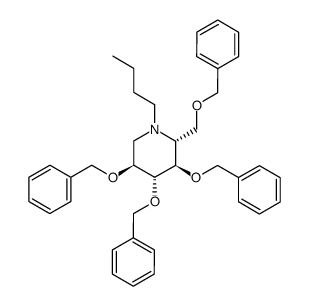
N-Boc-tetra-O-benzyl-1-deoxynojirimycin is a Boc and Benzyl protected form of 1-Deoxynojirimycin (also called duvoglustat or moranolin) which is an alpha-glucosidase inhibitor, most commonly found in mulberry leaves. Although it can be obtained in small quantities by brewing an herbal tea from mulberry leaves, interest in commercial production has led to research on developing mulberry tea higher in DNJ, and on alternate routes of production, such as via Bacillus species.
Physicochemical Properties
| Molecular Formula | C38H45NO4 |
| Molecular Weight | 579.76800 |
| Exact Mass | 579.335 |
| CAS # | 227932-82-3 |
| Related CAS # | 19130-96-2; 73285-50-4 |
| PubChem CID | 133556114 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 7.381 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 16 |
| Heavy Atom Count | 43 |
| Complexity | 711 |
| Defined Atom Stereocenter Count | 4 |
| SMILES | CCCCN1[C@H](COCC2=CC=CC=C2)[C@@H](OCC2=CC=CC=C2)[C@H](OCC2=CC=CC=C2)[C@@H](OCC2=CC=CC=C2)C1 |
| InChi Key | DZPAABGOOAWABR-NAQJMGRXSA-N |
| InChi Code | InChI=1S/C38H45NO4/c1-2-3-24-39-25-36(41-27-32-18-10-5-11-19-32)38(43-29-34-22-14-7-15-23-34)37(42-28-33-20-12-6-13-21-33)35(39)30-40-26-31-16-8-4-9-17-31/h4-23,35-38H,2-3,24-30H2,1H3/t35-,36+,37-,38-/m0/s1 |
| Chemical Name | (2S,3S,4S,5R)-1-butyl-3,4,5-tris(phenylmethoxy)-2-(phenylmethoxymethyl)piperidine |
| Synonyms | 227932-82-3; (2R,3R,4R,5S)-3,4,5-Tris(benzyloxy)-2-((benzyloxy)methyl)-1-butylpiperidine; (2S,3S,4S,5R)-1-butyl-3,4,5-tris(phenylmethoxy)-2-(phenylmethoxymethyl)piperidine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | α-glucosidase |
| ln Vitro |
α-Glucosidase inhibitory activities [2] For decades, researchers have shown that rat and human α-glucosidase is strongly inhibited by mulberry leaf extract (Anno et al., 2004, Miyahara et al., 2004, Oku et al., 2006). α-Glucosidase, located in the brush-border surface membranes of intestinal cells, is considered the most important enzyme in digestion of starch and other carbohydrates (Herscovics, 1999). Modification of carbohydrate metabolism by dietary foods and drugs may have therapeutic value. Mulberry 1-Deoxynojirimycin (Duvoglustat)/DNJ binds to the active center of α-glucosidase and is a potent inhibitor of this enzyme in the small intestine (Junge, Matzke, & Stoltefuss, 1996). For commercial development of nutraceutical products, the target compound and its concentration in the product should be known in order to achieve the best therapeutic results. In the case of mulberry dry tea, we think DNJ is the key compound because it strongly inhibits α-glucosidase and mulberry leaves contain high concentrations of it (50% of total imino sugars) (Asano et al., 2001). α-Glucosidase inhibition was highly correlated with both pure DNJ (r = 0.96) (Fig. 4B) and DNJ content of mulberry leaves (r = 0.84) (Fig. 4A). At comparable 1-Deoxynojirimycin (Duvoglustat)/DNJ concentrations, mulberry leaf extract had more α-glucosidase inhibitory activity than the DNJ standard: for example at 5 μg DNJ/ml, α-glucosidase activity was inhibited 27% by mulberry leaf extract and 23% by pure DNJ. The additional inhibition can be explained by presence in mulberry extract of other imino sugars (i.e., N-methyl-DNJ, 2-O-α-d-galactopyanosyl-DNJ and fagomine) and other ingredients such as isoquercitrin, quercetin and rutin. |
| ln Vivo |
1-Deoxynojirimycin (Duvoglustat) (20 -80 mg/kg; iv; once daily for 4 weeks) has consequences that are antiphysiological [3]. 1-Deoxynojirimycin enhances insulin symptoms noticeably by triggering the db/db shark shark pattern.
