Physicochemical Properties
| Molecular Formula | C21H31N3O5S |
| Molecular Weight | 437.5529 |
| Exact Mass | 437.198 |
| Elemental Analysis | C, 57.65; H, 7.14; N, 9.60; O, 18.28; S, 7.33 |
| CAS # | 59880-97-6 |
| PubChem CID | 443295 |
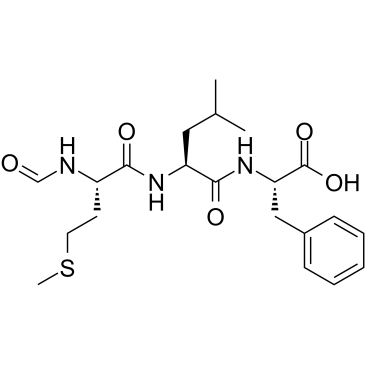
| Sequence | Formyl-Met-Leu-Phe; N-formyl-L-methionyl-L-leucyl-L-phenylalanine |
| SequenceShortening | MLF; Formyl-MLF |
| Appearance | White to off-white solid powder |
| Density | 1.2±0.1 g/cm3 |
| Boiling Point | 783.5±60.0 °C at 760 mmHg |
| Melting Point | 271-274 °C(lit.) |
| Flash Point | 427.6±32.9 °C |
| Vapour Pressure | 0.0±2.9 mmHg at 25°C |
| Index of Refraction | 1.551 |
| LogP | 1.5 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 13 |
| Heavy Atom Count | 30 |
| Complexity | 567 |
| Defined Atom Stereocenter Count | 3 |
| SMILES | CC(C)C[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)O)NC(=O)[C@H](CCSC)NC=O |
| InChi Key | PRQROPMIIGLWRP-BZSNNMDCSA-N |
| InChi Code | InChI=1S/C21H31N3O5S/c1-14(2)11-17(23-19(26)16(22-13-25)9-10-30-3)20(27)24-18(21(28)29)12-15-7-5-4-6-8-15/h4-8,13-14,16-18H,9-12H2,1-3H3,(H,22,25)(H,23,26)(H,24,27)(H,28,29)/t16-,17-,18-/m0/s1 |
| Chemical Name | (2S)-2-[[(2S)-2-[[(2S)-2-formamido-4-methylsulfanylbutanoyl]amino]-4-methylpentanoyl]amino]-3-phenylpropanoic acid |
| Synonyms | Chemotactic peptide; fMLF; 59880-97-6; fMetLeuPhe; N-Formyl-L-methionyl-L-leucyl-L-phenylalanine; CHEBI:53490; DTXSID9041077; RefChem:1091810; DTXCID00820624; N-Formyl-Met-Leu-Phe; FMLP |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
N-formyl peptide receptor 1 (FPR1) (EC₅₀ = 1.2 nM in calcium mobilization assay) |
| ln Vitro |
The surface cell receptor N-formyl peptide receptor (FPR) is bound by N-Formyl-Met-Leu-Phe, which sets off a distinct series of metabolic reactions that result in cellular activation. Under circumstances of osteoblast secretion, the G-coupled peptide receptor N-Formyl-Met-Leu-Phe, or FPR, stimulates osteoblast commitment and suppresses adipogenesis. N-Formyl-Met-Leu-Phe marks the creation of bones and promotes osteogenesis. The expression of peroxisome proliferator-activated receptor-γ1 is inhibited by N-formyl-met-leu-phen. The Ca2+-calmodulin inhibitor II-ERK-CREB signaling pathway combines with FPR1-phospholipase C/phospholipase D to drive the osteochondrogenesis induced by N-Formyl-Met-Leu-Phe [1]. N-Formyl-Met-Leu-Phe is a peptide produced from bacteria that causes human peripheral blood mononuclear cells to express proinflammatory cytokines. By means of signaling, the viral products LPS and N-Formyl-Met-Leu-Phe numerous signals work together to generate responses. TNF-α co-sensing via p65 nuclear translocation is the mechanism that TLR4, IKKβ-IκBα, and NF-κB signaling rely on [2].
