Physicochemical Properties
| Molecular Formula | C6H18N2O4 |
| Molecular Weight | 182.2181 |
| Exact Mass | 182.126 |
| Elemental Analysis | C, 39.55; H, 9.96; N, 15.37; O, 35.12 |
| CAS # | 86426-17-7 |
| Related CAS # | Meldonium;76144-81-5; 86426-17-7 (dihydrate); 839675-65-9 (fumarate); 76144-81-5 (free); 839675-63-7 (phoshate); 1608503-17-8 (hydrate); |
| PubChem CID | 6918082 |
| Appearance | White to off-white solid powder |
| Hydrogen Bond Donor Count | 3 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 12 |
| Complexity | 112 |
| Defined Atom Stereocenter Count | 0 |
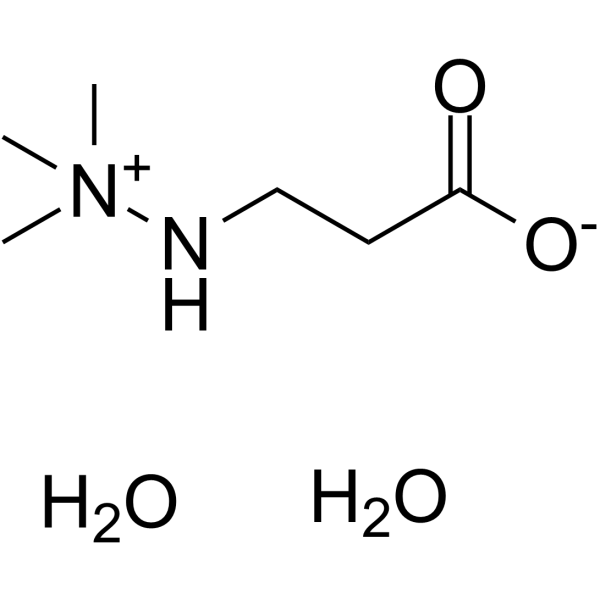
| SMILES | C[N+](C)(C)NCCC(=O)[O-].O.O |
| InChi Key | JFWLFLLRLZSBRA-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H14N2O2.2H2O/c1-8(2,3)7-5-4-6(9)10;;/h7H,4-5H2,1-3H3;2*1H2 |
| Chemical Name | 3-[(trimethylazaniumyl)amino]propanoate;dihydrate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- Carnitine palmitoyltransferase I (CPT I)
- Mildronate (Meldonium) inhibits CPT I, the rate-limiting enzyme of fatty acid β-oxidation, with an IC50 of 3.8 mM for rat liver CPT I and 2.1 mM for human recombinant CPT I [1] - Platelet-type phosphofructokinase (PFK-P) - Mildronate modulates PFK-P activity (a key enzyme in glycolysis) by increasing its phosphorylation (activation) at Ser32, with no direct inhibitory/activatory IC50/Ki reported [3] |
| ln Vitro |
- Cardiometabolic regulation (myocardial cells): 1. Fatty acid oxidation (FAO) inhibition: In neonatal rat ventricular myocytes (NRVMs) treated with Mildronate (1–10 mM) for 24 hours, FAO rates were reduced by 30–50% (measured by [14C]-palmitate oxidation assay). Concurrently, glucose oxidation (measured by [14C]-glucose) increased by 40–60%, shifting energy metabolism toward glucose utilization [1] 2. Mitochondrial function protection: Mildronate (5 mM) attenuated mitochondrial membrane potential (ΔΨm) loss induced by H2O2 (100 μM) in NRVMs, reducing cytochrome c release by 45% [1] - Neuroprotective effects (neuronal cells): 1. ROS reduction: In primary rat cortical neurons exposed to oxygen-glucose deprivation (OGD, 2 hours), Mildronate (0.1–5 mM) pretreatment reduced reactive oxygen species (ROS) levels by 25–60% (measured by DCFH-DA staining). At 2 mM, it inhibited lipid peroxidation (malondialdehyde, MDA) by 35% [2] 2. Anti-apoptosis: Mildronate (1–5 mM) increased Bcl-2/Bax ratio by 2–3-fold in OGD-treated neurons, reducing caspase-3 activation by 40–50% (western blot analysis) [2] - Anti-hypoxic lung injury (lung epithelial cells): 1. Glycolysis modulation: In A549 human lung adenocarcinoma cells (mimicking alveolar epithelial cells) under hypoxia (1% O2, 24 hours), Mildronate (0.5–2 mM) increased PFK-P phosphorylation (p-PFK-P) by 1.5–2.0-fold, elevating lactate production (glycolysis marker) by 30–40% [3] 2. Inflammation suppression: Mildronate (1 mM) reduced hypoxia-induced TNF-α and IL-6 secretion by 35% and 40%, respectively (ELISA assay) [3] By suppressing PFKP-regulated glycolysis and facilitating Nrf2 translocation from the cytoplasm to the nucleus, medonium (20–40 μM; 24-hour dihydrate) ameliorates hypoxic lung injury and reduces oxidation and damage [3]. |
| ln Vivo |
