Physicochemical Properties
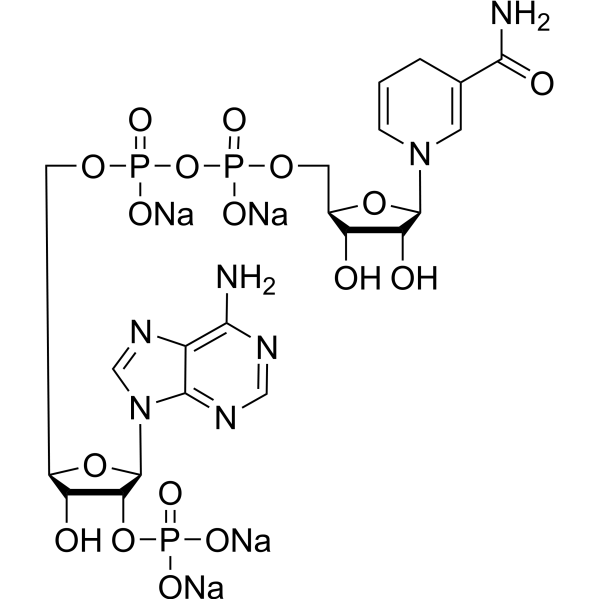
| Molecular Formula | C21H26N7O17P3 |
| Molecular Weight | 741.3891 |
| Exact Mass | 833.018 |
| CAS # | 2646-71-1 |
| Related CAS # | NADPH tetracyclohexanamine;100929-71-3 |
| PubChem CID | 5884 |
| Appearance | White to light yellow solid powder |
| Density | 2.28 g/cm C |
| Boiling Point | 1175.1ºC at 760 mmHg |
| Melting Point | >250ºC (dec.) |
| Flash Point | 664.5ºC |
| LogP | 0.402 |
| Hydrogen Bond Donor Count | 9 |
| Hydrogen Bond Acceptor Count | 22 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 48 |
| Complexity | 1410 |
| Defined Atom Stereocenter Count | 8 |
| SMILES | C1C=CN(C=C1C(=O)N)[C@H]2[C@@H]([C@@H]([C@H](O2)COP(=O)(O)OP(=O)(O)OC[C@@H]3[C@H]([C@H]([C@@H](O3)N4C=NC5=C(N=CN=C54)N)OP(=O)(O)O)O)O)O |
| InChi Key | ACFIXJIJDZMPPO-NNYOXOHSSA-N |
| InChi Code | InChI=1S/C21H30N7O17P3/c22-17-12-19(25-7-24-17)28(8-26-12)21-16(44-46(33,34)35)14(30)11(43-21)6-41-48(38,39)45-47(36,37)40-5-10-13(29)15(31)20(42-10)27-3-1-2-9(4-27)18(23)32/h1,3-4,7-8,10-11,13-16,20-21,29-31H,2,5-6H2,(H2,23,32)(H,36,37)(H,38,39)(H2,22,24,25)(H2,33,34,35)/t10-,11-,13-,14-,15-,16-,20-,21-/m1/s1 |
| Chemical Name | [[(2R,3R,4R,5R)-5-(6-aminopurin-9-yl)-3-hydroxy-4-phosphonooxyoxolan-2-yl]methoxy-hydroxyphosphoryl] [(2R,3S,4R,5R)-5-(3-carbamoyl-4H-pyridin-1-yl)-3,4-dihydroxyoxolan-2-yl]methyl hydrogen phosphate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture.(2). This product is not stable in solution, please use freshly prepared working solution for optimal results. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The de novo synthesis of NADPH tetrasodium salt is conducted by NAD kinase (NADK), which catalyzes the phosphorylation of NAD+ to generate NADP+. NADPH tetrasodium salt is predominantly involved in catabolic reactions, while NADPH tetrasodium salt is mainly involved in cellular antioxidant effects and anabolic reactions. Glutathione reductase (GR) employs NADPH tetrasodium salt to decrease oxidized glutathione (GSSG) to GSH[1]. The rate of regeneration of NADPH tetrasodium salt is often the rate-limiting step in overproduction of the desired chemical while sustaining strong cell growth [2]. NADPH tetrasodium homeostasis is regulated by numerous signaling pathways and multiple metabolic enzymes that undergo adaptive alterations in cancer cells. Metabolic reprogramming by NADPH tetrasodium salt makes cancer cells highly dependent on the antioxidant activity of this metabolic network and more vulnerable to oxidative stress [3]. NADPH tetrasodium salt is a key intracellular reducing agent required for the removal of lipid hydroperoxides. In fact, NADPH tetrasodium salt levels are a diagnostic of ferroptosis susceptibility in several cancer cell lines [4]. |
| References |
[1]. NAD+ Kinase as a Therapeutic Target in Cancer. Clin Cancer Res. 2016;22(21):5189-5195. [2]. Rational design of a synthetic Entner-Doudoroff pathway for improved and controllable NADPH regeneration. Metab Eng. 2015;29:86-96. [3]. NADPH homeostasis in cancer: functions, mechanisms and therapeutic implications. Signal Transduct Target Ther. 2020;5(1):231. Published 2020 Oct 7. [4]. Ferroptosis: A Regulated Cell Death Nexus Linking Metabolism, Redox Biology, and Disease. Cell. 2017;171(2):273-285. |
| Additional Infomation |
NADPH is the reduced form of NADP+; used in anabolic reactions, such as lipid and nucleic acid synthesis, which require NADPH as a reducing agent. It has a role as a fundamental metabolite and a cofactor. It is a NAD(P)H and a NADP. It is a conjugate acid of a NADPH(4-). NADPH is a metabolite found in or produced by Escherichia coli (strain K12, MG1655). NADPH has been reported in Homo sapiens, Bos taurus, and Schizosaccharomyces pombe with data available. NADPH is a metabolite found in or produced by Saccharomyces cerevisiae. Nicotinamide adenine dinucleotide phosphate. A coenzyme composed of ribosylnicotinamide 5'-phosphate (NMN) coupled by pyrophosphate linkage to the 5'-phosphate adenosine 2',5'-bisphosphate. It serves as an electron carrier in a number of reactions, being alternately oxidized (NADP+) and reduced (NADPH). (Dorland, 27th ed) |
Solubility Data
| Solubility (In Vitro) |
H2O : ≥ 35 mg/mL (~42.00 mM) DMSO :< 1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: 100 mg/mL (120.00 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with sonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.3488 mL | 6.7441 mL | 13.4882 mL | |
| 5 mM | 0.2698 mL | 1.3488 mL | 2.6976 mL | |
| 10 mM | 0.1349 mL | 0.6744 mL | 1.3488 mL |