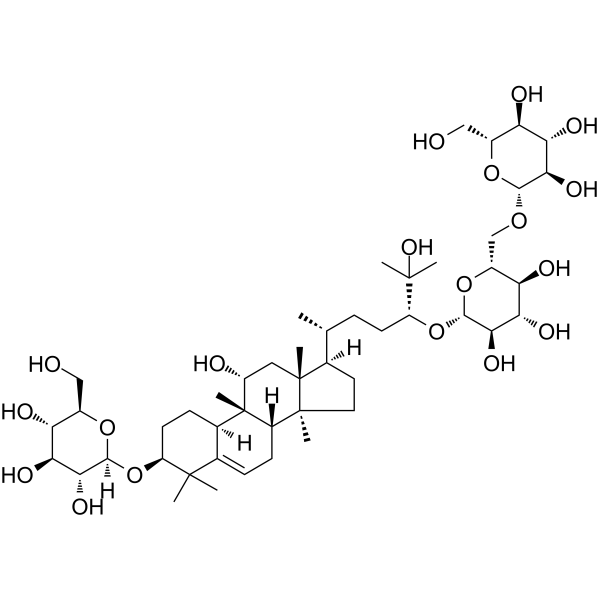
Physicochemical Properties
| Molecular Formula | C48H82O19 |
| Molecular Weight | 963.15328 |
| Exact Mass | 962.545 |
| CAS # | 130567-83-8 |
| PubChem CID | 24720988 |
| Appearance | White to off-white solid powder |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 1048.3±65.0 °C at 760 mmHg |
| Flash Point | 587.8±34.3 °C |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.619 |
| LogP | 1.87 |
| Hydrogen Bond Donor Count | 13 |
| Hydrogen Bond Acceptor Count | 19 |
| Rotatable Bond Count | 14 |
| Heavy Atom Count | 67 |
| Complexity | 1720 |
| Defined Atom Stereocenter Count | 25 |
| SMILES | C[C@H](CC[C@H](C(C)(C)O)O[C@H]1[C@@H]([C@H]([C@@H]([C@H](O1)CO[C@H]2[C@@H]([C@H]([C@@H]([C@H](O2)CO)O)O)O)O)O)O)[C@H]3CC[C@@]4([C@@]3(C[C@H]([C@@]5([C@H]4CC=C6[C@H]5CC[C@@H](C6(C)C)O[C@H]7[C@@H]([C@H]([C@@H]([C@H](O7)CO)O)O)O)C)O)C)C |
| InChi Key | KYVIPFHNYCKOMQ-YMRJDYICSA-N |
| InChi Code | InChI=1S/C48H82O19/c1-21(9-13-31(45(4,5)61)67-43-40(60)37(57)34(54)27(65-43)20-62-41-38(58)35(55)32(52)25(18-49)63-41)22-15-16-46(6)28-12-10-23-24(48(28,8)29(51)17-47(22,46)7)11-14-30(44(23,2)3)66-42-39(59)36(56)33(53)26(19-50)64-42/h10,21-22,24-43,49-61H,9,11-20H2,1-8H3/t21-,22-,24-,25-,26-,27-,28+,29-,30+,31-,32-,33-,34-,35+,36+,37+,38-,39-,40-,41-,42+,43+,46+,47-,48+/m1/s1 |
| Chemical Name | (2R,3S,4S,5R,6R)-2-(hydroxymethyl)-6-[[(2R,3S,4S,5R,6S)-3,4,5-trihydroxy-6-[(3R,6R)-2-hydroxy-6-[(3S,8S,9R,10R,11R,13R,14S,17R)-11-hydroxy-4,4,9,13,14-pentamethyl-3-[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl]oxy-2,3,7,8,10,11,12,15,16,17-decahydro-1H-cyclopenta[a]phenanthren-17-yl]-2-methylheptan-3-yl]oxyoxan-2-yl]methoxy]oxane-3,4,5-triol |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Enzymes involved in its biosynthesis pathway, as identified through functional expression and protein modeling: Squalene epoxidases (SQE), Cucurbitadienol synthase (CDS), Epoxide hydrolase (EPH3), Cytochrome P450 monooxygenase (CYP87D18), and UDP-glucosyltransferases (UGT720-269-1, UGT94 family). Docking studies indicated calculated binding affinities of -12.3 kcal/mol for cucurbitadienol and -12.5 kcal/mol for 24,25-epoxycucurbitadienol to CDS. [1] |
| ln Vitro |
A large-scale functional expression survey of nearly 200 candidate genes was conducted to identify the enzymes responsible for mogroside V biosynthesis. This involved heterologous expression in yeast (Saccharomyces cerevisiae) and bacteria (Escherichia coli) systems to demonstrate the catalytic activity of SQE, CDS, EPH, CYP450s, and UGTs in converting substrates along the mogroside pathway. [1] Metabolite analysis of developing Siraitia grosvenorii fruit showed that mogroside V (M5) is the major sweet component in ripe fruit (103 days after anthesis, DAA), accumulating at the expense of earlier glycosylated forms (M4 compounds) during the final stages of ripening. [1] |
| Enzyme Assay |