1-Deoxynojirimycin (Duvoglustat)/DNJ is widely used for the treatment of diabetes mellitus as an inhibitor of intestinal α-glucosidase. However, there are few reports about its effect on insulin sensitivity improvement. The aim of the present study was to investigate whether DNJ decreased hyperglycemia by improving insulin sensitivity. An economical method was established to prepare large amounts of DNJ. Then, db/db mice were treated with DNJ intravenously (20, 40 and 80 mg·kg(-1)·day(-1)) for four weeks. Blood glucose and biochemical analyses were conducted to evaluate the therapeutic effects on hyperglycemia and the related molecular mechanisms in skeletal muscle were explored. DNJ significantly reduced body weight, blood glucose and serum insulin levels. DNJ treatment also improved glucose tolerance and insulin tolerance. Moreover, although expressions of total protein kinase B (AKT), phosphatidylinositol 3 kinase (PI3K), insulin receptor beta (IR-β), insulin receptor substrate-1 (IRS1) and glucose transporter 4 (GLUT4) in skeletal muscle were not affected, GLUT4 translocation and phosphorylation of Ser473-AKT, p85-PI3K, Tyr1361-IR-β and Tyr612-IRS1 were significantly increased by DNJ treatment. These results indicate that DNJ significantly improved insulin sensitivity via activating insulin signaling PI3K/AKT pathway in skeletal muscle of db/db mice. [3] |
| Enzyme Assay |
α-Glucosidase inhibition assay [1] α-Glucosidase inhibitory activity was measured by a modification of the procedure described by Ma, Hattori, Daneshtalab, and Wang (2008). Briefly, rat-intestine acetone powder (1 g) was suspended in 100 mM potassium phosphate buffer (pH 7.0) and the suspension was sonicated for 20 min. After centrifugation at 3000 rpm for 30 min, the supernatant was used as the source of α-glucosidase. Substrate (2 mM 4-nitrophenyl-α-d-glucopyranoside) in 100 mM potassium phosphate buffer (pH 7.0) was pipetted into 96-well plates (40 μl/well). Five μl mulberry sample or control solution (a 50:50 mixture of ethanol and distilled water) was added and the solution was mixed. After addition of enzyme (5 μl), the mixture was incubated at 37 °C for 20 min, and then UV absorbance (405 nm) was measured. The percent α-glucosidase inhibitory activity of mulberry samples and standard 1-Deoxynojirimycin (Duvoglustat)/DNJ was calculated as: (ΔAcontrol-ΔAsample) × 100/ΔAcontrol, where ΔA is absorbance at 405 nm. |
| Cell Assay |
Western Blot [3] In order to investigate the effects of 1-Deoxynojirimycin (Duvoglustat)/DNJ on insulin signaling pathways, western blot analysis was performed as previously described. Briefly, skeletal muscle tissues (0.1 g) were lysed in lysis buffer (50 mM Tris (pH 7.4), 150 mM NaCl, 0.1% SDS, 0.5% sodium deoxycholate, 1% NP40, 10 μL phosphatase inhibitors, 1 μL protease inhibitor and 5 μL 100 mM PMSF), centrifuged for 15 min at 16,000× g at 4 °C, and protein concentration was quantified by bicinchonininc acid protein assay. Equal amounts of protein (70 μg) were loaded on 10% SDS-PAGE and transferred onto PVDF membranes. After membranes were blocked, they incubated with the primary antibodies against IR-β, p-Tyr1361-IR-β, IRS1, p-Tyr612-IRS1, PI3K, p-p85-PI3K, AKT, p-Ser473-AKT, GLUT4, β-actin or Na+K+-ATPase α1 overnight at 4 °C followed by HRP conjugated secondary antibody for 2 h at room temperature. Protein bands were visualized using an ECL detection kit. Normalization of total protein expression was carried out by using β-actin as control. Normalization of m-GLUT4 expression was carried out using Na+K+-ATPase α1 as control |
| Animal Protocol |
Animal/Disease Models: db/db mice[3] Doses: 20, 40, 80 mg/kg Route of Administration: intravenous (iv) (iv)injection; signal load PI3K/AKT[ 3]. one time/day for four weeks Experimental Results: Significant reduction in body weight, blood glucose, and serum insulin levels; improved glucose tolerance and insulin tolerance. At the end of ten weeks, wild-type C57BLKS mice, which received intravenously normal saline, served as a normal control (N control) (n = 6). The db/db mice were divided into four groups (n = 6): Group I served as a diabetic control and received intravenously normal saline (D control). Group II, III, and IV were treated intravenously with 1-Deoxynojirimycin (Duvoglustat)/DNJ 20, 40, and 80 mg·kg−1·day−1, respectively. An intravenous injection was selected to avoid the function of DNJ as an α-Glycosidase inhibitor inthe gastrointestinal tract. For DNJ doses selection, in our previous study, we screened a large number of Chinese traditional medicines including mulberry leaves by glucose tolerance test of ICR mice. We found the alkaloids (DNJ 40 mg·kg−1) isolated from mulberry leaves could improve the