Physicochemical Properties
| Molecular Formula | C29H29D6NO2 |
| Molecular Weight | 435.63 |
| Exact Mass | 432.285 |
| CAS # | 1228097-18-4 |
| PubChem CID | 46782374 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 3.8 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 32 |
| Complexity | 921 |
| Defined Atom Stereocenter Count | 5 |
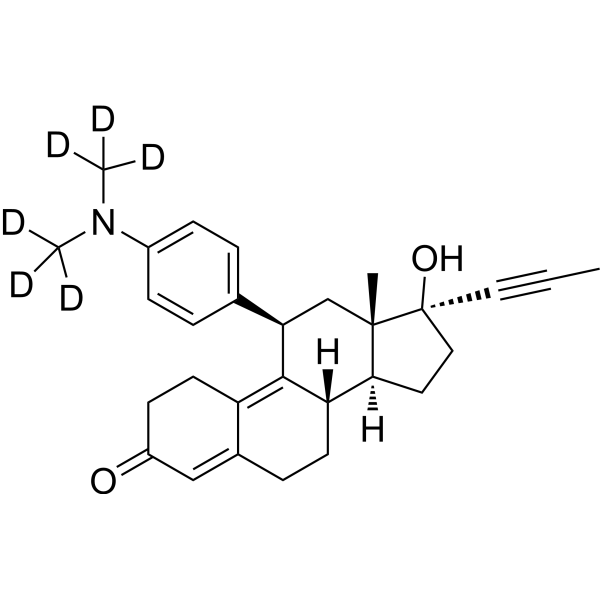
| SMILES | [2H]C([2H])([2H])N(C)C1=CC=C(C=C1)[C@H]2C[C@]3([C@@H](CC[C@]3(C#CC)O)[C@H]4C2=C5CCC(=O)C=C5CC4)C |
| InChi Key | VKHAHZOOUSRJNA-PBWRPEOTSA-N |
| InChi Code | InChI=1S/C29H35NO2/c1-5-15-29(32)16-14-26-24-12-8-20-17-22(31)11-13-23(20)27(24)25(18-28(26,29)2)19-6-9-21(10-7-19)30(3)4/h6-7,9-10,17,24-26,32H,8,11-14,16,18H2,1-4H3/t24-,25+,26-,28-,29-/m0/s1/i3D3 |
| Chemical Name | (8S,11R,13S,14S,17S)-17-hydroxy-13-methyl-11-[4-[methyl(trideuteriomethyl)amino]phenyl]-17-prop-1-ynyl-1,2,6,7,8,11,12,14,15,16-decahydrocyclopenta[a]phenanthren-3-one |
| Synonyms | Mifepristone-d6; RU486-d6; RU 38486-d6 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Stable heavy isotopes of hydrogen, carbon, and other elements have been incorporated into drug molecules, largely as tracers for quantitation during the drug development process. Studies involving the human use of drugs with labeled deuterium suggest that these compounds may offer some advantages when compared with their nondeuterated counterparts. Deuteration has gained attention because of its potential to affect the pharmacokinetic and metabolic profiles of drugs. Deutetrabenazine is the first deuterated drug to receive Food and Drug Administration approval. This deuterated form of the drug tetrabenazine is indicated for the treatment of chorea associated with Huntington's disease as well as tardive dyskinesia. Ongoing clinical trials suggest that a number of other deuterated compounds are being evaluated for the treatment of human diseases and not merely as research tools. [1] |
| ln Vivo | The cervix tumor xenograft models are treated with NSC 119875 alone, there is a tumor growth inhibition compare with control group. However, the tumor weight loss is even more significant (p<0.05) with the combination of NSC 119875 and Mifepristone at the doses used, showing a decrease of ~50% compared with the treatments alone by the end of the study[3]. Adult male Sprague-Dawley rats are subjected to a 4-day binge-like EtOH administration regimen (3 to 5 g/kg/i.g. every 8 hours designed to produce peak blood EtOH levels (BELs) of <300 mg/dL). Subgroups of animals receive s.c. injection of Mifepristone (20 or 40 mg/kg in peanut oil). Although Mifepristone produces no significant changes in behavior of EtOH-na?ve animals, pretreatment with Mifepristone (40 mg/kg) significantly reducesthe severity of EtOH withdrawal. Asignificant interaction between diet and drug, F(5,55)=3.92, p<0.05, such that EtOH-treated animals receiving vehicle or 20 mg/kg of Mifepristone displayssignificantly more signs of EtOH withdrawal than does EtOH-na?ve animals receiving the same drug treatment. Importantly, treatment with 40 mg/kg of Mifepristone significantly reduces the severity of EtOH withdrawal, in a dose-dependent manner[4]. |
| References |
[1]. Impact of Deuterium Substitution on the Pharmacokinetics of Pharmaceuticals. Ann Pharmacother. 2019 Feb;53(2):211-216. [2]. NSC 119875 cytotoxicity is increased by mifepristone in cervical carcinoma: an in vitro and in vivo study. Oncol Rep. 2009 Nov;22(5):1237-45. [3]. Mifepristone Pretreatment Reduces Ethanol Withdrawal Severity In Vivo. Alcohol Clin Exp Res. 2013 Aug;37(8):1417-23. [4]. New progesterone receptor antagonists: phosphorus-containing 11beta-aryl-substituted steroids. Bioorg Med Chem. 2006 Oct 1;14(19):6726-32. [5]. Progesterone Promotes Endothelial Nitric Oxide Synthase Expression Through Enhancing Nuclear Progesterone receptor-SP1 Formation. Am J Physiol Heart Circ Physiol. 2020 Jul 3. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.2955 mL | 11.4776 mL | 22.9553 mL | |
| 5 mM | 0.4591 mL | 2.2955 mL | 4.5911 mL | |
| 10 mM | 0.2296 mL | 1.1478 mL | 2.2955 mL |