For squalene epoxidase (SQE) and cucurbitadienol synthase (CDS) activity, genes were cloned into yeast expression vectors (e.g., pESC-URA) and transformed into the yeast strain GIL77 (erg7 mutant). Transformed yeast was cultured, induced with galactose, and metabolites were extracted using organic solvents (e.g., n-hexane). The extracts were analyzed by liquid chromatography-mass spectrometry (LC-MS) to identify products like cucurbitadienol and 24,25-epoxycucurbitadienol. [1] For cytochrome P450 (CYP) hydroxylase activity, candidate CYP genes were co-expressed with CDS and a NADPH-CYP reductase in yeast strain BY4743_YHR072. In some experiments, the lanosterol synthase inhibitor RO 48-8071 was added to shunt flux toward the mogroside pathway. Cells were lysed, extracted with ethyl acetate, and analyzed by LC-MS to detect hydroxylated intermediates like 11-hydroxycucurbitadienol and the final aglycone mogrol. [1] For UDP-glucosyltransferase (UGT) activity, UGT genes were expressed in E. coli as His-tagged proteins. Crude enzyme extracts were incubated with mogrol or various mogroside substrates (dissolved in DMSO) and UDP-glucose in a Tris-HCl buffer. After overnight incubation, reactions were stopped with methanol, centrifuged, and products were analyzed by LC-MS to identify glucosylated derivatives. [1] |
| References |
[1]. The biosynthetic pathway of the nonsugar, high-intensity sweetener mogroside V from Siraitia grosvenorii.Proc Natl Acad Sci U S A. 2016 Nov 22;113(47):E7619-E7628. |
| Additional Infomation |
Mogroside IIIA is a beta-D-glucoside and a mogroside. It has a role as a plant metabolite. It is functionally related to a Mogroside II-E. Mogroside III has been reported in Siraitia grosvenorii with data available. Mogroside V is a triterpenoid glycoside natural sweetener isolated from the ripe fruit of Siraitia grosvenorii (monk fruit or luo-han-guo). It has a sweetening strength approximately 250 times that of sucrose and is non-caloric. [1] The biosynthetic pathway involves five enzyme families: 1) Squalene epoxidase (SQE) epoxidizes squalene to 2,3;22,23-diepoxysqualene; 2) Cucurbitadienol synthase (CDS) cyclizes diepoxysqualene to 24,25-epoxycucurbitadienol; 3) Epoxide hydrolase (EPH3) hydrolyzes the epoxide at C24,C25 to form 24,25-dihydroxycucurbitadienol; 4) Cytochrome P450 (CYP87D18) hydroxylates at C11; and 5) UDP-glucosyltransferases (primarily UGT720-269-1) sequentially glucosylate the C24 and C3 positions of mogrol to form di-glucosylated M2, which is further branched by UGT94 family enzymes to produce penta-glucosylated mogroside V. [1] The study found that the genes encoding this pathway are not physically clustered in the genome but exhibit a highly coordinated transcriptional pattern specifically in young Siraitia fruit, which is unique compared to other cucurbit species. This coordinated expression is key to the evolution of this sweetener pathway. [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~103.83 mM) H2O : ~100 mg/mL (~103.83 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (2.60 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (2.60 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (2.60 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.0383 mL | 5.1913 mL | 10.3826 mL | |
| 5 mM | 0.2077 mL | 1.0383 mL | 2.0765 mL | |
| 10 mM | 0.1038 mL | 0.5191 mL | 1.0383 mL |