Physicochemical Properties
| Molecular Formula | C20H13F6N3O2S |
| Molecular Weight | 473.3934 |
| Exact Mass | 473.063 |
| CAS # | 887148-69-8 |
| Related CAS # | (Rac)-Monepantel-d5;2747918-33-6;(Rac)-Monepantel sulfone-d5;2747918-68-7;(R)-Monepantel |
| PubChem CID | 44449087 |
| Appearance | White to off-white solid powder |
| LogP | 5.67 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 32 |
| Complexity | 763 |
| Defined Atom Stereocenter Count | 1 |
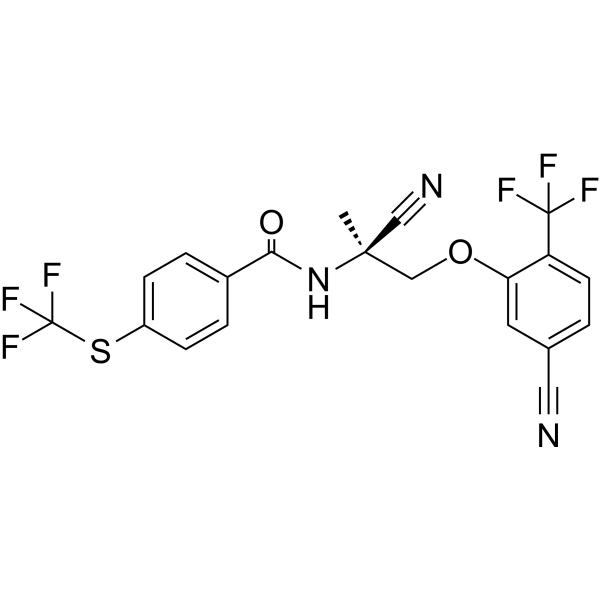
| SMILES | C[C@@](COC1=C(C=CC(=C1)C#N)C(F)(F)F)(C#N)NC(=O)C2=CC=C(C=C2)SC(F)(F)F |
| InChi Key | WTERNLDOAPYGJD-SFHVURJKSA-N |
| InChi Code | InChI=1S/C20H13F6N3O2S/c1-18(10-28,11-31-16-8-12(9-27)2-7-15(16)19(21,22)23)29-17(30)13-3-5-14(6-4-13)32-20(24,25)26/h2-8H,11H2,1H3,(H,29,30)/t18-/m0/s1 |
| Chemical Name | N-[(2S)-2-cyano-1-[5-cyano-2-(trifluoromethyl)phenoxy]propan-2-yl]-4-(trifluoromethylsulfanyl)benzamide |
| Synonyms | AAD-1566; AAD1566; AAD 1566 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | The presence of monepantel (25 μM) causes acidic vacuoles to accumulate. Liver cancer lines are very sensitive to Monepantel, with IC50 values of 7.2±0.2 μM (OVCAR-3) and 10.5±0.4 μM (A2780). Monepantel (0, 10 and 25 μM) stimulates autophagosome production in these Manhattan lines. Monepantel (0, 10, and 25 μM) revealed significantly lower levels of punctate staining, indicating suppression of Ser2448 phosphorylated mTOR. Monepantel also reduces the phosphorylated expression of Raptor at Ser792, a member of the mTORC1 coMonepantelex [2]. Monepantel (250 μg/mL) resulted in higher acetone expression of multiple ABC transporter genes in both worm isolates. Larvae exposed to 250 μg/mL Monepantel showed increased efflux of rhodamine-123, and some larval populations showed the ability to tolerate higher concentrations of IVM in migration assays [3]. |
| ln Vivo | In sheep, monepantel (10 μM) dramatically raises CYP3A24 mRNA and all CYP-related activity [4]. |
| References |
[1]. Assessment of the nematocidal activity of metallocenyl analogues of monepantel. Dalton Trans. 2016 Nov 28;45(44):17662-17671. [2]. Monepantel induces autophagy in human ovarian cancer cells through disruption of the mTOR/p70S6K signalling pathway. Am J Cancer Res. 2014 Sep 6;4(5):558-71. [3]. Increased expression of ATP binding cassette transporter genes following exposure of Haemonchus contortus larvae to a high concentration of monepantel in vitro. Parasit Vectors. 2016 Sep 29;9(1):522. [4]. Monepantel induces hepatic cytochromes p450 in sheep in vitro and in vivo. Chem Biol Interact. 2015 Feb 5;227:63-8. ?. |
| Additional Infomation |
Monepantel is a secondary carboxamide resulting from the formal condensation of the carboxy group of p-[(trifluoromethyl)sulfanyl]benzoic acid with the amino group of (2S)-2-amino-3-hydroxy-2-methylpropanenitrile in which the hydroxy group has been converted to the corresponding 5-cyano-2-(trifluoromethyl)phenyl ether. A broad-spectrum nematicide, it is used to control gastrointestinal roundworms in sheep and goats. It has a role as an anthelminthic drug and a nematicide. It is a nitrile, an aryl sulfide, an aromatic ether, a member of (trifluoromethyl)benzenes and a secondary carboxamide. Drug Indication Zolvix oral solution is a broad spectrum anthelmintic for the treatment and control of gastro-intestinal nematode infections and associated diseases in sheep including lambs, hoggets, breeding rams and ewes. , , Spectrum of activity includes fourth larvae and adults of: , , , Haemonchus contortus*; , Teladorsagia circumcincta*; , Teladorsagia trifurcata*; , Teladorsagia davtiani*; , Trichostrongylus axei*; , Trichostrongylus colubriformis; , Trichostrongylus vitrinus; , Cooperia curticei; , Cooperia oncophora; , Nematodirus battus; , Nematodirus filicollis; , Nematodirus spathiger; , Chabertia ovina; , Oesophagostomum venulosum. , , , * including inhibited larvae. , , The veterinary medicinal product is effective against strains of these parasites resistant to (pro)benzimidazoles, levamisole, morantel, macrocyclic lactones and H. contortus strains resistant to salicylanilides. , |
Solubility Data
| Solubility (In Vitro) | DMSO : ≥ 100 mg/mL (~211.24 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: 2.5 mg/mL (5.28 mM) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), suspension solution; with sonication. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (5.28 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1124 mL | 10.5621 mL | 21.1242 mL | |
| 5 mM | 0.4225 mL | 2.1124 mL | 4.2248 mL | |
| 10 mM | 0.2112 mL | 1.0562 mL | 2.1124 mL |