Physicochemical Properties
| Molecular Formula | C11H9NAO5S |
| Molecular Weight | 276.24 |
| Exact Mass | 276.006 |
| CAS # | 130-37-0 |
| Related CAS # | Menadione;58-27-5 |
| PubChem CID | 23665888 |
| Appearance | White to off-white solid powder |
| Melting Point | 121-124ºC |
| LogP | 1.84 |
| Hydrogen Bond Donor Count | 0 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 18 |
| Complexity | 466 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | XDPFHGWVCTXHDX-UHFFFAOYSA-M |
| InChi Code | InChI=1S/C11H10O5S.Na/c1-11(17(14,15)16)6-9(12)7-4-2-3-5-8(7)10(11)13;/h2-5H,6H2,1H3,(H,14,15,16);/q;+1/p-1 |
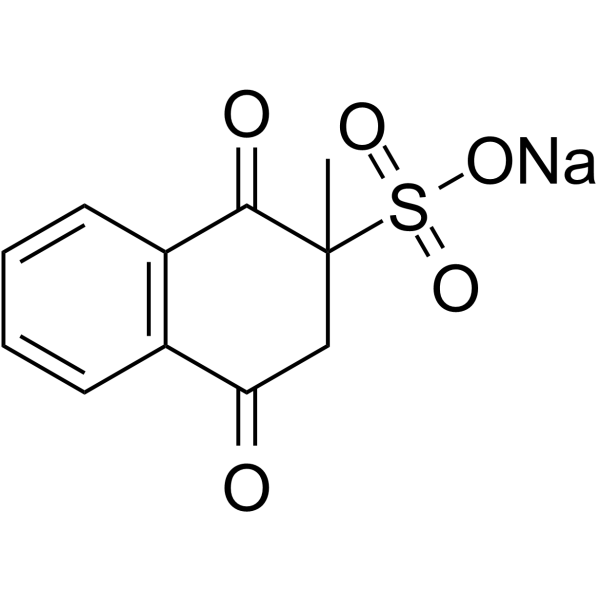
| Chemical Name | sodium;2-methyl-1,4-dioxo-3H-naphthalene-2-sulfonate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: Please store this product in a sealed and protected environment, avoid exposure to moisture. |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Menadione bisulfite sodium is a pro-oxidant compound. Upon cellular internalization and metabolism, it generates reactive oxygen species (ROS), specifically superoxide anion (O₂⁺•) and hydrogen peroxide (H₂O₂), through redox cycling. [1] |
| ln Vitro |
Menadione sodium bisulfite (500 μM) treatment and the accompanying increase in ROS have major impacts on the photosynthetic activity of cells. Treatment with menadione sodium bisulfite (500 μM) lowers global and particular antioxidant defenses [1]. Menadione bisulfite and PABA trigger similar defensive responses (PR, ROS and HR). Menadione sodium bisulfite enhances local and systemic resistance to stem point canker caused by Macrococcus sp. Treatment with menadione and sodium bisulfite can generate resistance to downy mildew in pearl millet. Menadione sodium bisulfite works by activating specific plant inherent defense systems. Menadione treatment with sodium bisulfite upregulates the expression of the gene producing GRX480 protein, a member of the glutaredoxin family that regulates the redox status of proteins [1]. Treatment of two Symbiodinium dinoflagellate species (S. kawagutii clade F and Symbiodinium sp. clade A temperate) with 500 µM menadione bisulfite sodium for 1.5 hours in the dark induced a significant increase in intracellular ROS levels. Compared to controls, ROS increased 11.30-fold in S. kawagutii and 6.70-fold in the temperate clade A species. [1] The MSB-induced oxidative stress significantly reduced the maximal photochemical quantum yield (Fv/Fm) of Photosystem II by 49.96% in S. kawagutii and by 33.76% in the temperate clade A species. [1] MSB treatment decreased the total oxygen radical-scavenging capacity (assessed by a fluorescence-based assay) in S. kawagutii to 62.77% of control levels, while it remained near control levels (97.17%) in the temperate clade A species. [1] Total superoxide dismutase (SOD) activity decreased significantly in both species upon MSB treatment, reaching 69.74% and 68.78% of control levels for S. kawagutii and the temperate clade A species, respectively. [1] MSB treatment caused a significant decrease in chlorophyll a and c₂ concentrations, and altered carotenoid levels (diatoxanthin, peridinin, diadinoxanthin) in both species, with S. kawagutii generally showing greater reductions. [1] The treatment increased protein ubiquitination (6.42 and 11.67-fold increase vs. control for temperate clade A and S. kawagutii, respectively) and malondialdehyde (MDA) content (a marker of lipid peroxidation) to 133.87% of control in S. kawagutii, but only to 108.01% in the temperate clade A species. Protein carbonylation was not significantly affected. [1] Caspase-like activity (measured with Ac-DEVD-AFC substrate) decreased twofold in S. kawagutii after MSB treatment but remained at control levels in the temperate clade A species. [1] |
| Enzyme Assay |
Superoxide Dismutase (SOD) Activity Assay: Total SOD activity was determined using a colorimetric microplate assay. The assay mixture contained phosphate buffer (pH 8.0), diethylenetriamine-pentaacetic acid, hypoxanthine, the water-soluble tetrazolium salt WST-1, catalase, and xanthine oxidase. Xanthine oxidase and hypoxanthine generate superoxide anions (O₂⁺•), which convert WST-1 to a colored WST-1 formazan product, measurable at 450 nm. SOD activity in cell extracts competitively inhibits this reaction. Cell extracts or a bovine Cu,Zn-SOD standard were added to the wells, and the rate of absorbance increase at 450 nm was monitored for 15 minutes at 25°C. Activity was calculated based on the amount of sample causing 50% inhibition of formazan formation. [1] Oxygen Radical-Scavenging Capacity Assay: This fluorometric assay evaluates the overall capacity of water-soluble cellular antioxidants to neutralize peroxyl radicals. Cell extracts or a Trolox standard were incubated with phosphate buffer (pH 7.0), the fluorescent probe 6-carboxyfluorescein, and the peroxyl radical generator AAPH (2,2'-azobis(2-amidinopropane) dihydrochloride). The fluorescence decay at 520/495 nm (emission/excitation) was recorded every minute for 60 minutes. The antioxidant capacity of samples was determined by comparison to a Trolox standard curve and expressed as Trolox equivalents. [1] |
| Cell Assay |
