Physicochemical Properties
| Molecular Formula | C11H23N3O5S |
| Molecular Weight | 309.382421731949 |
| Exact Mass | 309.135 |
| Elemental Analysis | C, 42.70; H, 7.49; N, 13.58; O, 25.86; S, 10.36 |
| CAS # | 89344-48-9 |
| Related CAS # | 616-91-1 (Ac-cysteine);56-87-1 (lysine);89344-48-9 |
| PubChem CID | 9883132 |
| Appearance | Typically exists as solid at room temperature |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 8 |
| Heavy Atom Count | 20 |
| Complexity | 254 |
| Defined Atom Stereocenter Count | 2 |
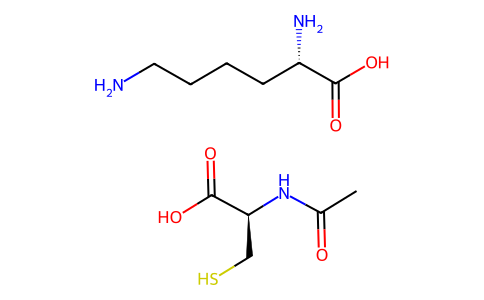
| SMILES | CC(=O)N[C@@H](CS)C(=O)O.C(CCN)C[C@@H](C(=O)O)N |
| InChi Key | YLCSLYZPLGQZJS-VDQHJUMDSA-N |
| InChi Code | InChI=1S/C6H14N2O2.C5H9NO3S/c7-4-2-1-3-5(8)6(9)10;1-3(7)6-4(2-10)5(8)9/h5H,1-4,7-8H2,(H,9,10);4,10H,2H2,1H3,(H,6,7)(H,8,9)/t5-;4-/m00/s1 |
| Chemical Name | (2R)-2-acetamido-3-sulfanylpropanoic acid;(2S)-2,6-diaminohexanoic acid |
| Synonyms | Nacystelyn; N-Acetylcysteine lysinate; 89344-48-9; UNII-8G1KV0RT1P; 8G1KV0RT1P; Lysine salt of N-acetylcysteine; n-acetylcysteinate lysine; ACETYLCYSTEINE LYSINE; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | N-Acetylcysteine derivative |
| ln Vitro |
Nacystelyn efficiently inhibited the procoagulant activity of human recombinant tissue factor, primary duct cells and human islet cell preparations at clinically relevant concentrations without cellular toxicity.
Conclusions/interpretation: Nacystelyn is a pharmaceutical candidate to reduce early beta cell loss related to tissue factor-dependent coagulation after islet transplantation.[1] We observed that Nacystelyn dose-dependently inhibited CD40 expression on CAPAN-2 cells as well as CD40-induced nuclear factor kappaB activation and proinflammatory cytokines up-regulation.[3] |
| ln Vivo | A newly synthesized mucolytic agent, N-acetylcysteine L-lysinate (Nacystelyn) was studied. Tracheal mucus velocity (TMV), transepithelial potential difference (PD), rheological properties, and ion content of collected airway secretions were evaluated in six healthy mongrel dogs after placebo, Nacystelyn (NAL) and acetylcysteine (NAC) metered dose inhaler (MDI) aerosols. Although TMV was increased and viscoelasticity decreased after both treatments, the treatment effect with NAL was significantly greater. Furthermore, NAL increased the negative PD and CI- content of secretions in the trachea, an effect not observed after NAC. Both compounds increased ciliary beat frequency (CBF) on the frog palate at a concentration range similar to that approximated in dog airways. The increased mucociliary clearance could be partially explained by favourable rheological changes combined with stimulation of CBF. Since both compounds break disulfide bonds in mucus polymers, the greater change in mucus rheology and clearance rate after NAL, without change in water content, could be explained by the increase in CI- content. Nacystelyn appears to combine different modes of action which synergistically cause an increase in the clearance rate of airway secretions.[2] |
| Cell Assay |
The effects of Nacystelyn, a salt derivative of N-acetyl-L-cysteine, were first assessed on procoagulant activity induced in human plasma by recombinant tissue factor, human primary duct cells or human islet cell preparations. The influence of Nacystelyn on clot formation, platelet counts and D-dimers were measured in a whole blood tubing loop model. Human beta cell viability and insulin synthesis after Nacystelyn treatment were assessed to exclude cytotoxicity of Nacystelyn.[1] Conclusions: Our data suggest that Nacystelyn could be considered as a useful tool to prevent immune and inflammatory responses in pancreatic disorders by interfering with the CD40 pathway in pancreatic duct cells. We assessed the effects of Nacystelyn on CD40 expression and function in human caucasian pancreatic adenocarcinoma, ATCC n degrees THB-80 (CAPAN-2) cells, a human pancreatic duct cell line. CD40 expression was analyzed by flow cytometry. To assess CAPAN-2 cell responses to CD40 engagement, we looked at nuclear factor-kappaB transcription factor activation using enzyme-linked immunosorbent assay and electrophoretic mobility shift assay and cytokine mRNA levels by quantitative real-time reverse transcriptase polymerase chain reaction.[3] |
| References |
[1]. N-Acetylcysteine derivative inhibits procoagulant activity of human islet cells. Diabetologia. 2007 Feb;50(2):343-7. [2]. A comparison of a new mucolytic N-acetylcysteine L-lysinate with N-acetylcysteine: airway epithelial function and mucus changes in dog. Pulm Pharmacol. 1995 Dec;8(6):259-65. [3]. N-acetylcysteine derivative inhibits CD40-dependent proinflammatory properties of human pancreatic duct cells. Pancreas. 2008 May;36(4):363-8. |
| Additional Infomation |
The early loss of beta cells after islet cell transplantation has been attributed in part to blood coagulation at the implant site. Tissue factor expressed by beta cells and contaminating duct cells is considered to activate this process. Here, we investigated the ability of N-acetyl-L-cysteine to suppress the in vitro procoagulant activity of duct cells and human islet cell preparations.[1] Objectives: We recently observed that duct cells constitutively express CD40, a membrane molecule whose engagement results in duct cell activation and proinflammatory cytokine secretion. This observation suggests a potential role of this pathway in the pathogenesis of type 1 diabetes, islet graft rejection, or acute pancreatitis. In this article, we investigated whether a salt derivative of N-acetyl-L-cysteine, Nacystelyn, could modulate CD40 expression on duct cells and the response of activated duct cells to CD40 engagement.[3] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.2323 mL | 16.1614 mL | 32.3227 mL | |
| 5 mM | 0.6465 mL | 3.2323 mL | 6.4645 mL | |
| 10 mM | 0.3232 mL | 1.6161 mL | 3.2323 mL |