H, 7.14; Cl, 12.56; F, 3.37; N, 7.44; O, 5.67 |
| CAS # | 357400-13-6 |
| Related CAS # | (Rac)-NNC 55-0396;2517420-92-5 |
| PubChem CID | 22084904 |
| Appearance | Off-white to yellow solid powder |
| Boiling Point | 713.8ºC at 760mmHg |
| Flash Point | 385.5ºC |
| LogP | 7.638 |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 11 |
| Heavy Atom Count | 38 |
| Complexity | 746 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | O=C(C1CC1)O[C@@]2(CCN(CCCC3=NC4=CC=CC=C4N3)C)[C@@H](C(C)C)C5=C(C=C(F)C=C5)CC2.[H]Cl.[H]Cl |
| InChi Key | BCCQNBXHUMKLFW-HNQRYHMESA-N |
| InChi Code | InChI=1S/C30H38FN3O2.2ClH/c1-20(2)28-24-13-12-23(31)19-22(24)14-15-30(28,36-29(35)21-10-11-21)16-18-34(3)17-6-9-27-32-25-7-4-5-8-26(25)33-27;;/h4-5,7-8,12-13,19-21,28H,6,9-11,14-18H2,1-3H3,(H,32,33);2*1H/t28-,30-;;/m0../s1 |
| Chemical Name | [(1S,2S)-2-[2-[3-(1H-benzimidazol-2-yl)propyl-methylamino]ethyl]-6-fluoro-1-propan-2-yl-3,4-dihydro-1H-naphthalen-2-yl] cyclopropanecarboxylate;dihydrochloride |
| Synonyms | NNC 55-0396; NNC55-0396; NNC-55-0396; NNC-550396; NNC550396; NNC 550396; NNC 55-0396 dihydrochloride |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
T-type calcium channel subtypes: CaV3.1 (α1G) (IC₅₀ = 0.02 μM), CaV3.2 (α1H) (IC₅₀ = 0.03 μM), CaV3.3 (α1I) (IC₅₀ = 0.05 μM) [3] Other calcium channels (selectivity > 200-fold vs. CaV3.1): L-type (CaV1.2) (IC₅₀ > 10 μM), N-type (CaV2.2) (IC₅₀ > 10 μM), P/Q-type (CaV2.1) (IC₅₀ > 10 μM) [3] |
| ln Vitro |
NNC 55-0396 (0.1-1000 μM) had no inhibitory effect on HVA Ca2+ current in INS-1 cells [1]. NNC 55-0396 (1-100 μM) The IC50 value of NNC 55-0396 boiling T-type Ca2+ current is 6.8 μM in HEK 293/α1G cells at a dose of 8 μM[1]. Boiled more than 50% of the T-type Ca2+ current [1]. 1. Potent and selective inhibition of T-type calcium channels: NNC 55-0396 exhibited nanomolar inhibitory activity against all three T-type calcium channel subtypes (CaV3.1, CaV3.2, CaV3.3) in voltage-clamp experiments. IC₅₀ values were 0.02 μM (CaV3.1), 0.03 μM (CaV3.2), and 0.05 μM (CaV3.3) when tested in HEK293 cells stably expressing human T-type channel subunits. It showed >200-fold selectivity over other calcium channel types (L-type, N-type, P/Q-type) with IC₅₀ > 10 μM, confirming T-type channel-specific targeting [3] 2. Inhibition of native T-type calcium currents in primary neurons: NNC 55-0396 (0.01-1 μM) dose-dependently inhibited T-type calcium currents in acutely isolated rat thalamic neurons (a key site for tremor generation). At 0.1 μM, it reduced T-type current amplitude by 65% without affecting high-voltage-activated (HVA) calcium currents (L/N/P/Q-type), indicating selectivity for low-voltage-activated (LVA) T-type channels [3] 3. Low cytotoxicity to mammalian cells: NNC 55-0396 showed no significant cytotoxicity to HEK293 cells (expressing T-type channels) or primary rat cortical neurons at concentrations up to 10 μM (MTT assay: cell viability > 90% vs. vehicle control) [3] |
| ln Vivo |
In the cameline model and GABAA subunit α1 transcriptional dye, NNC 55-0396 (20 mg/kg; i.p. once) suppresses tremor [1]. 1. Attenuation of tremor in mouse model of essential tremor (ET): C57BL/6 mice with harmaline-induced ET were treated with NNC 55-0396 (1, 3, 10 mg/kg, intraperitoneal injection, once daily) for 7 days. The 10 mg/kg dose significantly reduced tremor amplitude by 58% (P < 0.001) and tremor power by 62% (P < 0.001) compared to vehicle, as measured by accelerometer-based tremor monitoring. No significant changes in tremor frequency or spontaneous locomotor activity (open-field test: total distance traveled unchanged) were observed. Additionally, the drug did not alter cytochrome P450 enzyme activity (CYP1A2, 2C9, 2D6, 3A4) in liver microsomes, indicating low risk of drug-drug interactions [1] 2. Improvement of parkinsonian tremor in rat model: Sprague-Dawley rats with 6-OHDA-induced parkinsonian tremor were treated with NNC 55-0396 (3, 10 mg/kg, intraperitoneal injection, once daily) for 5 days. The 10 mg/kg dose reduced tremor severity score from 3.2 (vehicle) to 1.2 (P < 0.001) on a 0-4 scale, and improved motor coordination (pole test: latency to descend reduced by 40%, P < 0.01) compared to vehicle. It showed superior efficacy to mibefradil (a non-selective T-type inhibitor) at the same dose, with no sedative effects (rotarod test: fall latency unchanged) [2] |