glucose tolerance test of ICR mice (Figure A1). We then tested doses of 10, 20, and 40 mg·kg−1, but both 10 and 20 mg·kg−1 did not have any effect (Figure A2). Therefore, we selected the 1-Deoxynojirimycin (Duvoglustat)/DNJ doses as 20, 40, and 80 mg·kg−1·day−1. All these doses were given for 4 weeks. The blood glucose, body weight and average food intake, water intake, and urine output were measured every week. At the end of the experimental period, the mice were anesthetized with chloral hydrate after withholding food for 12 h, and blood samples were taken to determine the serum insulin levels. Besides, skeletal muscle were removed after the blood was collected, then rinsed with a physiological saline solution, and immediately stored at −80 °C [3]. |
| References |
[1]. 1-Deoxynojirimycin: Occurrence, Extraction, Chemistry, Oral Pharmacokinetics, Biological Activities and In Silico Target Fishing. Molecules. 2016 Nov 23;21(11). pii: E1600. [2]. Development of high 1-deoxynojirimycin (DNJ) content mulberry tea and use of response surface methodology to optimize tea-making conditions for highest DNJ extraction. LWT - Food Science and Technology. Volume 45, Issue 2, March 2012, Pages 226-232. [3]. 1-Deoxynojirimycin Alleviates Insulin Resistance via Activation of Insulin Signaling PI3K/AKT Pathway in Skeletal Muscle of db/db Mice. Molecules. 2015 Dec 4;20(12):21700-14. |
| Additional Infomation |
Duvoglustat is an optically active form of 2-(hydroxymethyl)piperidine-3,4,5-triol having 2R,3R,4R,5S-configuration. It has a role as an EC 3.2.1.20 (alpha-glucosidase) inhibitor, an anti-HIV agent, an anti-obesity agent, a bacterial metabolite, a hypoglycemic agent, a hepatoprotective agent and a plant metabolite. It is a 2-(hydroxymethyl)piperidine-3,4,5-triol and a piperidine alkaloid. An alpha-glucosidase inhibitor with antiviral action. Derivatives of deoxynojirimycin may have anti-HIV activity. 1-Deoxynojirimycin has been reported in Parmotrema austrosinense, Parmotrema praesorediosum, and other organisms with data available. An alpha-glucosidase inhibitor with antiviral action. Derivatives of deoxynojirimycin may have anti-HIV activity. See also: Fagomine (annotation moved to). 1-Deoxynojirimycin (DNJ, C₆H13NO₄, 163.17 g/mol), an alkaloid azasugar or iminosugar, is a biologically active natural compound that exists in mulberry leaves and Commelina communis (dayflower) as well as from several bacterial strains such as Bacillus and Streptomyces species. Deoxynojirimycin possesses antihyperglycemic, anti-obesity, and antiviral features. Therefore, the aim of this detailed review article is to summarize the existing knowledge on occurrence, extraction, purification, determination, chemistry, and bioactivities of DNJ, so that researchers may use it to explore future perspectives of research on DNJ. Moreover, possible molecular targets of DNJ will also be investigated using suitable in silico approach.[1] Mulberry 1-deoxynojirimycin (DNJ), a potent α-glucosidase inhibitor, suppresses postprandial blood glucose, thereby possibly preventing diabetes mellitus. At present, mulberry dry teas are commercially supplied as functional foods in many countries, but these products may not provide an effective dose (6 mg DNJ/60 kg human wt) due to their low DNJ content (about 100 mg/100 g of dry wt). Therefore, development of tea with higher DNJ content is desirable. To do this, we investigated distribution of DNJ content and α-glucosidase inhibitory activity in 35 Thai mulberry varieties. DNJ content in young leaves varied among mulberry varieties from 30 to 170 mg/100 g of dry leaves. Varieties having highest DNJ content were Kam, Burirum 60 and Burirum 51. Leaf position affected DNJ content: shoots > young leaves > mature leaves. DNJ concentration and α-glucosidase inhibitory activity were highly correlated (r = 0.84), suggesting that α-glucosidase inhibitory activity of mulberry leaves is mainly due to DNJ. Consequently, high DNJ content mulberry tea was produced from shoots of varieties such as Burirum 60, which contains 300 mg/100 g of dry wt. Tea-making conditions were optimized for highest DNJ extraction using response surface methodology. Approximate 95% of total DNJ in high DNJ content dry tea was extracted when temperature was maintained at 98 °C for 400 s; these conditions could be applicable for preparation of commercial products with high DNJ content. One cup (230 ml, a normal serving) of DNJ-enriched mulberry tea contained enough DNJ (6.5 mg) to effectively suppress postprandial blood glucose.[2] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.7248 mL | 8.6241 mL | 17.2482 mL | |
| 5 mM | 0.3450 mL | 1.7248 mL | 3.4496 mL | |
| 10 mM | 0.1725 mL | 0.8624 mL | 1.7248 mL |