Physicochemical Properties
| Molecular Formula | C22H28O4CL2 |
| Molecular Weight | 427.36132 |
| Exact Mass | 520.142 |
| CAS # | 105102-22-5 |
| Related CAS # | Mometasone-d5 |
| PubChem CID | 441335 |
| Appearance | White to off-white solid powder |
| Density | 1.35g/cm3 |
| Boiling Point | 586.6ºC at 760mmHg |
| Melting Point | 220ºC (decomposes) |
| Flash Point | 308.5ºC |
| Vapour Pressure | 4.47E-18mmHg at 25°C |
| Index of Refraction | 1.603 |
| LogP | 4.869 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 28 |
| Complexity | 806 |
| Defined Atom Stereocenter Count | 8 |
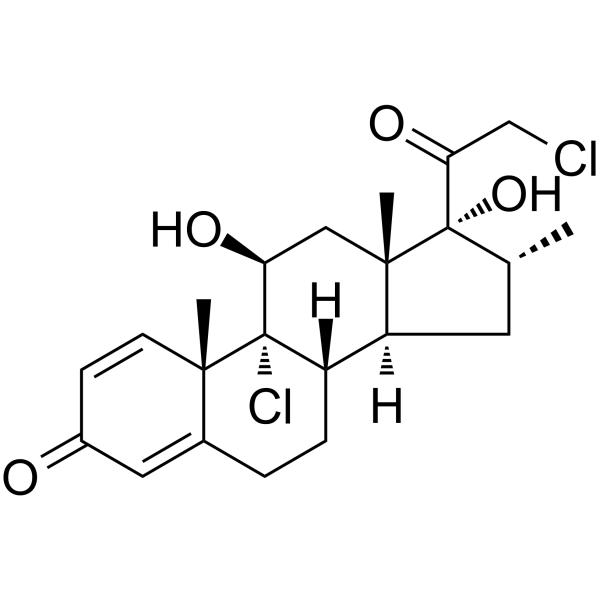
| SMILES | C[C@@H]1C[C@H]2[C@@H]3CCC4=CC(=O)C=C[C@@]4([C@]3([C@H](C[C@@]2([C@]1(C(=O)CCl)O)C)O)Cl)C |
| InChi Key | QLIIKPVHVRXHRI-CXSFZGCWSA-N |
| InChi Code | InChI=1S/C22H28Cl2O4/c1-12-8-16-15-5-4-13-9-14(25)6-7-19(13,2)21(15,24)17(26)10-20(16,3)22(12,28)18(27)11-23/h6-7,9,12,15-17,26,28H,4-5,8,10-11H2,1-3H3/t12-,15+,16+,17+,19+,20+,21+,22+/m1/s1 |
| Chemical Name | (8S,9R,10S,11S,13S,14S,16R,17R)-9-chloro-17-(2-chloroacetyl)-11,17-dihydroxy-10,13,16-trimethyl-6,7,8,11,12,14,15,16-octahydrocyclopenta[a]phenanthren-3-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vivo | Mometasone (0.1-3 mg/kg; intranasal) reduces the enhanced airway sensitivity to aerosolized methacholine and suppresses mild allergic lung inflammation in a dosage-dependent manner at the highest dose tested, 3 mg/kg. Allergen-induced rise in Penh in murine mice [3]. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Nasal spray is virtually undetectable in plasma Metabolism / Metabolites Hepatic. Extensive metabolism to multiple metabolites. There are no major metabolites detectable in plasma. Upon in vitro incubation, one of the minor metabolites formed is 6ß-hydroxy-mometasone furoate. In human liver microsomes, the formation of the metabolite is regulated by cytochrome P-450 3A4. Biological Half-Life 5.8 hours |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Neither topical mometasone nor the mometasone nasal implant have been studied during breastfeeding. Since only extensive application of the most potent corticosteroids may cause systemic effects in the mother, it is unlikely that short-term application of topical corticosteroids or the slow-release implant would pose a risk to the breastfed infant by passage into breastmilk. However, it would be prudent to use the least potent drug on the smallest area of skin possible. It is particularly important to ensure that the infant's skin does not come into direct contact with the areas of skin that have been treated. Current guidelines allow topical corticosteroids to be applied to the nipples just after nursing for eczema, with the nipples cleaned gently before nursing. Only water-miscible cream or gel products should be applied to the breast because ointments may expose the infant to high levels of mineral paraffins via licking. ◉ Effects in Breastfed Infants Topical application of a corticosteroid with relatively high mineralocorticoid activity (isofluprednone acetate) to the mother's nipples resulted in prolonged QT interval, cushingoid appearance, severe hypertension, decreased growth and electrolyte abnormalities in her 2-month-old breastfed infant. The mother had used the cream since birth for painful nipples. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. ◉ Summary of Use during Lactation Neither inhaled mometasone nor mometasone nasal implants have been studied during breastfeeding. Although not measured, the amounts of inhaled and nasal corticosteroids absorbed into the maternal bloodstream and excreted into breastmilk are probably too small to affect a breastfed infant. Expert opinion considers inhaled, nasal and oral corticosteroids acceptable to use during breastfeeding. See also Mometasone, Topical. ◉ Effects in Breastfed Infants None reported with any corticosteroid. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. Protein Binding 98% to 99% (in a concentration range of 5 to 500 ng/mL). |
| References |
[1]. Long-term, intermittent treatment of chronic hand eczema with mometasone furoate. Br J Dermatol. 1999 May;140(5):882-6. [2]. Mometasone or Tiotropium in Mild Asthma with a Low Sputum Eosinophil Level. N Engl J Med. 2019 May 23;380(21):2009-2019. [3]. Synergistic effect of formoterol and mometasone in a mouse model of allergic lung inflammation. Br J Pharmacol. 2007 Sep;152(1):83-90. |