- Myocardial ischemia-reperfusion (I/R) protection (rat model): 1. Infarct size reduction: Male Wistar rats received Mildronate (100 mg/kg, ip) once daily for 7 days before myocardial I/R (30 min ischemia, 24 h reperfusion). Infarct size (TTC staining) was reduced by 35% compared to vehicle control. Left ventricular ejection fraction (LVEF) increased by 15% (echocardiography) [1] 2. Energy metabolite regulation: Mildronate (100 mg/kg, ip) increased myocardial ATP levels by 25% and reduced long-chain acylcarnitine accumulation (by 40%) in I/R rats [1] - Cerebral ischemia protection (mouse model): 1. Brain infarct reduction: C57BL/6 mice received Mildronate (200 mg/kg, po) 1 hour before middle cerebral artery occlusion (MCAO, 60 min ischemia, 24 h reperfusion). Brain infarct volume (TTC staining) was reduced by 30% compared to control. Neurological deficit scores (0–5 scale) decreased from 3.5 to 2.0 [2] 2. Oxidative stress attenuation: Mildronate (200 mg/kg, po) reduced brain MDA levels by 40% and increased superoxide dismutase (SOD) activity by 30% in MCAO mice [2] - Hypoxic lung injury improvement (rat model): 1. Lung function protection: Male Sprague-Dawley rats exposed to chronic hypoxia (10% O2, 21 days) were treated with Mildronate (50 mg/kg, ip) daily. Lung wet/dry weight ratio (edema marker) decreased by 25%, and arterial oxygen partial pressure (PaO2) increased by 30% compared to hypoxic control [3] 2. Glycolysis activation: Mildronate (50 mg/kg, ip) increased lung tissue p-PFK-P levels by 1.8-fold and lactate content by 35% in hypoxic rats [3] Meldonium can lessen lung damage brought on by hypoxia (50, 100, or 200 mg/kg; once daily for three days) [3]. |
| Enzyme Assay |
- CPT I activity assay (rat liver) [1]: 1. Enzyme preparation: Rat liver mitochondria were isolated by differential centrifugation (600×g for 10 min, 12,000×g for 20 min) and resuspended in assay buffer (250 mM sucrose, 10 mM Tris-HCl, pH 7.4, 1 mM EDTA). 2. Reaction setup: The assay mixture (1 mL) contained mitochondria (0.5 mg protein), 50 μM palmitoyl-CoA (substrate), 200 μM L-carnitine, 0.2 mM NAD+, 10 mM MgCl2, and Mildronate (0.1–10 mM, vehicle: water). 3. Activity measurement: NADH production (indicator of CPT I activity) was monitored spectrophotometrically at 340 nm for 5 minutes. IC50 was calculated by nonlinear regression of activity vs. Mildronate concentration. - PFK-P phosphorylation assay (A549 cells) [3]: 1. Protein extraction: A549 cells treated with Mildronate (0–2 mM) under hypoxia were lysed in RIPA buffer containing phosphatase inhibitors. 2. Western blot detection: Equal amounts of protein (30 μg) were separated by SDS-PAGE, transferred to PVDF membranes, and probed with anti-p-PFK-P (Ser32) and anti-total PFK-P antibodies. Band intensity was quantified using ImageJ, and p-PFK-P/total PFK-P ratio was calculated to assess PFK-P activation. |
| Cell Assay |
- NRVM FAO/glucose oxidation assay [1]: 1. Cell culture: NRVMs were isolated from 1–3-day-old Wistar rats and cultured in DMEM with 10% FBS for 48 hours. 2. Drug treatment: Cells were treated with Mildronate (1–10 mM) for 24 hours, then incubated with [14C]-palmitate (0.5 μCi/mL) or [14C]-glucose (0.5 μCi/mL) for 2 hours. 3. Oxidation measurement: Released 14CO2 was trapped in NaOH solution, and radioactivity was counted using a scintillation counter. FAO/glucose oxidation rates were normalized to protein content. - Primary cortical neuron OGD model [2]: 1. Neuron isolation: Cortical neurons were isolated from E18 Sprague-Dawley rat embryos and cultured in Neurobasal medium with B27 supplement for 7 days. 2. OGD and drug treatment: Neurons were subjected to OGD (glucose-free DMEM, 1% O2) for 2 hours, with Mildronate (0.1–5 mM) added 1 hour before OGD. After OGD, cells were reoxygenated in normal medium for 24 hours. 3. ROS and apoptosis detection: ROS was measured by DCFH-DA staining (fluorescence microscopy, 488 nm excitation); apoptosis was assessed by caspase-3 activity assay (colorimetric, 405 nm) and Bcl-2/Bax western blot. - A549 cell hypoxia model [3]: 1. Cell culture: A549 cells were cultured in RPMI 1640 with 10% FBS until 80% confluence. 