|
NMS-P118 (NMS-P-118) is a novel potent, orally bioavailable, and highly selective PARP-1 (protein poly(ADP-ribose) polymerase-1) inhibitor with anticancer activity. It demonstrates a 150-fold preference for PARP-1 over PARP-2 (Kd 0.009 μM versus 1.39 μM, in that order). Treatment for cancer with it has been studied. Out of eight isoforms tested, NMS-P118 modestly inhibits two members of the cytochrome P450 family (CYP-2B6 IC50, 8.15 μM; CYP-2D6 IC50, 9.51 μM). It also demonstrates excellent ADME and pharmacokinetic properties and is metabolically stable. NMS-P118 has a full oral bioavailability and a low in vivo clearance.
|
Physicochemical Properties
| Molecular Formula |
C20H24F3N3O2
|
| Molecular Weight |
395.42
|
| Exact Mass |
395.18
|
| Elemental Analysis |
C, 60.75; H, 6.12; F, 14.41; N, 10.63; O, 8.09
|
| CAS # |
1262417-51-5
|
| Related CAS # |
|
| PubChem CID |
49843531
|
| Appearance |
White to off-white solid powder
|
| Density |
1.4±0.1 g/cm3
|
| Boiling Point |
508.3±50.0 °C at 760 mmHg
|
| Flash Point |
261.2±30.1 °C
|
| Vapour Pressure |
0.0±1.3 mmHg at 25°C
|
| Index of Refraction |
1.591
|
| LogP |
0.15
|
| Hydrogen Bond Donor Count |
1
|
| Hydrogen Bond Acceptor Count |
6
|
| Rotatable Bond Count |
3
|
| Heavy Atom Count |
28
|
| Complexity |
614
|
| Defined Atom Stereocenter Count |
0
|
| SMILES |
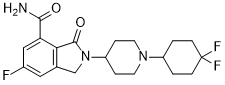
FC1(C([H])([H])C([H])([H])C([H])(C([H])([H])C1([H])[H])N1C([H])([H])C([H])([H])C([H])(C([H])([H])C1([H])[H])N1C(C2C(C(N([H])[H])=O)=C([H])C(=C([H])C=2C1([H])[H])F)=O)F
|
| InChi Key |
ARYVAQSYRLZVQD-UHFFFAOYSA-N
|
| InChi Code |
InChI=1S/C20H24F3N3O2/c21-13-9-12-11-26(19(28)17(12)16(10-13)18(24)27)15-3-7-25(8-4-15)14-1-5-20(22,23)6-2-14/h9-10,14-15H,1-8,11H2,(H2,24,27)
|
| Chemical Name |
2-[1-(4,4-difluorocyclohexyl)piperidin-4-yl]-6-fluoro-3-oxo-1H-isoindole-4-carboxamide
|
| Synonyms |
NMS-P118; NMS-P 118; NMS P118
|
|
| HS Tariff Code |
2934.99.9001
|
| Storage |
Powder-20°C 3 years
4°C 2 years
In solvent -80°C 6 months
-20°C 1 month
|
| Shipping Condition |
Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs)
|
|
Biological Activity
| Targets |
PARP-1 ( Kd = 9 nM ); PARP-2 ( Kd = 1390 nM )
|
| ln Vitro |
In vitro activity: NMS-P118 is discovered to be less myelotoxic in vitro than the dual PARP-1/-2 inhibitor olaparib, which is currently marketed as Lynparza. Out of eight isoforms tested, NMS-P118 demonstrates metabolic stability and modest inhibition of two cytochrome P450 family members (CYP-2B6 IC50: 8.15 μM; CYP-2D6 IC50: 9.51 μM). Depending on the species, it can inhibit bone marrow cell proliferation 5–60 times less effectively than olaparib. |
|
| ln Vivo |
| NMS-P118 is demonstrated to be metabolically stable and, of the eight isoforms examined, it only slightly inhibits two members of the cytochrome P450 family (CYP-2B6 IC50, 8.15 μM; CYP-2D6 IC50, 9.51 μM). NMS-P118 has a full oral bioavailability and a low in vivo clearance. The pharmacokinetic profile of NMS-P118 in rats administered orally at 10 and 100 mg/kg and intravenously at 10 mg/kg is similar to that seen in mice, with oral bioavailability exceeding 65% and dose-dependent exposure linearity. At 1, 2, and 6 hours after administration, its treatment significantly lowers intratumoral PAR levels; at 24 hours, partial recovery of PAR levels is seen. In BRCA1-mutant MDA-MB-436 and BRCA2 deficient Capan-1 human tumor xenograft models, respectively, NMS-P118 exhibits excellent ADME and pharmacokinetic profiles, high oral availability in mice and rats, and high efficacy both as a single agent and in combination with Temozolomide. |
