Physicochemical Properties
| Molecular Formula | C8H8O4 |
| Molecular Weight | 168.1467 |
| Exact Mass | 168.042 |
| CAS # | 2150-43-8 |
| Related CAS # | Methyl 3,4-dihydroxybenzoate-d3-1;2733147-54-9 |
| PubChem CID | 287064 |
| Appearance | White to off-white solid |
| Density | 1.4±0.1 g/cm3 |
| Boiling Point | 351.5±22.0 °C at 760 mmHg |
| Melting Point | 134-135°C |
| Flash Point | 148.5±15.8 °C |
| Vapour Pressure | 0.0±0.8 mmHg at 25°C |
| Index of Refraction | 1.588 |
| LogP | 1.69 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 12 |
| Complexity | 168 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O(C([H])([H])[H])C(C1C([H])=C([H])C(=C(C=1[H])O[H])O[H])=O |
| InChi Key | CUFLZUDASVUNOE-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C8H8O4/c1-12-8(11)5-2-3-6(9)7(10)4-5/h2-4,9-10H,1H3 |
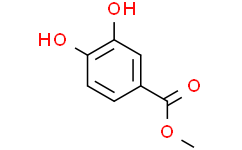
| Chemical Name | methyl 3,4-dihydroxybenzoate |
| Synonyms | Methyl 3,4-dihydroxybenzoate; Methyl protocatechuate; Protocatechuic acid methyl ester |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Methyl 3,4-dihydroxybenzoate targets oxidative stress regulatory pathways, inflammatory response-related molecules, and apoptosis signaling proteins [1,2] |
| ln Vitro |
In A549 cells, methyl 3,4-dihydroxybenzoate (also known as methyl protocatechuate; protocatechuic acid methyl ester) reduces the toxicity of F- by modifying its bioavailability, intracellular calcium concentration, mitochondrial membrane integrity, and redox signaling[1]. Methyl 3,4-dihydroxybenzoate (10 μM–40 μM) dose-dependently protected A549 cells from fluoride-induced toxicity: 40 μM increased cell viability from 52% (fluoride alone) to 85% after 48 hours, as assessed by MTT assay [1] The compound (10 μM–40 μM) reduced fluoride-induced oxidative stress in A549 cells: 40 μM decreased intracellular reactive oxygen species (ROS) levels by 62%, malondialdehyde (MDA) content by 58%, and increased superoxide dismutase (SOD) activity by 45% and glutathione peroxidase (GSH-Px) activity by 53% [1] Methyl 3,4-dihydroxybenzoate (40 μM) inhibited fluoride-induced apoptosis in A549 cells: apoptotic rate decreased from 38% to 12% (Annexin V+/PI+), accompanied by a 2.3-fold increase in Bcl-2 protein expression, 65% reduction in Bax protein, and 70% decrease in cleaved caspase-3 levels (Western blot detection) [1] |
| ln Vivo |
Methyl 3,4-dihydroxybenzoate, also known as methyl protocatechuate (25 or 50 mg/kg bw/day), reduces oxidative stress and cellular F- accumulation. By reactivating RAGE and Nrf2 expression, methyl 3,4-dihydroxybenzoate slows the progression of inflammation and the fibrosis it causes[2]. In Wistar rats with fluoride-induced pulmonary toxicity (100 ppm NaF in drinking water for 8 weeks), oral administration of Methyl 3,4-dihydroxybenzoate (50 mg/kg, 100 mg/kg, q.d.) for 8 weeks dose-dependently alleviated lung injury [2] The 100 mg/kg dose reduced lung tissue MDA content by 63%, increased SOD activity by 58% and GSH-Px activity by 61%, and decreased myeloperoxidase (MPO) activity (inflammatory marker) by 65% [2] Methyl 3,4-dihydroxybenzoate (100 mg/kg) downregulated serum pro-inflammatory cytokines: TNFα levels decreased by 68% and IL-6 levels by 72%, and improved lung histopathology (reduced alveolar damage and inflammatory cell infiltration) [2] |
| Enzyme Assay |
Antioxidant enzyme activity assay: A549 cell lysates or rat lung tissue homogenates were incubated with assay buffers specific for SOD, GSH-Px, or catalase (CAT). For SOD, xanthine oxidase method was used to measure inhibition of superoxide anion; for GSH-Px, dithionitrobenzoic acid method was used to detect GSH oxidation; absorbance was measured at specific wavelengths to quantify enzyme activity [1,2] Myeloperoxidase (MPO) activity assay: Rat lung tissue homogenates were mixed with assay buffer containing o-phenylenediamine and hydrogen peroxide. The reaction was conducted at 37°C for 30 minutes, and absorbance at 460 nm was measured to reflect MPO activity (inflammatory index) [2] |
