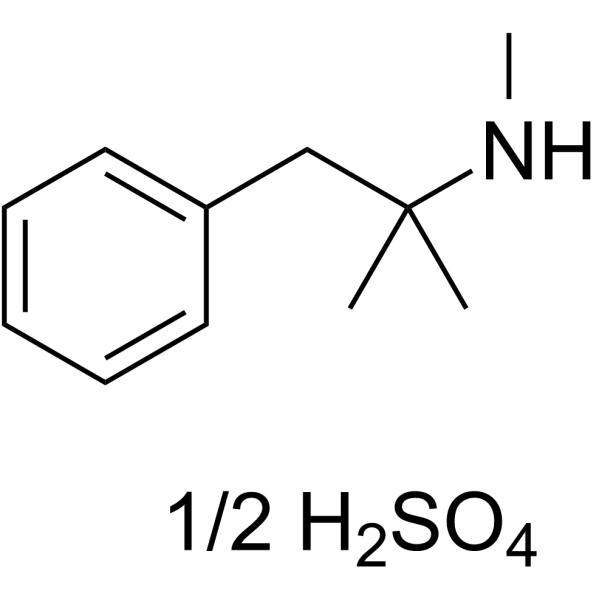
Physicochemical Properties
| Molecular Formula | C11H17N.1/2H2SO4 |
| Molecular Weight | 212.30 |
| Exact Mass | 424.24 |
| CAS # | 1212-72-2 |
| Related CAS # | Mephentermine hydrochloride;3978-34-5 |
| PubChem CID | 3677 |
| Appearance | LIQUID |
| Boiling Point | 229.4ºC at 760mmHg |
| Melting Point |
< 25 °C YELLOW CRYSTALS; MP: 155-156 °C /PICRATE/ < 25 °C |
| Flash Point | 90ºC |
| Vapour Pressure | 1.94E-20mmHg at 25°C |
| LogP | 5.664 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 12 |
| Complexity | 123 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | CC(C)(CC1=CC=CC=C1)NC.CC(C)(CC1=CC=CC=C1)NC.OS(=O)(=O)O |
| InChi Key | RXQCGGRTAILOIN-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H17N/c1-11(2,12-3)9-10-7-5-4-6-8-10/h4-8,12H,9H2,1-3H3 |
| Chemical Name | N,2-dimethyl-1-phenylpropan-2-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion ...MEPHENTERMINE.../IS/ WELL ABSORBED FROM DIGESTIVE TRACT. .../IT/ MAY BE ABSORBED TO A GREATER OR LESSER EXTENT FROM NASAL MUCOSA. THE MAJORITY OF THE DRUG IS EXCRETED IN THE URINE WITHIN 24 HR. MEPHENTERMINE IS REABSORBED IN THE RENAL TUBULES. EXCRETION OF THE DRUG & ITS METABOLITES IS MORE RAPID IN ACIDIC URINE. Metabolism / Metabolites Hepatic, by N-demethylation and then p-hydroxylation. MEPHENTERMINE IS METABOLIZED IN THE LIVER BY N-DEMETHYLATION & SUBSEQUENT P-HYDROXYLATION TO NORMEPHENTERMINE & P-HYDROXYNORMEPHENTERMINE. /SULFATE/ MEPHENTERMINE WAS METABOLIZED IN VITRO BY RABBIT LIVER MICROSOMES TO PHENTERMINE (II), N-HYDROXYMEPHENTERMINE (III), & N-HYDROXYPHENTERMINE. METABOLITES (II) & (III) PLUS UNCHANGED MEPHENTERMINE FOUND IN URINE OF HUMAN ADMIN SINGLE DOSE OF MEPHENTERMINE SULFATE. Metabolites of mephentermine (MP), phentermine (Ph), p-hydroxy-MP, p-hydroxy-Ph, N-hydroxy-MP and N-hydroxy-Ph on incubation with rat liver microsomal and cytosolic preparations were identified by glc and glc mass spectrometry. Identification of the metabolites indicated the following new metabolic routes of MP: NADPH dependent microsomal formation of p-hydroxy-MP from MP, of p-hydroxy-Ph from p-hydroxy-MP, and the NADH-dependent microsomal formation of Ph from N-hydroxy-Ph. The urinary excretion of mephentermine (I) and its major metabolite phentermine (II) in human volunteers over a period of several days after oral admin of the drug is described. The total proportion of the drug excreted during 54 hr was 57 to 83%. The ingestion of acetazolamide shortly after admin of I resulted in decr in excretion of both I and II during one day. The administration of furosemide only produced a urinary diluting effect during 2-4 hr after admin. Biological Half-Life 17 to 18 hours. 17 to 18 hours |
| Toxicity/Toxicokinetics |