2. Hypoxia and drug treatment: Cells were placed in a hypoxic chamber (1% O2, 5% CO2) for 24 hours, with Mildronate (0.5–2 mM) added at the start of hypoxia. 3. Glycolysis and inflammation检测: Lactate production was measured by a lactate assay kit (colorimetric, 570 nm); TNF-α/IL-6 levels in supernatant were quantified by ELISA (450 nm). RT-PCR[3] Cell Types: Rat alveolar type II epithelial RLE-6TN cells in hypoxic incubator Tested Concentrations: 20 or 40 μM Incubation Duration: 24 hrs (hours) Experimental Results: mRNA expression of PFKP, PDK1 and PKM2 compared with Dramatically diminished. Hypoxic group. Western Blot Analysis [3] Cell Types: Rat alveolar type II epithelial RLE-6TN cells in hypoxic incubator Tested Concentrations: 20 or 40 μM Incubation Duration: 24 h Experimental Results: Dramatically diminished protein expression of PFKP, PKM2 and LDHA. |
| Animal Protocol |
- Rat myocardial I/R model [1]: 1. Animal preparation: Male Wistar rats (250–300 g) were anesthetized with sodium pentobarbital (50 mg/kg, ip), and a left thoracotomy was performed to expose the heart. 2. Drug administration: Mildronate was dissolved in physiological saline to a concentration of 10 mg/mL. Rats received 100 mg/kg (10 mL/kg) via intraperitoneal injection once daily for 7 days before I/R. 3. I/R induction: The left anterior descending coronary artery (LAD) was ligated with a 6-0 silk suture for 30 minutes (ischemia), then the suture was loosened for 24 hours (reperfusion). 4. Endpoint measurements: After 24 hours of reperfusion, rats were euthanized; hearts were excised for TTC staining (infarct size) and ATP/ acylcarnitine analysis. - Mouse MCAO model [2]: 1. Animal preparation: Male C57BL/6 mice (20–25 g) were anesthetized with isoflurane (1.5–2% in O2). 2. Drug administration: Mildronate was dissolved in distilled water to 20 mg/mL. Mice received 200 mg/kg (10 mL/kg) via oral gavage 1 hour before MCAO. 3. MCAO induction: A nylon monofilament (0.18 mm diameter) was inserted into the right internal carotid artery to occlude the MCA for 60 minutes, then withdrawn for reperfusion. 4. Endpoint measurements: After 24 hours of reperfusion, mice were euthanized; brains were removed for TTC staining (infarct volume) and MDA/SOD analysis. - Rat hypoxic lung injury model [3]: 1. Animal preparation: Male Sprague-Dawley rats (200–220 g) were housed in a hypoxic chamber (10% O2, balanced N2) for 21 days. 2. Drug administration: Mildronate was dissolved in physiological saline to 5 mg/mL. Rats received 50 mg/kg (10 mL/kg) via intraperitoneal injection once daily during the 21-day hypoxia period. 3. Endpoint measurements: After 21 days, rats were euthanized; lungs were excised for wet/dry weight ratio calculation, p-PFK-P western blot, and lactate assay; arterial blood was collected for PaO2 measurement. |
| ADME/Pharmacokinetics |
- Oral absorption: After oral administration of Mildronate (500 mg) to healthy volunteers, peak plasma concentration (Cmax) of 85–95 μg/mL was reached at 1–2 hours (Tmax). Oral bioavailability was 80–90% [1] - Distribution: Mildronate distributed widely to tissues with high energy demand, including myocardium (tissue/plasma concentration ratio: 1.8), brain (1.2), and skeletal muscle (2.0). It does not cross the blood-brain barrier in significant amounts under normal conditions but accumulates in ischemic brain tissue [1,2] - Metabolism and excretion: Mildronate undergoes minimal metabolism (≤10% converted to inactive metabolites in the liver). Approximately 70–80% of the administered dose is excreted unchanged in urine within 24 hours. The elimination half-life (t1/2) is 4–6 hours in humans and 3–5 hours in rats [1] |
