Physicochemical Properties
| Molecular Formula | C37H26N4NA4O15S4 |
| Molecular Weight | 986.83 |
| Exact Mass | 985.986 |
| Elemental Analysis | C, 45.03; H, 2.66; N, 5.68; Na, 9.32; O, 24.32; S, 13.00 |
| CAS # | 202982-98-7 |
| PubChem CID | 90488883 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 15 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 64 |
| Complexity | 1850 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | [Na+].[Na+].[Na+].[Na+].O=C(NC1C(C)=CC=C(C(NC2=CC(S([O-])(=O)=O)=CC3C=CC(=CC2=3)S([O-])(=O)=O)=O)C=1)NC1=C(C)C=CC(C(NC2=CC(S([O-])(=O)=O)=CC3C=CC(=CC2=3)S([O-])(=O)=O)=O)=C1 |
| InChi Key | SJMHXBFWMZYDBY-UHFFFAOYSA-J |
| InChi Code | InChI=1S/C37H30N4O15S4.4Na/c1-19-3-5-23(35(42)38-33-17-27(59(51,52)53)11-21-7-9-25(15-29(21)33)57(45,46)47)13-31(19)40-37(44)41-32-14-24(6-4-20(32)2)36(43)39-34-18-28(60(54,55)56)12-22-8-10-26(16-30(22)34)58(48,49)50;;;;/h3-18H,1-2H3,(H,38,42)(H,39,43)(H2,40,41,44)(H,45,46,47)(H,48,49,50)(H,51,52,53)(H,54,55,56);;;;/q;4*+1/p-4 |
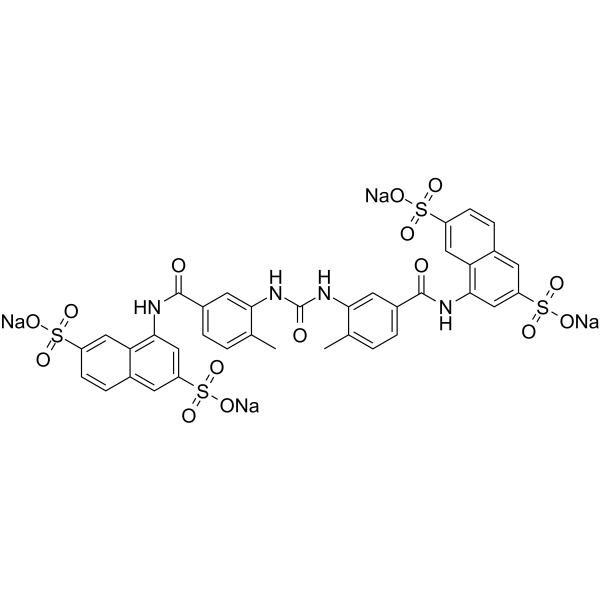
| Chemical Name | tetrasodium;4-[[3-[[5-[(3,7-disulfonatonaphthalen-1-yl)carbamoyl]-2-methylphenyl]carbamoylamino]-4-methylbenzoyl]amino]naphthalene-2,6-disulfonate |
| Synonyms | NF340; NF-340; NF 340; 202982-98-7; tetrasodium;4-[[3-[[5-[(3,7-disulfonatonaphthalen-1-yl)carbamoyl]-2-methylphenyl]carbamoylamino]-4-methylbenzoyl]amino]naphthalene-2,6-disulfonate; 4,4'-(Carbonylbis(imino-3,1-(4-methyl-phenylene)carbonylimino))bis(naphthalene-2,6-disulfonic acid) tetrasodium salt; 4,4'-(Carbonylbis(imino-3,1-(4-methyl-phenylene)carbonylimino))bis(naphthalene-2,6-disulfonicacid)tetrasodiumsalt; NF 340 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | P2Y11 receptor |
| ln Vitro |
Blockage of P2Y11R by NF340 inhibits IL-1β-induced matrix metalloproteinase protein expression as indicated by the levels of MMP-1, MMP-3, and MMP-13. Mechanistically, blockage of P2Y11R mitigates IL-1β-activated NFκB signaling, which was revealed by reduced IκBα phosphorylation, nuclear p65 accumulation, and NFκB promoter activity. Our study provides evidence of a protective mechanism of P2Y11R antagonist NF340 against cytokine-induced inflammation. Therefore, targeting P2Y11R could have potential therapeutic implication in the treatment of RA. [1] Blockage of P2Y11R inhibits IL-1β-induced expression of TNF-α and IL-6 [1] Next, we sought to elucidate the role of P2Y11R in the inflammatory response. We applied the P2Y11R-specific antagonist NF340 to block the receptor's activity and assessed the response to IL-1β-triggered inflammation in FLS. At the mRNA level and compared to nontreated cells, IL-1β induced approximately 5.8-fold TNF-α expression. However, blockage of P2Y11R by 10 and 20 μM NF340 ameliorated IL-1β-induced expression of TNF-α in a dose-dependent manner to only 3.5- and 2.2-fold, respectively (Figure 3a). Similarly, IL-1β induced 4.5-fold IL-6 mRNA expression, while IL-1β-induced expression of IL-6 was only approximately 3- and 1.5-fold in the presence of the two doses of NF340 (Figure 3a). We were able to confirm this P2Y11R-dependent inhibitory effect at the protein level. Again, when compared to nontreated cells, IL-1β induced about fivefold expression of TNF-α protein. However, IL-1β only caused approximately 3- and 1.7-fold TNF-α protein induction when the two doses of antagonist were present in the media, respectively (Figure 3b). Similarly, IL-1β induced around 4.2-fold expression of IL-6 protein. However, IL-1β only caused approximately 2.8- and 1.5-fold expression of IL-6 protein when the two doses of antagonist were present in the media, respectively (Figure 3b). Blockage of P2Y11R inhibits IL-1β-induced oxidative stress [1] We then assessed the effect of blockage of P2Y11R on cytokine-induced cellular stress response. We tested two aspects of cellular oxidative stress including directly measuring the total levels of ROS and 4-HNE, the lipid byproduct of oxidative stress. Compared to nontreated FLS, IL-1β gave rise to about fourfold the level of total cellular ROS. However, it only caused about three- and two-fold production of ROS when the two doses ofNF340 were present in the media, respectively (Figure 4a). As compared with nontreated cells, the change in the level of 4-HNE production induced by IL-1β treatment generated around 3.8-fold production of 