Interactions IN ANESTHETIZED DOGS, RESERPINE POTENTIATED THE PRESSOR RESPONSE WHEN ADMIN AFTER MEPHENTERMINE. MEPHENTERMINE & DEXAMPHETAMINE SHOWED RAPID TACHYPHYLAXIS OF RESPONSES ON BLOOD PRESSURE WHEN REPEATED INJECTIONS ADMIN TO CATS & RATS. TACHYPHYLAXIS TO THESE SUBSTANCES WAS MUTUALLY CROSSED & WAS CROSSED TO PHENYLETHYLAMINE BUT NOT TO TYRAMINE BECAUSE OF SITE OF ACTION. TYRAMINE PROBABLY RELEASES NORADRENALINE FROM A DIFFERENT STORAGE SITE THAN DEXAMPHETAMINE, MEPHENTERMINE, & PHENYLETHYLAMINE BUT THE TWO STORES MAY BE FUNCTIONALLY CONNECTED. MEPHENTERMINE IN NONSTIMULATING DOSES OF 1 MG/KG PRODUCED AN ACUTE ERGOTROPIC RESPONSE (AER) WHEN ADMIN IV WITH 1 MG/KG RESERPINE TO DOGS & RABBITS. RABBITS PRETREATED WITH ALPHA-METHYL-M-TYROSINE (100 MG/KG) APPEARED NORMAL, CONSISTENTLY MANIFESTED ELECTROENCEPHALOGRAPHIC ACTIVATION TO EXTEROCEPTIVE STIMULI, & DISPLAYED AER IN RESPONSE TO DRUG COMBINATION. 5 MG/KG OF IMIPRAMINE-HCL PREVENTED THE AER, WHILE DEMETHYLIMIPRAMIME DID NOT BLOCK OR ALTER ITS COURSE. 1-5 MG/KG OF SCOPOLAMINE HYDROGEN BROMIDE PREVENTED ONLY THE ELECTROENCEPHALIC ACTIVATION OF THE AER. PRETREATMENT WITH IPRONIAZID (100 MG/KG) PROLONGED THE STIMULATION FOLLOWING THE DRUG COMBINATION BUT DID NOT CHANGE THE ACTIONS OF MEPHENTERMINE ALONE. PRETREATMENT OF RABBITS WITH RESERPINE REDUCED OR ABOLISHED THE CNS STIMULATION CAUSED BY MEPHENTERMINE BUT THRESHOLD DOSES OF METHAMPHETAMINE (1-5 MG/KG) PRODUCED CNS STIMULATION. RESERPINE (1 MG/KG) IN COMBINATION WITH MEPHENTERMINE (1 MG/KG) BUT NOT RESERPINE OR MEPHENTERMINE ALONE, PRODUCED ELECTROENCEPHALOGRAPH ACTIVATION PATTERN & ABOLISHED SINGLE SHOCK RESPONSES WITHIN MIDBRAIN RETICULAR FORMATION FOR ABOUT 28 MIN IN CURARIZED RABBITS. CHLORPROMAZINE (5 MG/KG) BLOCKED THE COMBINED EFFECT OF THE DRUGS. RESERPINE APPARENTLY FACILITATES INDIRECT ACTIONS OF MEPHENTERMINE, & THE PRESENCE OF BRAIN BIOGENIC AMINES ARE A REQUISITE TO THIS STIMULATION. For more Interactions (Complete) data for MEPHENTERMINE (27 total), please visit the HSDB record page. |
| References |
[1]. Mephentermine Misuse: An Impending Crisis among Sportspersons [published online ahead of print, 2021 Jun 14]. J Psychoactive Drugs. 2021;1-3. |
| Additional Infomation |
Mephentermine is a member of amphetamines. A sympathomimetic agent with mainly indirect effects on adrenergic receptors. It is used to maintain blood pressure in hypotensive states, for example, following spinal anesthesia. Although the central stimulant effects of mephentermine are much less than those of amphetamine, its use may lead to amphetamine-type dependence. (From Martindale, The Extra Pharmacopoeia, 30th ed, p1248) A sympathomimetic agent with specificity for alpha-1 adrenergic receptors. It is used to maintain BLOOD PRESSURE in hypotensive states such as following SPINAL ANESTHESIA. See also: Mephentermine sulfate dihydrate (annotation moved to). Drug Indication Used to maintain blood pressure in hypotensive states. Mechanism of Action Mephentermine is an alpha adrenergic receptor agonist, but also acts indirectly by releasing endogenous norepinephrine. Cardiac output and systolic and diastolic pressures are usually increased. A change in heart rate is variable, depending on the degree