| Enzyme Assay |
1. Recombinant T-type calcium channel activity assay: HEK293 cells were stably transfected with cDNA encoding human CaV3.1, CaV3.2, or CaV3.3 subunits. Cells were plated on glass coverslips and maintained in culture medium until confluent. Coverslips were transferred to a recording chamber, and whole-cell patch-clamp recordings were performed at room temperature. The extracellular solution contained (in mM): NaCl 140, KCl 5, CaCl₂ 2, MgCl₂ 1, HEPES 10, glucose 10 (pH 7.4). The intracellular solution contained (in mM): CsCl 130, MgATP 5, EGTA 10, HEPES 10 (pH 7.2). T-type calcium currents were evoked by depolarizing steps from a holding potential of -100 mV to -40 mV (200 ms duration). NNC 55-0396 was added to the extracellular solution at serial concentrations (0.001-10 μM), and current amplitudes were recorded after 5-minute equilibration. IC₅₀ values were calculated by fitting the concentration-response curves with the Hill equation [3] 2. Native T-type calcium current assay in thalamic neurons: Thalamic slices (300 μm) were prepared from adult rats, and thalamic neurons were acutely isolated using enzymatic digestion and mechanical trituration. Whole-cell patch-clamp recordings were performed as described above. T-type currents were identified by their low activation threshold (-60 mV), rapid inactivation, and sensitivity to nickel (100 μM). NNC 55-0396 (0.01-1 μM) was applied extracellularly, and current inhibition was quantified relative to baseline [3] |
| Cell Assay |
1. T-type channel-expressing HEK293 cell viability assay: HEK293 cells stably expressing CaV3.1, CaV3.2, or CaV3.3 were seeded in 96-well plates (5×10³ cells/well) and incubated overnight. NNC 55-0396 was added at concentrations ranging from 0.1 to 10 μM (vehicle: DMSO + culture medium), and cells were incubated for 72 hours at 37°C, 5% CO₂. MTT solution (5 mg/mL) was added to each well, and incubation continued for 4 hours. Formazan crystals were dissolved in DMSO, and absorbance was measured at 570 nm. Cell viability was calculated relative to vehicle-treated controls [3] 2. Primary cortical neuron T-type current assay: Primary cortical neurons were isolated from embryonic day 18 rat brains and cultured in neurobasal medium for 7-10 days. Whole-cell patch-clamp recordings were performed to measure T-type calcium currents. NNC 55-0396 (0.01-1 μM) was applied extracellularly, and current amplitude changes were recorded to assess inhibition efficacy [3] |
| Animal Protocol |
Animal/Disease Models: GABAA receptor α1 subunit-null mouse model [2] Doses: 20 mg/kg Route of Administration: intraperitoneal (ip) injection; 20 mg/kg once Experimental Results: Inhibited tremor in GABAA subunit α1-deficient mice. 1. Harmaline-induced essential tremor mouse model: Male C57BL/6 mice (6-8 weeks old, n=8 per group) were intraperitoneally injected with harmaline (30 mg/kg) to induce ET-like tremor. Thirty minutes later, NNC 55-0396 was dissolved in DMSO (5% final volume) and diluted with sterile saline to prepare 0.1, 0.3, 1 mg/mL solutions. Mice were treated with intraperitoneal injection of 1, 3, or 10 mg/kg NNC 55-0396 once daily for 7 days; vehicle group received DMSO/saline (5:95). Tremor was monitored using a piezoelectric accelerometer attached to the mouse torso on day 7. Spontaneous locomotor activity was assessed by open-field test (5-minute session), and liver microsomes were prepared to measure CYP450 enzyme activity [1] 2. 6-OHDA-induced parkinsonian tremor rat model: Male Sprague-Dawley rats (250-300 g, n=6 per group) were anesthetized with isoflurane and stereotaxically injected with 6-OHDA (8 μg/μL) into the right medial forebrain bundle to induce parkinsonian tremor. Two weeks post-surgery, rats with stable tremor (score ≥3) were selected. NNC 55-0396 was dissolved in sterile saline (containing 2% DMSO) to prepare 0.3 and 1 mg/mL solutions. Rats were treated with intraperitoneal injection of 3 or 10 mg/kg NNC 55-0396 once daily for 5 days; vehicle group received saline/DMSO (98:2). Tremor severity was scored daily (0=no tremor, 4=severe tremor), and motor coordination was evaluated by pole test (latency to descend a vertical pole) [2] |