4-HNE. However, it only produced approximately 2.5- and 1.8-fold 4-HNE production in the presence of the same two doses of NF340 (Figure 4b). Blockage of P2Y11R inhibits IL-1β-induced MMP expression [1] Increased activity of MMPs is an important feature of tissue remodeling in RA development, and it has been shown that MMPs can be induced by pro-inflammatory cytokines. We tested the effect of P2Y11R blockage on IL-1β-induced MMP expression in FLS. At the mRNA level and compared to the nontreated control, IL-1β triggered an average increase of five- to six-fold expression of MMP-1, MMP-3, and MMP-13 at the mRNA transcript level, respectively. But this induction of MMPs by IL-1β was only about two- to three-fold when 10 and 20 μM NF340 was added to the media to block P2Y11R activity (Figure 5a). At the protein level and compared to nontreated cells, IL-1β triggered an average increase of four- to five-fold in MMP-1, MMP-3, and MMP-13 mRNA protein expression, respectively. However, IL-1β only induced an average increase of two- to three-fold when the same two doses of NF340 were added to the media (Figure 5b). These changes in the MMP expression profile suggest that cytokine-induced expression of MMPs in FLS relies on the activity of P2Y11R. Blockage of P2Y11R mitigates IL-1β-induced NF-κB activation [1] Cytokine-induced pro-inflammatory factors are known to be largely dependent on NF-κB activation. We tested the status of cellular IκBα kinase phosphorylation when P2Y11R was inhibited. Compared with non-treated FLS, IL-1β induced around fivefold phosphorylation of IκBα. However, it only caused about 3.5- and 2-fold phosphorylation of IκBα when P2Y11R was blocked using the same two doses of NF340 (Figure 6). Next, we tested the status of nuclear NF-κB via direct detection of p65 accumulation and transfected NF-κB promoter activity. Nuclear p65 accumulation is the precedent event of nuclear NF-κB activation, so we compared changes in the level of p65 to nontreated cells. IL-1β induced approximately 3.2-fold more accumulation of p65 in nuclei, but only about 2- and 1.5-fold more nuclear p65 when the two doses of NF340 were added to the media, respectively (Figure 7a). When NF-κB luciferase promoter was transfected into FLS, its activity was increased to around 80-fold upon IL-1β treatment. However, the increase in promoter activity induced by IL-1β was decreased to approximately 50- and 25-fold in the presence of the two doses of NF340, respectively (Figure 7b). Together, these experiments firmly confirm P2Y11R activity is required for cytokine-induced NF-κB activation. |
| Enzyme Assay |
Promoter assay [1] The 3x NFκB luciferase vectors were purchased from Thermo Fisher Scientific. Cells were cotransfected with a GL3-Renilla control plasmid and a NF-κB–firefly reporter plasmid using Lipofectamine 2000 reagent. At 24 hr post-transfection, the cells were treated with 10 ng/mL IL-1β in presence or absence of 10 and 20 μM NF340 for an additional 24 hr. The total cell lysates were collected to measure the dual luciferase activity of renilla and firefly luciferase. The relative luciferase was calculated by normalizing the activity of firefly luciferase to renilla luciferase. |
| Cell Assay |
Cell treatment experiment [1] Human FLS were maintained in 10% FBS containing complete DMEM media. For cytokine treatment experiments, FLS were plated in 6-well plates and grown to 80 to 90% confluence. The cells were then treated with 5, 10, and 20 ng/mL of IL-1β for 24 hr. To block the activity of P2Y11R with NF340 treatment, 10 and 20 μM concentration of fresh NF340 were added to cell growth media for 24 hr. The controls were treated with the solvent DMSO. |
| References |
[1]. P2Y11 receptor antagonist NF340 ameliorates inflammation in human fibroblast-like synoviocytes: An implication in rheumatoid arthritis. IUBMB Life. 2019 Oct;71(10):1552-1560. |
| Additional Infomation |