| Additional Infomation |
Mometasone is an 11beta-hydroxy steroid, a 17alpha-hydroxy steroid, a 20-oxo steroid, a 3-oxo-Delta(1),Delta(4)-steroid, a chlorinated steroid and a tertiary alpha-hydroxy ketone. It has a role as an anti-inflammatory drug, a dermatologic drug, a vasoconstrictor agent and an anti-allergic agent. It is functionally related to a Delta(1)-progesterone. Mometasone is a corticosteroid not currently used in medical products. [Mometasone furoate] however, is still in use. Mometasone is a Corticosteroid. The mechanism of action of mometasone is as a Corticosteroid Hormone Receptor Agonist, and Corticosteroid Hormone Receptor Agonist. Mometasone is a synthetic topical glucocorticoid receptor (GR) agonist with anti-inflammatory, anti-pruritic and vasoconstrictive properties. Upon administration, mometasone binds to cytoplasmic GRs and subsequently activates GR-mediated gene expression. This results in the synthesis of certain anti-inflammatory proteins, while inhibiting the synthesis of certain inflammatory mediators. Specifically, mometasone appears to induce phospholipase A2 inhibitory proteins, thereby controlling the release of the inflammatory precursor arachidonic acid from phospholipid membrane by phospholipase A2. A pregnadienediol derivative ANTI-ALLERGIC AGENT and ANTI-INFLAMMATORY AGENT that is used in the management of ASTHMA and ALLERGIC RHINITIS. It is also used as a topical treatment for skin disorders. See also: Mometasone Furoate (has salt form); Mometasone Furoate Monohydrate (active moiety of). Drug Indication The inhaler is indicated for the maintenance treatment of asthma as prophylactic therapy. The nasal spray is indicated for the treatment of the nasal symptoms of seasonal allergic and perennial allergic rhinitis. Mechanism of Action Unbound corticosteroids cross cell membranes and bind with high affinity to specific cytoplasmic receptors. Inflammation is decreased by diminishing the release of leukocytic acid hydrolases, prevention of macrophage accumulation at inflamed sites, interference with leukocyte adhesion to the capillary wall, reduction of capillary membrane permeability, reduction of complement components, inhibition of histamine and kinin release, and interference with the formation of scar tissue. The antiinflammatory actions of corticosteroids are thought to involve phospholipase A2 inhibitory proteins, lipocortins, which control the biosynthesis of potent mediators of inflammation such as prostaglandins and leukotrienes. Mometasone furoate has been shown in vitro to exhibit a binding affinity for the human glucocorticoid receptor which is approximately 12 times that of dexamethasone, 7 times that of triamcinolone acetonide, 5 times that of budesonide, and 1.5 times that of fluticasone. Pharmacodynamics Mometasone is a medium-potency synthetic corticosteroid with antiinflammatory, antipruritic, and vasoconstrictive properties. Studies in asthmatic patients have demonstrated that mometasone provides a favorable ratio of topical to systemic activity due to its primary local effect along with the extensive hepatic metabolism and the lack of active metabolites. Though effective for the treatment of asthma, glucocorticoids do not affect asthma symptoms immediately. Maximum improvement in symptoms following inhaled administration of mometasone furoate may not be achieved for 1 to 2 weeks or longer after starting treatment. When glucocorticoids are discontinued, asthma stability may persist for several days or longer. Mometasone has been shown in vitro to exhibit a binding affinity for the human glucocorticoid receptor which is approximately 12 times that of dexamethasone, 7 times that of triamcinolone acetonide, 5 times that of budesonide, and 1.5 times that of fluticasone. The clinical significance of these findings is unknown. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~125 mg/mL (~292.49 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (4.87 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (4.87 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (4.87 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.3399 mL | 11.6997 mL | 23.3995 mL | |
| 5 mM | 0.4680 mL | 2.3399 mL | 4.6799 mL | |
| 10 mM | 0.2340 mL | 1.1700 mL | 2.3399 mL |