Induction of Oxidative Stress and ROS Measurement: Cultures of Symbiodinium cells were exposed to 500 µM menadione bisulfite sodium in the dark for 1.5 hours at 26°C to induce an acute oxidative burst. Control cultures were incubated under identical conditions without MSB. [1] Intracellular ROS Detection: ROS production was assessed using the fluorescent probe carboxy-H₂DCFDA. Cells were incubated with 5 µM carboxy-H₂DCFDA for 30 minutes in the dark. After washing, they were exposed to MSB or control conditions for 1.5 hours. Fluorescence was measured in black 96-well plates using a spectrofluorometer (excitation/emission: 485/528 nm). [1] Chlorophyll Fluorescence Measurement: After treatment, cell aliquots were concentrated and dark-adapted. The maximum photochemical quantum yield of Photosystem II (Fv/Fm) was measured using a fluorescence imaging system after applying a saturating pulse of light. [1] Pigment Analysis: Pigments were extracted from cells with 100% methanol at 4°C for 24 hours. Chlorophyll a and c₂ concentrations were determined spectrophotometrically. Detailed carotenoid profiles (peridinin, diadinoxanthin, diatoxanthin, β-carotene) were analyzed by reverse-phase HPLC with photodiode array detection. [1] Protein Ubiquitination (Dot Blot): Proteins were extracted from cell pellets. For the assay, denatured proteins were fixed on a nitrocellulose membrane. The membrane was incubated with a primary anti-ubiquitin antibody, followed by a horseradish peroxidase-conjugated secondary antibody. Detection was performed using enhanced chemiluminescence and autoradiography. Spot density was quantified and normalized to total protein content revealed by Amido Black staining. [1] Protein Carbonylation (ELISA): Carbonyl content, an indicator of protein oxidation, was measured using an ELISA. Proteins were derivatized with dinitrophenylhydrazine (DNPH). The assay used a biotin-conjugated anti-DNP antibody and streptavidin-biotinylated horseradish peroxidase. After substrate incubation, absorbance was read at 492 nm, and carbonyl content was calculated using a molar absorption coefficient. [1] Lipid Peroxidation (TBARS Assay): Lipid peroxidation was evaluated by measuring thiobarbituric acid reactive substances (TBARS), primarily malondialdehyde (MDA). Cell extracts were reacted with a TBA reagent containing thiobarbituric acid, butylated hydroxytoluene, and HCl. The concentration of the resulting red MDA-TBA adduct was determined spectrophotometrically at 535 nm using a specific extinction coefficient. [1] Caspase-like Activity Assay: Cells were lysed by sonication in an appropriate buffer. Cell extracts were incubated in a reaction buffer containing the fluorogenic caspase-3 substrate Ac-DEVD-AFC. The release of fluorescent AFC was measured every 3 minutes for 90 minutes in a black 96-well plate using a spectrofluorometer (excitation/emission: 400/505 nm). Activity was expressed as pmol AFC released per minute. [1] |
| Toxicity/Toxicokinetics |
The study demonstrates the in vitro toxicity of menadione bisulfite sodium (at 500 µM for 1.5 h) to Symbiodinium cells, manifested as oxidative stress, photosynthetic damage, pigment degradation, lipid peroxidation, protein damage (increased ubiquitination), and inhibition of caspase-like activity. The temperate Symbiodinium sp. (clade A) was more tolerant to this chemically-induced oxidative stress than the tropical S. kawagutii (clade F). [1] |
| References |
[1]. Differential antioxidant response between two Symbiodinium species from contrasting environments. Plant Cell Environ. 2016 Dec;39(12):2713-2724. |
| Additional Infomation |
Menadione sodium sulfonate is an organic sodium salt that is the monosodium salt of menadione sulfonate. A synthetic naphthoquinone without the isoprenoid side chain and biological activity, but can be converted into active vitamin K2, menaquinone, after alkylation in vivo. It contains a menadione sulfonate. It is functionally related to a menadione. A synthetic naphthoquinone without the isoprenoid side chain and biological activity, but can be converted to active vitamin K2, menaquinone, after alkylation in vivo. Menadione bisulfite sodium was used in this study as a chemical tool to induce an acute, endogenous oxidative stress in Symbiodinium cells, bypassing the effects of light or temperature. This allowed for a direct comparison of the antioxidant network responses between two species from contrasting environments (tropical vs. temperate). [1] The ROS (O₂⁺• and H₂O₂) generated by MSB are similar to those produced by the Mehler reaction in photosynthesis, which is a significant alternative electron sink in Symbiodinium and increases during thermal stress. [1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~362.00 mM) H2O : ≥ 100 mg/mL (~362.00 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (9.05 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (9.05 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (9.05 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 120 mg/mL (434.40 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 3.6200 mL | 18.1002 mL | 36.2004 mL | |
| 5 mM | 0.7240 mL | 3.6200 mL | 7.2401 mL | |
| 10 mM | 0.3620 mL | 1.8100 mL | 3.6200 mL |