Rheumatoid arthritis is a common chronic inflammatory joint disease. Fibroblast-like synoviocytes-mediated inflammation is closely associated with the development of rheumatoid arthritis. In this study, we report that P2Y11 receptor activity is required for cytokine-induced inflammation in primary fibroblast-like synoviocytes (FLS). P2Y11R is fairly expressed in primary FLS isolated from healthy subjects and is elevated to around three- to four-fold in rheumatoid arthritis-derived FLS. The expression of P2Y11R is inducible upon IL-1β treatment. Blockage of P2Y11R by its antagonist suppresses IL-1β-induced TNF-α and IL-6 induction and ameliorates oxidative stress as determined by levels of cellular ROS and the oxidative byproduct 4-HNE. Moreover, blockage of P2Y11R by NF340 inhibits IL-1β-induced matrix metalloproteinase protein expression as indicated by the levels of MMP-1, MMP-3, and MMP-13. Mechanistically, blockage of P2Y11R mitigates IL-1β-activated NFκB signaling, which was revealed by reduced IκBα phosphorylation, nuclear p65 accumulation, and NFκB promoter activity. Our study provides evidence of a protective mechanism of P2Y11R antagonist NF340 against cytokine-induced inflammation. Therefore, targeting P2Y11R could have potential therapeutic implication in the treatment of RA. [1] Researchers conducted a series of inhibition experiments to explore the significance of lowering P2Y11R. First, our experiments show that blockage of P2Y11R by its specific antagonist NF340 indeed weakened IL-1β-induced expression of the pro-inflammatory cytokines TNF-α and IL-6. Second, we show that blockage of this receptor by NF340 reduced IL-1β-induced cellular ROS production and decreased the oxidative stress marker 4-HNE. Third, NF340 appears to potently suppress IL-1β-induced MMP protease activity. NF340 suppresses at least three primary members of the MMP family that indicates that it could hamper the proteolytic erosive activity of FLS, thereby exerting a protective role in joints. It has been reported that FLS are responsible for eroding bone and destroying cartilage through the release of MMPs The suppression of IL-1β induction and MMP activity by NF340 implies that this compound could have therapeutic potential. Indeed, a very recent report showed that 0.3 to 20 μM NF340 reduced a variety of arthritic lesions and improved animal joint conditions after 21 day treatment in an RA rat model, suggesting that NF340 might have additional targets and effects in rats. Several purinergic receptors have been documented to be involved in the pathological development of RA. It has been reported that the P2X7 receptor is expressed by synoviocytes from the joints of RA patients. Activation of the P2X7 receptor stimulates secretion of the pro-inflammatory cytokine IL-6. Deficiency of the P2X7 receptor induced a loss of ATP-dependent leukocyte function, IL-1β production, and L-selectin shedding. Blockage of this receptor has been associated with lower incidence and severity of arthritis in animal models. Interestingly, there exists crosstalk between the P2Y11 receptor and the P2X7 receptor. The P2Y11 receptor can interfere with P2X7 receptor pore formation and thus prevent cell death of human CD8+ T lymphocytes. We found that the expression of P2X7R in RA-FLS was higher than that of the control. Indeed, several preliminary experiments in our laboratory have demonstrated that the P2X7 receptor played a key role in the inflammatory response in FLS (data not shown). Future investigations will provide us with a complete picture of the underlying mechanisms. This study has the following unanswered questions: more detailed understanding of the mechanism of action is required to evaluate the effect of NF340. One of the major problems we face is that the murine genome does not have a human P2Y11R orthologue, and there is no available P2Y11R specific knockout mouse model. Some other preclinical studies were based on rats, but still provided only a limited understanding of this receptor.13 In rats, NF340 has been shown to act on glial cells and participate in the maintenance of neuropathic pain. In human monocyte-derived dendritic cells, NF340 suppresses thrombospondin-1 secretion and reverses LPS stimulated interleukin-12 release. In conclusion, our understanding of the P2Y11R receptor and experience with its antagonist in pharmacological study are still limited. Our study using human FLS provides mechanistic insight into the effect of P2Y11R and its antagonist NF340. Nevertheless, targeting P2Y11R using NF340 could have the potential to be used in clinical trials for RA.[1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.0133 mL | 5.0667 mL | 10.1335 mL | |
| 5 mM | 0.2027 mL | 1.0133 mL | 2.0267 mL | |
| 10 mM | 0.1013 mL | 0.5067 mL | 1.0133 mL |