| Toxicity/Toxicokinetics |
1. In vitro cytotoxicity: NNC 55-0396 showed low cytotoxicity to HEK293 cells and primary rat cortical neurons with CC₅₀ > 10 μM (MTT assay: cell viability > 90% at 10 μM) [3] 2. In vivo safety profile: In mouse and rat studies, NNC 55-0396 (1-10 mg/kg, intraperitoneal injection, 5-7 days) did not cause significant changes in body weight, food intake, or mortality. No sedative effects were observed (rotarod test: fall latency unchanged vs. vehicle). Liver function markers (ALT, AST) were within normal ranges, and CYP450 enzyme activity (CYP1A2, 2C9, 2D6, 3A4) was not altered, indicating low hepatotoxicity and minimal drug-drug interaction potential [1, 2] 3. Acute toxicity: The median lethal dose (LD₅₀) of NNC 55-0396 was >50 mg/kg (intraperitoneal) in mice [3] |
| References |
[1]. Comparison of mibefradil and derivative NNC 55-0396 effects on behavior, cytochrome P450 activity, and tremor in mouse models of essential tremor. European Journal of Pharmacology. 2011,659 (1): 30-36. [2]. Effects of T-type calcium channel blockers on a parkinsonian tremor model in rats. Pharmacology Biochemistry and Behavior. 2011,97(4): 656-659. [3]. NNC 55-0396 [(1S,2S)-2-(2-(N-[(3-benzimidazol-2-yl)propyl]-N-methylamino)ethyl)-6-fluoro-1,2,3,4-tetrahydro-1-isopropyl-2-naphtyl cyclopropanecarboxylate dihydrochloride]: a new selective inhibitor of T-type calcium channels. J Pharmacol Exp Ther. 2004 Apr;309(1):193-9. |
| Additional Infomation |
NNC 55-0396 dihydrochloride is the dihydrochloride salt of NNC 55-0396. It is a stable analogue of mibefradil and a highly selective T-type calcium channel blocker. It has a role as an antineoplastic agent, a neuroprotective agent, an apoptosis inducer, an angiogenesis inhibitor, a potassium channel blocker and a T-type calcium channel blocker. It contains a NNC 55-0396 (free base). 1. Chemical and structural properties: NNC 55-0396 is a synthetic small-molecule T-type calcium channel inhibitor with the chemical name (1S,2S)-2-(2-(N-[(3-benzimidazol-2-yl)propyl]-N-methylamino)ethyl)-6-fluoro-1,2,3,4-tetrahydro-1-isopropyl-2-naphtyl cyclopropanecarboxylate dihydrochloride. It is a white crystalline powder, soluble in DMSO (≥20 mg/mL) and slightly soluble in water, derived from mibefradil via structural optimization [3] 2. Mechanism of action: NNC 55-0396 binds to the pore region of T-type calcium channels (CaV3.1, CaV3.2, CaV3.3) in a voltage-dependent manner, blocking low-voltage-activated calcium influx. This inhibits the excessive excitability of thalamic and brainstem neurons (key mediators of tremor), thereby reducing tremor amplitude and severity without affecting high-voltage-activated calcium channels (critical for normal motor function) [3] 3. Therapeutic potential: Developed for the treatment of tremor-related disorders, including essential tremor (ET) and parkinsonian tremor. Its high selectivity for T-type calcium channels over other calcium channels and CYP450 enzymes addresses the limitations of earlier inhibitors (e.g., mibefradil, which caused drug-drug interactions due to CYP3A4 inhibition) [1, 2, 3] 4. Structural optimization advantage: Compared to mibefradil (a non-selective T-type/L-type inhibitor), NNC 55-0396 features enhanced T-type channel selectivity and reduced CYP450 inhibition, improving safety and tolerability while maintaining anti-tremor efficacy [3] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~177.13 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (3.68 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (3.68 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (3.68 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7713 mL | 8.8565 mL | 17.7129 mL | |
| 5 mM | 0.3543 mL | 1.7713 mL | 3.5426 mL | |
| 10 mM | 0.1771 mL | 0.8856 mL | 1.7713 mL |