Physicochemical Properties
| Molecular Formula | C6H11NO3 |
| Molecular Weight | 145.15644 |
| Exact Mass | 145.074 |
| CAS # | 115819-92-6 |
| Related CAS # | N-Hydroxypipecolic acid potassium;2253632-01-6 |
| PubChem CID | 269025 |
| Appearance | White to off-white solid powder |
| LogP | 0.252 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 10 |
| Complexity | 137 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | SEWARTPIJFHCRP-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H11NO3/c8-6(9)5-3-1-2-4-7(5)10/h5,10H,1-4H2,(H,8,9) |
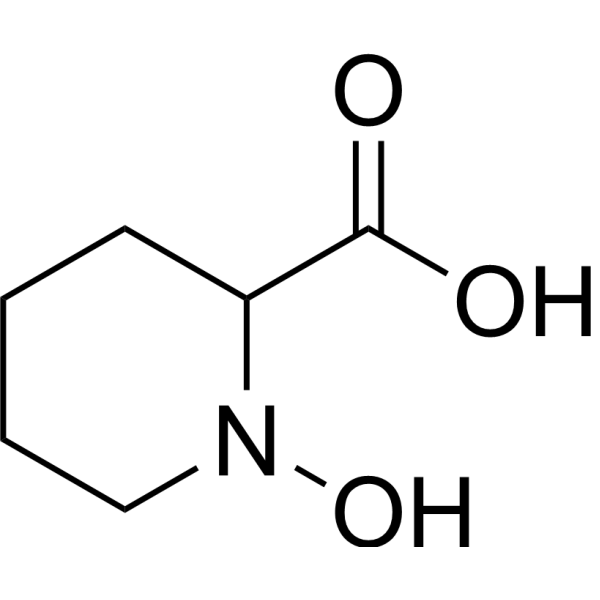
| Chemical Name | 1-hydroxypiperidine-2-carboxylic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | In order to ensure improved plant immunity, N-hydroxypicolic acid (NHP) works through a variety of mechanisms in SAR, including direct sensing of SAR gene expression, signal amplification, the start of heightened defensive activation, and positive responses to salicylic acid signals. In order to produce NHP, monooxygenase 1 (FMO1) operates downstream of Pip by foaming Pip[1][3]. |
| References |
[1]. Pipped at the Post: Pipecolic Acid Derivative Identified as SAR Regulator. Cell. 2018 Apr 5;173(2):286-287. [2]. Signals of Systemic Immunity in Plants: Progress and Open Questions. Int J Mol Sci. 2018 Apr 10;19(4). pii: E1146. [3]. N-hydroxypipecolic acid and salicylic acid: a metabolic duo for systemic acquired resistance. Curr Opin Plant Biol. 2019 Aug;50:44-57. |
| Additional Infomation | N-hydroxypipecolic acid is an N-hydroxy-alpha-amino-acid resulting from the formal N-hydroxylation of the amino group of piperidine-carboxylic acid (pipecolic acid). It is a N-hydroxy-alpha-amino-acid and a piperidinemonocarboxylic acid. It is functionally related to a pipecolic acid. |
Solubility Data
| Solubility (In Vitro) |
H2O : ~50 mg/mL (~344.45 mM) DMSO : ~25 mg/mL (~172.22 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (14.33 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (14.33 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (14.33 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 6.8890 mL | 34.4448 mL | 68.8895 mL | |
| 5 mM | 1.3778 mL | 6.8890 mL | 13.7779 mL | |
| 10 mM | 0.6889 mL | 3.4445 mL | 6.8890 mL |