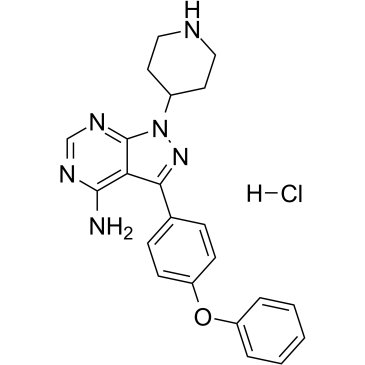
N-piperidine Ibrutinib HCl, the hydrochloride salt of N-piperidine Ibrutinib, is an intermediate used in the synthesis of BTK-targeted PROTACs such as SJF620 (DC50 of 7.9 nM for degrading BTK). N-piperidine Ibrutinib, an Ibrutinib derivative, likewise exhibits strong BTK inhibitory activity, with IC50s of 51.0 and 30.7 nM for BTK (WT) and BTK (C481S), respectively.
Physicochemical Properties
| Molecular Formula | C22H23CLN6O |
| Molecular Weight | 422.910622835159 |
| Exact Mass | 422.16 |
| Elemental Analysis | C, 62.48; H, 5.48; Cl, 8.38; N, 19.87; O, 3.78 |
| CAS # | 2231747-18-3 |
| Related CAS # | 2231747-18-3 |
| PubChem CID | 139465991 |
| Appearance | White to off-white solid |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 4 |
| Heavy Atom Count | 30 |
| Complexity | 515 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | ORBFZIXZKIUECG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C22H22N6O.ClH/c23-21-19-20(15-6-8-18(9-7-15)29-17-4-2-1-3-5-17)27-28(22(19)26-14-25-21)16-10-12-24-13-11-16;/h1-9,14,16,24H,10-13H2,(H2,23,25,26);1H |
| Chemical Name | 3-(4-phenoxyphenyl)-1-piperidin-4-ylpyrazolo[3,4-d]pyrimidin-4-amine;hydrochloride |
| Synonyms | Npiperidine; Ibrutinib HCl; N piperidine; Ibrutinib HCl |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture and light. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | WT BTK (IC50 = 51.0 nM); C481S BTK (IC50 = 30.7 nM) |
| ln Vitro | N-piperidine ibrutinib hydrochloride is a BTK ligand that can be utilized in the production of several PROTACs. Effective PROTAC BTK degraders are SJF638, SJF678, and SJF608, with DC50 values of 374, 162, and 8.3 nM, respectively [2]. |
| References |
[1]. Targeting the C481S Ibrutinib-Resistance Mutation in Bruton's Tyrosine Kinase Using PROTAC-Mediated Degradation. Biochemistry. 2018 Jul 3;57(26):3564-3575. [2]. Design, synthesis and biological evaluation of Proteolysis Targeting Chimeras (PROTACs) as a BTK degraders with improved pharmacokinetic properties. Bioorg Med Chem Lett. 2020 Feb 1;30(3):126877. |
Solubility Data
| Solubility (In Vitro) |
Water: ~85 mg/mL (~201 mM) DMSO: ~11 mg/mL (~26.0 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (5.91 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: 2.5 mg/mL (5.91 mM) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (5.91 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3646 mL | 11.8228 mL | 23.6457 mL | |
| 5 mM | 0.4729 mL | 2.3646 mL | 4.7291 mL | |
| 10 mM | 0.2365 mL | 1.1823 mL | 2.3646 mL |