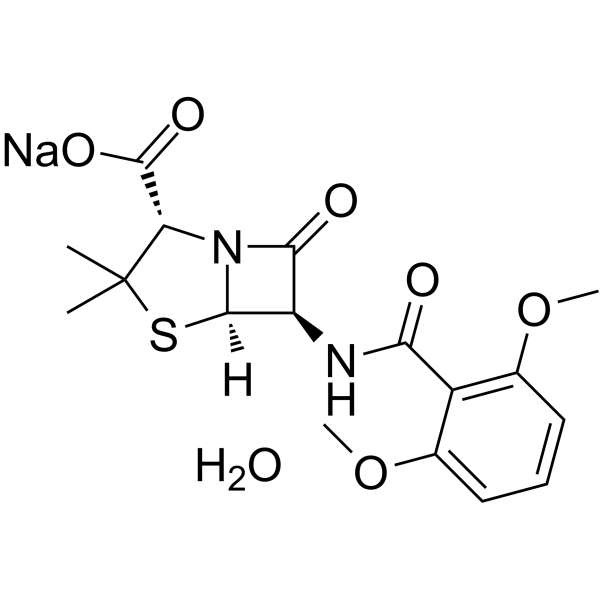
Description: Methicillin sodium hydrate is a narrow-spectrum β-lactam antibiotic of the penicillin class with the potential to be used for skin infections, osteomyelitis, and endocarditis. It has been used to treat infections caused by susceptible Gram-positive bacteria, in particular, penicillinase-producing organisms such as Staphylococcus aureus that would otherwise be resistant to most penicillins. Meticillin acts by inhibiting penicillin-binding proteins (PBPs) and is active against Staphylococcus aureus and Staphylococcus epidermidis that are resistant to other penicillins.
Physicochemical Properties
| Molecular Formula | C17H20N2O6S.NA+ |
| Molecular Weight | 403.4053 |
| Exact Mass | 402.086 |
| CAS # | 7246-14-2 |
| Related CAS # | Methicillin sodium salt;132-92-3;Methicillin;61-32-5 |
| PubChem CID | 23678596 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 1.334 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 5 |
| Heavy Atom Count | 28 |
| Complexity | 606 |
| Defined Atom Stereocenter Count | 3 |
| SMILES | CC1([C@@H](N2[C@H](S1)[C@@H](C2=O)NC(=O)C3=C(C=CC=C3OC)OC)C(=O)[O-])C.O.[Na+] |
| InChi Key | NRZPASQBOYNGHR-HWROMZCQSA-M |
| InChi Code | InChI=1S/C17H20N2O6S.Na.H2O/c1-17(2)12(16(22)23)19-14(21)11(15(19)26-17)18-13(20)10-8(24-3)6-5-7-9(10)25-4;;/h5-7,11-12,15H,1-4H3,(H,18,20)(H,22,23);;1H2/q;+1;/p-1/t11-,12+,15-;;/m1../s1 |
| Chemical Name | sodium;(2S,5R,6R)-6-[(2,6-dimethoxybenzoyl)amino]-3,3-dimethyl-7-oxo-4-thia-1-azabicyclo[3.2.0]heptane-2-carboxylate;hydrate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Staphylococcus aureus CFU rapidly decreases when exposed to 20 μg/mL of methylmethacillin sodium hydrate over 24 hours [2]. |
| ln Vivo | In rabbits infected with Staphylococcus aureus, methicillin sodium hydrate (40 mg/kg intramuscular injection every 6 hours for 5 days) [2]. |
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Amounts of methicillin ingested by the infant in breastmilk are small and would not be expected to cause any adverse effects. Occasionally disruption of the infant's gastrointestinal flora, resulting in diarrhea or thrush have been reported with penicillins, but these effects have not been adequately evaluated. Methicillin is acceptable in nursing mothers. ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References |
[1]. Sharon S. Castle. Methicillin. xPharm: The Comprehensive Pharmacology Reference 2007, Pages 1-4. [2]. Treatment of Experimental Staphylococcus aureus Endocarditis: Comparison of Cephalothin, Cefazolin, and Methicillin. Antimicrob Agents Chemother. 1978 Jan; 13(1): 74–77. |
| Additional Infomation |
Methicillin sodium monohydrate is a hydrate. It contains a methicillin sodium. Methicillin Sodium is the sodium salt form of methicillin, a semisynthetic beta-lactam penicillin antibiotic with beta-lactamase resistant activity. Methicillin sodium is bactericidal and binds to specific penicillin-binding proteins on the bacterium and inhibits the transpeptidation enzyme, thereby preventing the cross-linking of peptidoglycans. This leads to an interruption of the bacterial cell wall and causes bacterial lysis. Methicillin sodium is active against beta-lactamase producing staphylococci. One of the PENICILLINS which is resistant to PENICILLINASE but susceptible to a penicillin-binding protein. It is inactivated by gastric acid so administered by injection. See also: Methicillin Sodium (annotation moved to). |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.4789 mL | 12.3943 mL | 24.7887 mL | |
| 5 mM | 0.4958 mL | 2.4789 mL | 4.9577 mL | |
| 10 mM | 0.2479 mL | 1.2394 mL | 2.4789 mL |