1. Osteoblast Differentiation: - fMLP (10 nM–1 μM) significantly increased alkaline phosphatase (ALP) activity and mineralized nodule formation in human bone marrow mesenchymal stem cells (hBMSCs). This effect was abolished by FPR1 antagonist cyclosporine H, confirming FPR1 dependency. Western blot analysis revealed upregulation of osteogenic markers RUNX2 and Osterix, with concurrent activation of ERK1/2 and AKT signaling pathways 2. Inflammatory Synergy: - Co-treatment of RAW 264.7 macrophages with fMLP (100 nM) and lipopolysaccharide (LPS, 1 μg/mL) resulted in synergistic induction of TNF-α (2.8-fold increase) and IL-6 (3.5-fold increase) compared to LPS alone. This synergy was mediated by FPR1 and TLR4 cross-talk, as demonstrated by siRNA knockdown experiments 3. Calprotectin Release: - In human polymorphonuclear neutrophils (PMNs), fMLP (10 nM) induced calprotectin release in a dose-dependent manner (maximal 4.2-fold increase at 30 minutes). This effect was blocked by pertussis toxin, indicating G protein-coupled receptor (GPCR) dependency |
| ln Vivo |
N-Formyl-Met-Leu-Phe helps zebrafish and rabbits build stronger bones. More than 80% of zebrafish treated with N-formyl-Met-Leu-Phe showed signs of endpoint development at 5 dpf. Runx2 expression rose in response to N-Formyl-Met-Leu-Phe treatment. N-Formyl-Met-Leu-Phe-treated skulls have a large medullary cavity and dense connective tissue, such as periosteum, covering the bone [1]. Calprotectin release from PMN is mediated in vitro by N-Formyl-Met-Leu-Phe. It dose-dependently causes PMNs to release calprotectin. Roughly 10% of total PMN calprotectin is preserved at concentrations of N-Formyl-Met-Leu-Phe ranging from 0.1 to 10.0 nM [3].
1. Bone Regeneration Model: - Subcutaneous implantation of fMLP-loaded scaffolds in nude mice promoted ectopic bone formation, as confirmed by micro-CT and histological analysis. New bone volume/tissue volume (BV/TV) ratio increased by 45% compared to control scaffolds. Immunohistochemistry showed FPR1 expression in osteoblasts and endothelial cells within the regenerated tissue 2. Sepsis Model: - Intraperitoneal injection of fMLP (1 mg/kg) in LPS-challenged mice exacerbated systemic inflammation, characterized by increased plasma TNF-α (2.1-fold) and IL-6 (2.5-fold). Survival rate decreased from 60% to 30% compared to LPS alone. This effect was reversed by FPR1 knockout or cyclosporine H pretreatment |
| Enzyme Assay |
1. Calcium Mobilization Assay:
- CHO cells stably expressing FPR1 were loaded with Fluo-4 AM and stimulated with fMLP (0.1 nM–10 μM). Changes in intracellular calcium were measured by fluorescence microscopy. EC₅₀ value was calculated as 1.2 nM using sigmoidal dose-response curve fitting 2. GTPγS Binding Assay: - Membrane fractions from THP-1 cells were incubated with fMLP (0.01 nM–1 μM), GDP (10 μM), and [³⁵S]-GTPγS (0.1 nM). Bound radioactivity was measured by filtration. IC₅₀ value for FPR1-mediated G protein activation was 0.8 nM |
| Cell Assay |
1. Osteogenic Differentiation Assay:
- hBMSCs were cultured in osteogenic medium supplemented with fMLP (10 nM–1 μM). ALP activity was quantified using p-nitrophenyl phosphate substrate at day 7, and mineralization was assessed by alizarin red staining at day 21. FPR1 antagonist treatment abolished these effects 2. Neutrophil Chemotaxis: - Transwell migration assay showed fMLP (10 nM) induced dose-dependent PMN chemotaxis (maximal 3.2-fold increase). Migration was inhibited by anti-FPR1 antibody (70% reduction) and cytochalasin B, indicating actin-dependent motility |
