Physicochemical Properties
| Molecular Formula | C27H29N3O3 |
| Molecular Weight | 443.53746676445 |
| Exact Mass | 443.22 |
| CAS # | 2380230-73-7 |
| PubChem CID | 138811017 |
| Appearance | Off-white to gray solid powder |
| LogP | 1.2 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 7 |
| Heavy Atom Count | 33 |
| Complexity | 641 |
| Defined Atom Stereocenter Count | 0 |
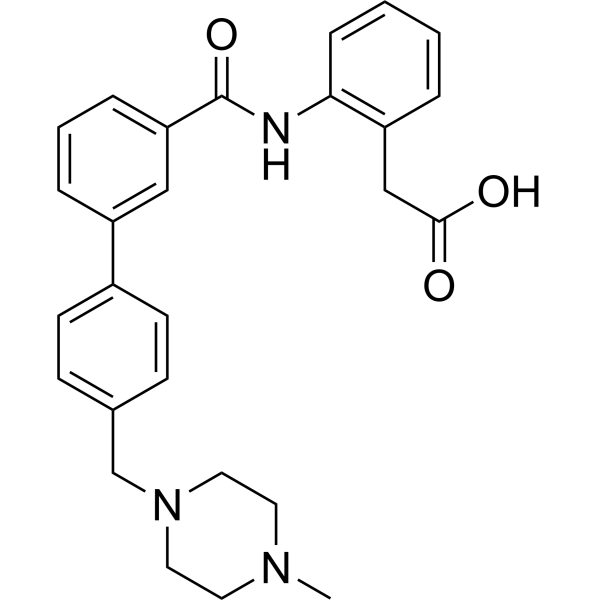
| SMILES | O=C(C1=CC=CC(=C1)C1C=CC(=CC=1)CN1CCN(C)CC1)NC1C=CC=CC=1CC(=O)O |
| InChi Key | UTWXDNZWMQAUKL-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C27H29N3O3/c1-29-13-15-30(16-14-29)19-20-9-11-21(12-10-20)22-6-4-7-24(17-22)27(33)28-25-8-3-2-5-23(25)18-26(31)32/h2-12,17H,13-16,18-19H2,1H3,(H,28,33)(H,31,32) |
| Chemical Name | 2-[2-[[3-[4-[(4-methylpiperazin-1-yl)methyl]phenyl]benzoyl]amino]phenyl]acetic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | SUCNR1 (GPR91)[1] |
| ln Vitro | Deep within the hydrophobic pocket, NF-56-EJ40 is bound. On one side of the hydrophobic pocket, the conserved residues Y832.64 and Y301.39, and on the other, R2817.39, coordinate the acid group. The preserved E181. It is anticipated that 27 will add another hydrogen bond to NF-56-EJ40's piperazine ring. Rat SUCNR1 K181.31 and K2697.32 replace human SUCNR1 E221.31 and N2747.32. Due to steric hindrance, these two amino acid substitutions may inhibit NF-56-EJ40 from binding to rat SUCNR1. Our homology model and radioligand-binding studies with human SUCNR1 partially agreed: the Y301.39F mutant of human SUCNR1 exhibits decreased binding of NF-56-EJ40. The E181.27K and E181.27R mutants exhibit similar effects, most likely as a result of steric clashes between the Lys and Arg residues of NF-56-EJ40 and the dissociation of a hydrogen bond from its piperazine ring[1]. ?The double mutant K181.31E/K2697.32N, also known as humanized rat SUCNR1, is created by introducing human SUCNR1 residues into rat SUCNR1. (Ki of 17.4 nM and human and humanized rat SUCNR1, respectively). The thermal stability of human and humanized rat SUCNR1 is increased by NF-56-EJ40, but not that of rat SUCNR1[1]. |
| References |
[1]. Structural basis of species-selective antagonist binding to the succinate receptor. Nature. 2019 Oct;574(7779):581-585. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 5 mg/mL (11.27 mM) H2O: 4.55 mg/mL (10.26 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2546 mL | 11.2729 mL | 22.5459 mL | |
| 5 mM | 0.4509 mL | 2.2546 mL | 4.5092 mL | |
| 10 mM | 0.2255 mL | 1.1273 mL | 2.2546 mL |