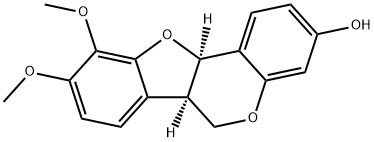
Methylnissolin, also known as Astrapterocarpan, isolated from Astragalus membranaceus, is a novel and potent bioactive compound. Methylnissolin prevents platelet-derived growth factor (PDGF)-BB-induced cell proliferation with an IC50 of 10 μM. Methylnissolin also prevents PDGF-BB-induced phosphorylation of extracellular signal-regulated kinase 1/2 (ERIC1/2) mitogen-activated protein (MAP) kinase. These effects are achieved by inhibiting the ERK1/2 MAP kinase cascade.
Physicochemical Properties
| Molecular Formula | C17H16O5 |
| Molecular Weight | 300.3059 |
| Exact Mass | 300.099 |
| CAS # | 73340-41-7 |
| Related CAS # | 73340-41-7 |
| PubChem CID | 14077830 |
| Appearance | White to off-white solid |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 428.9±45.0 °C at 760 mmHg |
| Flash Point | 213.2±28.7 °C |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.612 |
| LogP | 2.45 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 22 |
| Complexity | 403 |
| Defined Atom Stereocenter Count | 2 |
| SMILES | O1C2C(=C(C([H])=C([H])C=2[C@@]2([H])C([H])([H])OC3C([H])=C(C([H])=C([H])C=3[C@@]12[H])O[H])OC([H])([H])[H])OC([H])([H])[H] |
| InChi Key | UOVGCLXUTLXAEC-WFASDCNBSA-N |
| InChi Code | InChI=1S/C17H16O5/c1-19-13-6-5-10-12-8-21-14-7-9(18)3-4-11(14)15(12)22-16(10)17(13)20-2/h3-7,12,15,18H,8H2,1-2H3/t12-,15-/m0/s1 |
| Chemical Name | (6aR,11aR)-9,10-dimethoxy-6a,11a-dihydro-6H-[1]benzofuro[3,2-c]chromen-3-ol |
| Synonyms | Methylnissolin; Astrapterocarpan |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: This product requires protection from light (avoid light exposure) during transportation and storage. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | TrkC (IC50 = 3 nM); TrkB (IC50 = 4 nM); TrkA (IC50 = 6 nM) |
| ln Vitro | PF-06273340 is an exceptionally strong pan-Trk inhibitor that possesses a superior LipE profile. Through a series of in vitro safety assays, PF-06273340 is profiled and found to have little cytotoxicity in either HepG2 or THLE cell lines (IC50 > 300 μM or > 42 μM, respectively). With the exception of COX-1 (IC50 = 2.7 μM), dopamine transporter assays (Ki = 5.2 μM), and PDEs 4D, 5A, 7B, 8B, and 11 (54−89% inhibition at 10 μM), all IC50/Ki values in this broad panel were >10 μM. PF-06273340 is screened against 309 kinases in the Invitrogen wide kinase panel. All of the kinases, with the exception of MUSK (IC50 53 nM), FLT-3 (IC50 395 nM), IRAK1 (IC50 2.5 μM), MKK (90% @ 1 μM), and DDR1 (60% @ 1 μM), were inhibited by less than 40% when tested at 1 μM[1]. |
| ln Vivo | In rats, decreases in white blood cell count are observed from 150 mg/kg/day. Increases in food intake and body weight gain are seen at doses greater than 250 mg/kg; these effects may be explained by central inhibition of TrkB, agonists of which are known to be anorexigenic in rodents. Microscopic observations reveal adaptive alterations in the liver, which are correlated with elevated liver weight (≥250 mg/kg) and elevated cholesterol (1000 mg/kg). PF-06273340 is generally well tolerated up to 1000 mg/kg/day, with an IC50 of roughly 400×TrkA for unbound Cavg plasma exposure[1]. |
| Animal Protocol |
Male SD rats 0.25, 2.5 and 25 mg/kg oral administration |
| References |
[1]. J Med Chem . 2016 Nov 23;59(22):10084-10099. |
| Additional Infomation |
Astrapterocarpan is a member of pterocarpans. Astrapterocarpan has been reported in Dalbergia odorifera with data available. |
Solubility Data
| Solubility (In Vitro) |
DMSO: 50~95 mg/mL (166.5~198 mM) Ethanol: ~1 mg/mL (~2.1 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (8.32 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (8.32 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (8.32 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.3299 mL | 16.6495 mL | 33.2989 mL | |
| 5 mM | 0.6660 mL | 3.3299 mL | 6.6598 mL | |
| 10 mM | 0.3330 mL | 1.6649 mL | 3.3299 mL |