|
| Enzyme Assay |
NMS-P118 is characterized by 56 distinct kinases profiles (ABL, ACK1, AKT1, ALK, AUR1, AUR2, BRK, BUB1, CDC7/DBF4, CDK2/CYCA, CHK1, CK2, EEF2K, EGFR1, ERK2, EphA2, FAK, FGFR1, FLT3, GSK3beta, Haspin, IGFR1, IKK2, IR, JAK1, JAK2, JAK3, KIT, LCK, LYN, MAPKAPK2, MELK, MET, MNK2, MPS1, MST4, NEK6, NIM1, P38alpha, PAK4, POLYDATINGFRb, POLYDATINK1, PERK, PIM1, PIM2, PKAalpha, PLK1, RET, SULU1, Syk, TLK2, TRKA, TYK2, VEGFR2, ZAP70). For every enzyme tested, the IC50 values are found to be greater than 10 μM[1].
|
| Cell Assay |
NMS-P118 is diluted with the proper medium and dissolved in DMSO prior to usage. By evaluating the inhibition of hydrogen peroxide-induced PAR formation in HeLa cells (ECACC), PARP-1 inhibitors' cellular activity is determined. ArrayScan vTi is used to quantify the levels of cellular PAR after they are determined by immunocytochemistry[1].
|
| Animal Protocol |
| Ad Hoc pharmacokinetic studies have been conducted on rats to examine the oral bioavailability and pharmacokinetic profile of the compounds. NMS-P118 is designed to be administered as an intravenous bolus in a 20% DMSO + 40% PEG 400 solution in 5% milk sugar. With 0.5% methylcellulose, an NMS-P118 suspension is used for oral drug administration. For every delivery method, there is one oral administration at a dose of 100 mg/kg and one single administration at a dose of 10 mg/kg. Each study employs three male animals[1]. | |
|
| References |
[1]. Discovery of 2-[1-(4,4-Difluorocyclohexyl)piperidin-4-yl]-6-fluoro-3-oxo-2,3-dihydro-1H-isoindole-4-carboxamide (NMS-P118): A Potent, Orally Available, and Highly Selective PARP-1 Inhibitor for Cancer Therapy. J Med Chem. 2015 Sep 10;58(17):6875-98.
|
|
Solubility Data
| Solubility (In Vitro) |
| DMSO: 16~24 mg/mL (40.5~60.7 mM) | | Water: <1 mg/mL | | Ethanol: ~5 mg/mL (~12.6 mM) |
|
| Solubility (In Vivo) |
| O=C(C1=CC(F)=CC(CN2C3CCN(C4CCC(F)(F)CC4)CC3)=C1C2=O)N |
(Please use freshly prepared in vivo formulations for optimal results.)
|
| Preparing Stock Solutions |
|
1 mg |
5 mg |
10 mg |
| 1 mM |
2.5290 mL |
12.6448 mL |
25.2896 mL |
| 5 mM |
0.5058 mL |
2.5290 mL |
5.0579 mL |
| 10 mM |
0.2529 mL |
1.2645 mL |
2.5290 mL |
*Note: Please select an appropriate solvent for the preparation of stock solution based on your experiment needs. For most products, DMSO can be used for preparing stock solutions (e.g. 5 mM, 10 mM, or 20 mM concentration); some products with high aqueous solubility may be dissolved in water directly. Solubility information is available at the above Solubility Data section. Once the stock solution is prepared, aliquot it to routine usage volumes and store at -20°C or -80°C. Avoid repeated freeze and thaw cycles. |