| Cell Assay |
Cell viability assay: A549 cells were seeded in 96-well plates (5 × 10³ cells/well) and pre-treated with Methyl 3,4-dihydroxybenzoate (10 μM–40 μM) for 2 hours, then exposed to fluoride (200 μM) for 48 hours. MTT reagent was added, and absorbance at 570 nm was measured to calculate cell viability [1] ROS detection assay: A549 cells were loaded with DCFH-DA fluorescent probe (20 μM) for 30 minutes, pre-treated with Methyl 3,4-dihydroxybenzoate (10 μM–40 μM) for 2 hours, then exposed to fluoride. ROS levels were quantified by flow cytometry and fluorescence microscopy [1] Apoptosis and Western blot assay: A549 cells were treated with Methyl 3,4-dihydroxybenzoate (40 μM) and fluoride for 48 hours. Apoptosis was detected by Annexin V-FITC/PI staining and flow cytometry; cell lysates were prepared for Western blot to detect Bcl-2, Bax, and cleaved caspase-3 protein levels [1] |
| Animal Protocol |
Fluoride-induced pulmonary toxicity rat model: Wistar rats (180–220 g) were randomized into 4 groups (n=6/group): control (normal drinking water), fluoride alone (100 ppm NaF in drinking water), low-dose drug (fluoride + 50 mg/kg Methyl 3,4-dihydroxybenzoate), high-dose drug (fluoride + 100 mg/kg Methyl 3,4-dihydroxybenzoate). The compound was dissolved in normal saline and administered by gavage once daily for 8 weeks. Rats were sacrificed, and serum and lung tissues were collected for biochemical analysis and histopathological examination [2] |
| Toxicity/Toxicokinetics |
In A549 cells, Methyl 3,4-dihydroxybenzoate at concentrations up to 40 μM did not cause cytotoxicity (cell viability > 95%) [1] In rats treated with Methyl 3,4-dihydroxybenzoate (100 mg/kg, p.o., q.d. for 8 weeks): no significant changes in body weight, hematological parameters (WBC, RBC, platelets), or biochemical parameters (ALT, AST, BUN, creatinine) were observed. Histopathological examination of liver, kidney, and heart showed no drug-related lesions [2] |
| References |
[1]. Protocatechuic acid methyl ester ameliorates fluoride toxicity in A549 cells. Food Chem Toxicol. 2017 Nov;109(Pt 2):941-950. [2]. Protocatechuic acid methyl ester modulates fluoride induced pulmonary toxicity in rats. Food Chem Toxicol. 2018 Aug;118:235-244. |
| Additional Infomation |
Methyl 3,4-dihydroxybenzoate is a methyl ester resulting from the formal condensation of the carboxy group of 3,4-dihydroxybenzoic acid with methanol. It has a role as an antioxidant, a neuroprotective agent and a plant metabolite. It is a methyl ester and a member of catechols. It is functionally related to a 3,4-dihydroxybenzoic acid. Methyl 3,4-dihydroxybenzoate has been reported in Camellia sinensis, Perilla frutescens, and other organisms with data available. See also: Acai fruit pulp (part of). Methyl 3,4-dihydroxybenzoate (protocatechuic acid methyl ester) is a natural phenolic ester widely distributed in plants [1,2] Its mechanism of alleviating fluoride toxicity involves scavenging reactive oxygen species, enhancing antioxidant enzyme activity, inhibiting lipid peroxidation, downregulating pro-inflammatory cytokines, and suppressing the mitochondrial apoptotic pathway [1,2] The compound exhibits potent antioxidant and anti-inflammatory activities, with potential applications in mitigating fluoride-induced tissue damage [1,2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~50 mg/mL (~297.35 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (14.87 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (14.87 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (14.87 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.9471 mL | 29.7354 mL | 59.4707 mL | |
| 5 mM | 1.1894 mL | 5.9471 mL | 11.8941 mL | |
| 10 mM | 0.5947 mL | 2.9735 mL | 5.9471 mL |