| Animal Protocol |
1. Ectopic Bone Formation:
- fMLP (100 μg) was incorporated into poly(lactic-co-glycolic acid) (PLGA) scaffolds and implanted subcutaneously in 6-week-old nude mice. After 8 weeks, mice were euthanized, and scaffolds were analyzed by micro-CT and histology 2. Sepsis Model: - C57BL/6 mice received intraperitoneal LPS (5 mg/kg) followed by fMLP (1 mg/kg) 1 hour later. Survival was monitored for 72 hours. Plasma cytokines were measured by ELISA at 6 hours post-LPS |
| ADME/Pharmacokinetics |
- Absorption:
- Oral bioavailability in rats was 8.5%, with peak plasma concentration (Cₘₐₓ) of 15 ng/mL at 1 hour post-dose. Subcutaneous administration resulted in 92% bioavailability - Metabolism: - Primarily metabolized by plasma esterases to inactive fragments. Less than 2% of the dose was excreted unchanged in urine - Half-life: - Plasma t½ was 1.8 hours in mice, with rapid clearance from circulation due to receptor-mediated internalization |
| Toxicity/Toxicokinetics |
- Acute Toxicity:
- LD₅₀ in mice exceeded 2000 mg/kg (oral), with no significant organ damage observed in histopathological analysis - Immunotoxicity: - Repeated subcutaneous fMLP (0.5 mg/kg daily for 14 days) in rats caused neutrophilia (2.3-fold increase) and splenomegaly. These effects were reversible after treatment cessation |
| References |
[1]. N-formyl-methionyl-leucyl-phenylalanine (fMLP) promotes osteoblast differentiation via the N-formyl peptide receptor 1-mediated signaling pathway in human mesenchymal stem cells from bone marrow. J Biol Chem. 2011 May 13;286(19):17133-43. [2]. Synergistic induction of inflammation by bacterial products lipopolysaccharide and fMLP: an important microbial pathogenic mechanism. J Immunol. 2009 Feb 15;182(4):2518-24. [3]. Chemotaxins C5a and fMLP induce release of calprotectin (leucocyte L1 protein) from polymorphonuclear cells in vitro. Mol Pathol. 1998 Jun;51(3):143-8. |
| Additional Infomation |
- Background:
- fMLP is a bacterial-derived tripeptide that mimics host mitochondrial peptides. It acts as a pathogen-associated molecular pattern (PAMP) to activate innate immunity - Mechanism: - FPR1 activation by fMLP triggers Gαᵢ/o protein signaling, leading to calcium mobilization, MAPK activation, and NF-κB translocation. Synergy with TLR4 involves MyD88-dependent signaling cross-talk - Clinical Relevance: - FPR1-targeted therapies are being developed for osteoporosis and sepsis. However, fMLP-induced inflammation highlights the need for receptor-specific modulators N-formyl-L-methionyl-L-leucyl-L-phenylalanine is a tripeptide composed of L-Met, L-Leu and L-Phe in a linear sequence with a formyl group at the amino terminus. It acts as a potent inducer of leucocyte chemotaxis and macrophage activator as well as a ligand for the FPR receptor. It is functionally related to a N-formyl-L-methionine, a L-leucine and a L-phenylalanine. It is a conjugate acid of a N-formyl-L-methionyl-L-leucyl-L-phenylalaninate. A formylated tripeptide originally isolated from bacterial filtrates that is positively chemotactic to polymorphonuclear leucocytes, and causes them to release lysosomal enzymes and become metabolically activated. |
Solubility Data
| Solubility (In Vitro) |
DMSO : ≥ 82.5 mg/mL (~188.55 mM) H2O : < 0.1 mg/mL |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.75 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.75 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2855 mL | 11.4273 mL | 22.8545 mL | |
| 5 mM | 0.4571 mL | 2.2855 mL | 4.5709 mL | |
| 10 mM | 0.2285 mL | 1.1427 mL | 2.2855 mL |