of vagal tone. Sometimes the net vascular effect may be vasodilation. Large doses may depress the myocardium or produce central nervous system (CNS) effects. Mephentermine is a sympathomimetic drug that acts both directly and indirectly; it has many similarities to ephedrine. After an intramuscular injection, the onset of action is prompt (within 5 to 15 minutes), and effects may last for several hours. Since the drug releases norepinephrine, cardiac contraction is enhanced and cardiac output and systolic and diastolic pressures are usually increased. The change in heart rate is variable, depending on the degree of vagal tone. Therapeutic Uses Adrenergic alpha-Agonists; Adrenergic Agents; Sympathomimetics; Vasoconstrictor Agents Mephentermine ... is used to prevent hypotension, which frequently accompanies spinal anesthesia. IT IS NOT RECOMMENDED FOR ROUTINE USE IN MANAGEMENT OF SHOCK, ESPECIALLY HYPOVOLEMIC SHOCK, ALTHOUGH IT CAN BE GIVEN AS INTERIM DRUG WHILE PREPARATIONS ARE BEING MADE FOR FLUID REPLACeMENT & OTHER MEASURES. /SULFATE/ ...FREE BASE CAN BE EMPLOYED TOPICALLY AS MYDRIATIC. For more Therapeutic Uses (Complete) data for MEPHENTERMINE (9 total), please visit the HSDB record page. Drug Warnings Mephentermine may produce arrhythmias, including extrasystoles, and AV block and hypertension. Arrhythmias are most likely to occur in patients with heart disease or those receiving other drugs which may increase cardiac irritability such as cyclopropane or halogenated hydrocarbon general anesthetics. /Mephentermine sulfate/ The CNS stimulating effects of mephentermine may result in nervousness, anxiety, seizures, or tachycardia. Large overdosage of the drug has caused visual hallucinations of colored geometric forms, paranoid psychosis, and euphoria. Drowsiness, weeping, incoherence, weakness, numbness, and tingling of the extremities have been reported. Withdrawal of the drug results in rapid disappearance of adverse CNS effects. /Mephentermine sulfate/ AS THE FREE BASE IS CURRENTLY USED, UNTOWARD EFFECTS ARE VERY UNCOMMON. MEPHENTERMINE MAY INCR UTERINE CONTRACTIONS ESPECIALLY DURING THE THIRD TRIMESTER OF PREGNANCY &, THEREFORE, SHOULD NOT BE USED IN PREGNANT WOMEN UNLESS THE POTENTIAL BENEFITS OUTWEIGH THE POSSIBLE RISKS. /MEPHENTERMINE SULFATE/ For more Drug Warnings (Complete) data for MEPHENTERMINE (10 total), please visit the HSDB record page. Pharmacodynamics Mephentermine is a sympathomimetic agent with mainly indirect effects on adrenergic receptors. It is used to maintain blood pressure in hypotensive states, for example, following spinal anesthesia. Although the central stimulant effects of mephentermine are much less than those of amphetamine, its use may lead to amphetamine-type dependence. (From Martindale, The Extra Pharmacopoeia, 30th ed, p1248) |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.7103 mL | 23.5516 mL | 47.1032 mL | |
| 5 mM | 0.9421 mL | 4.7103 mL | 9.4206 mL | |
| 10 mM | 0.4710 mL | 2.3552 mL | 4.7103 mL |