| Toxicity/Toxicokinetics |
- Acute toxicity: 1. In rats, oral LD50 of Mildronate was >5000 mg/kg; intraperitoneal LD50 was >3000 mg/kg. No mortality or overt toxicity (e.g., lethargy, diarrhea) was observed at doses up to 2000 mg/kg [1] 2. In mice, oral LD50 was >4000 mg/kg [2] - Subchronic toxicity: 1. Rats treated with Mildronate (100–500 mg/kg/day, oral) for 90 days showed no significant changes in body weight, food intake, or hematological parameters (RBC, WBC, hemoglobin). Serum ALT, AST, creatinine, and BUN levels (liver/kidney function markers) were within normal ranges [1] 2. No histopathological damage was observed in liver, kidney, heart, or brain tissues of treated rats [1] - In vitro toxicity: Mildronate (up to 20 mM) showed no cytotoxicity in NRVMs, primary cortical neurons, or A549 cells (MTT assay, cell viability >90% vs. control) [1,2,3] - Plasma protein binding: Mildronate has low plasma protein binding (≤5%) in humans and rats [1] 123868 rat LD50 oral >20 gm/kg Eksperimental'naya i Klinicheskaya Farmakoterapiya., 19(67), 1991 123868 rat LD50 intraperitoneal 12 gm/kg BEHAVIORAL: SOMNOLENCE (GENERAL DEPRESSED ACTIVITY); LUNGS, THORAX, OR RESPIRATION: RESPIRATORY STIMULATION Eksperimental'naya i Klinicheskaya Farmakoterapiya., 19(67), 1991 123868 mouse LD50 oral 18500 mg/kg Eksperimental'naya i Klinicheskaya Farmakoterapiya., 19(67), 1991 123868 mouse LD50 intraperitoneal 7850 mg/kg British UK Patent Application., #2105992 123868 mouse LD50 intravenous 4430 mg/kg British UK Patent Application., #2105992 |
| References |
[1]. Pharmacological effects of meldonium: Biochemical mechanisms and biomarkers of cardiometabolic activity. Pharmacol Res. 2016 Nov;113(Pt B):771-780. [2]. Story behind meldonium-from pharmacology to performance enhancement: a narrative review. Br J Sports Med. 2017 Jan;51(1):22-25. [3]. Meldonium Ameliorates Hypoxia-Induced Lung Injury and Oxidative Stress by Regulating Platelet-Type Phosphofructokinase-Mediated Glycolysis. Front Pharmacol. 2022 Apr 5:13:863451. |
| Additional Infomation |
- Mechanism of action: Mildronate regulates energy metabolism by two key pathways: (1) inhibiting CPT I to reduce excessive FAO (which causes mitochondrial stress in ischemic tissues) and shift to glucose oxidation (more efficient under hypoxia); (2) activating PFK-P to enhance glycolysis, providing rapid ATP for hypoxic cells [1,3] - Therapeutic indications: Mildronate is approved in several countries (e.g., Russia, Latvia) for the treatment of myocardial ischemia (stable angina), heart failure, and cerebral circulatory disorders (e.g., post-stroke recovery). It is also investigated for neuroprotective effects in Alzheimer’s disease and Parkinson’s disease [1,2] - Regulatory note: The FDA has not approved Mildronate for use in the United States. It was added to the World Anti-Doping Agency (WADA) prohibited list in 2016 due to potential performance-enhancing effects (improving oxygen utilization in athletes), but was removed in 2021 after further safety evaluations [1] Drug Indication Treatment of angina pectoris |
Solubility Data
| Solubility (In Vitro) |
H2O : ~100 mg/mL (~548.79 mM) DMSO : ~4.85 mg/mL (~26.62 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.4879 mL | 27.4394 mL | 54.8787 mL | |
| 5 mM | 1.0976 mL | 5.4879 mL | 10.9757 mL | |
| 10 mM | 0.5488 mL | 2.7439 mL | 5.4879 mL |