Physicochemical Properties
| Molecular Formula | C16H13N |
| Molecular Weight | 219.28 |
| Exact Mass | 219.104 |
| CAS # | 90-30-2 |
| PubChem CID | 7013 |
| Appearance |
Prisms or needles from alcohol; leaflets from ligroin. Tan to purple crushed solid or crystals. White to yellowish crystals In its pure form crystallizes into lemon yellow prisms or needles ... marketed in the form of brown to dark violet crystals or light brown to light violet granules |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 370.7±0.0 °C at 760 mmHg |
| Melting Point | 60-62 °C(lit.) |
| Flash Point | 190.2±14.7 °C |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.702 |
| LogP | 4.2 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 17 |
| Complexity | 232 |
| Defined Atom Stereocenter Count | 0 |
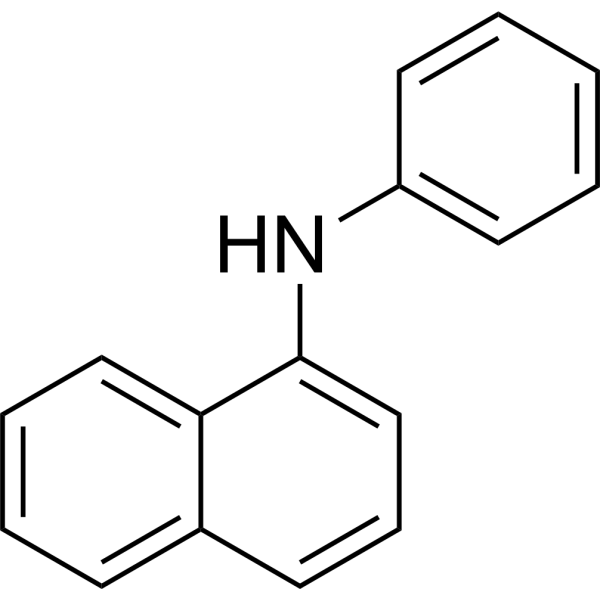
| SMILES | C1C=CC(NC2C3C(=CC=CC=3)C=CC=2)=CC=1 |
| InChi Key | XQVWYOYUZDUNRW-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C16H13N/c1-2-9-14(10-3-1)17-16-12-6-8-13-7-4-5-11-15(13)16/h1-12,17H |
| Chemical Name | N-phenylnaphthalen-1-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion In male Sprague-Dawley rats administered a single oral dose of 160 mg [14C] N-phenyl-1-naphthylamine/kg bw, the chemical was well absorbed, metabolized almost completely, and excreted primarily in the feces. Radioactivity was detected in plasma within 60 min, with the maximum concentration measured after 4 hr. After 24 hr, 20% of the radioactivity was found in the GI tract (including contents), 2.4% in fatty tissue, 0.4% in the liver, and 0.1% in the kidneys. 90% of the administered radioactivity was excreted within 48 hr; 95% was excreted within 72 hr (60% in the feces and 35% in the urine). High performance liquid chromatographic and gas chromatographic methods for the determination of N-phenyl-1-naphthylamine (I) in biological fluid and tissues were developed, and the biological fate of I was studied in male rats following single and multiple oral administration. It was assumed that the intestinal absorption of I was excellent, because the fecal excretion of unchanged I was extremely low (0.4% of dose), and there was little unchanged I excreted in the urine (0.01% of dose). A large amount of glucuronide and sulfate metabolites was excreted in the urine. After single or multiple (6 days) oral administration, a small amount of unchanged I was distributed in fat, but the distribution of unchanged I in liver, kidney, spleen, heart and lung was extremely low. Metabolism / Metabolites Mono- and dihydroxy-derivatives of N-phenyl-1-naphthylamine have been identified in in vitro metabolic studies conducted with rat liver microsomes. /It was/ suggested that the hydroxyl group in the mono-hydroxy derivative is in the naphthalene moiety at a para-position to the amino group, whereas at least one hydroxyl group in the dihydroxy-derivative is at the available para-position in the naphthyl ring. Pretreatment of male rats with phenobarbital or 3-methylcholanthrene increased the rate of microsomal metabolism, indicating that more than one P-450 enzyme is involved in the metabolism of N-phenyl-1-naphthylamine. ...In the ether extract of the urine /of male Sprague-Dawley rats administered a single oral dose of 160 mg [14C] N-phenyl-1-naphthylamine/kg bw/, at least five radioactive metabolites were detected but not identified. In studies conducted with human volunteers or laboratory animals, the isomer N-phenyl-2-naphthylamine was partially metabolized to the known human carcinogen 2-naphthylamine following ingestion or inhalation. Although data concerning the formation of this metabolite are not available for N-phenyl-1-naphthylamine, it should be noted that, based on its chemical structure, it is unlikely that N-phenyl-1-naphthylamine is metabolized to 2-naphthylamine. /N-phenyl-2-naphthylamine/ Biological Half-Life In male Sprague-Dawley rats administered a single oral dose of 160 mg [14C] N-phenyl-1-naphthylamine/kg bw, ...the elimination half-lives were reported as 1.68 hr for the fast elimination and 33 hr for the slow elimination. |
| Toxicity/Toxicokinetics |
Non-Human Toxicity Values LD50 Rat oral 1625 mg/kg LD50 Mouse oral 1231 mg/kg |
| References |
[1]. Biochemical characterization and bacterial expression of an odorant-binding protein from Locusta migratoria. Cell Mol Life Sci. 2003 Feb;60(2):390-400. |
| Additional Infomation |
N-phenyl-1-naphthylamine appears as white to slightly yellowish prisms or reddish brown crystalline powder. (NTP, 1992) N-Phenyl-1-naphthylamine is a member of naphthalenes. N-Phenyl-1-naphthylamine has been reported in Streptomyces, Arundo donax, and Triadica sebifera with data available. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.5604 mL | 22.8019 mL | 45.6038 mL | |
| 5 mM | 0.9121 mL | 4.5604 mL | 9.1208 mL | |
| 10 mM | 0.4560 mL | 2.2802 mL | 4.5604 mL |