Physicochemical Properties
| Molecular Formula | CH6O6P2 |
| Molecular Weight | 176.0023 |
| Exact Mass | 175.963 |
| CAS # | 1984-15-2 |
| Related CAS # | 25681-89-4 (di-hydrochloride salt);6145-29-5 (tetra-hydrochloride salt) |
| PubChem CID | 16124 |
| Appearance | White to off-white solid powder |
| Density | 2.1±0.1 g/cm3 |
| Boiling Point | 583.2±60.0 °C at 760 mmHg |
| Melting Point | 197-199 °C(lit.) |
| Flash Point | 306.5±32.9 °C |
| Vapour Pressure | 0.0±3.5 mmHg at 25°C |
| Index of Refraction | 1.550 |
| LogP | -4.11 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 6 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 9 |
| Complexity | 152 |
| Defined Atom Stereocenter Count | 0 |
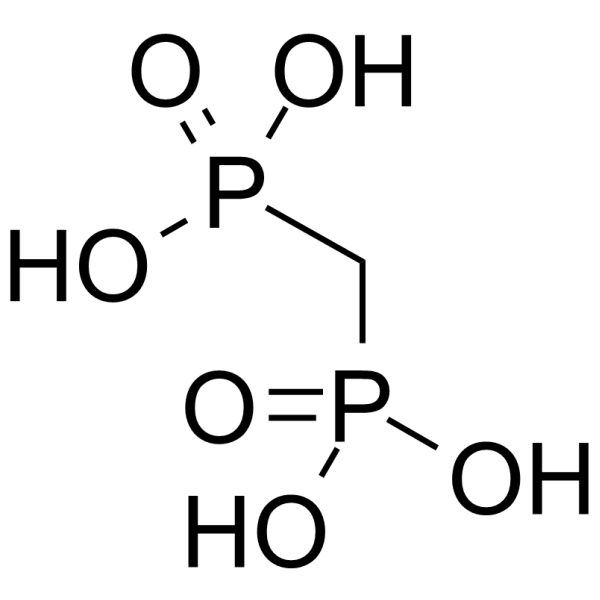
| SMILES | P(C([H])([H])P(=O)(O[H])O[H])(=O)(O[H])O[H] |
| InChi Key | MBKDYNNUVRNNRF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/CH6O6P2/c2-8(3,4)1-9(5,6)7/h1H2,(H2,2,3,4)(H2,5,6,7) |
| Chemical Name | phosphonomethylphosphonic acid |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Toxicity/Toxicokinetics |
Effects During Pregnancy and Lactation ◉ Summary of Use during Lactation Information in this record refers to the use of technetium Tc 99m medronate (Tc 99m methylene diphosphonate; Tc 99m MDP) as a diagnostic agent. A US Nuclear Regulatory Commission subcommittee has recommended that nursing be discontinued for 24 hours after administration of all technetium Tc 99m diagnostic products to simplify guidance recommendations, although this time interval may be longer than necessary.[1] The International Commission on Radiological Protection also recommends that breastfeeding need not be interrupted after administration technetium Tc 99m medronate.[2] However, to follow the principle of keeping exposure "as low as reasonably achievable", some experts recommend nursing the infant just before administration of the radiopharmaceutical and interrupting breastfeeding for 3 to 6 hours after the dose, then expressing the milk completely once and discarding it. If the mother has expressed and saved milk prior to the examination, she can feed it to the infant during the period of nursing interruption.[3-5] Mothers need not refrain from close contact with their infants after usual clinical doses.[6] Mothers concerned about the level of radioactivity in their milk could ask to have it tested at a nuclear medicine facility at their hospital. When the radioactivity is at a safe level, she may resume breastfeeding. A method for measuring milk radioactivity and determining the time when a mother can safely resume breastfeeding has been published.[7] For nursing mothers who work with Tc 99m substances in their workplace, there is no need to take any precautions other than those appropriate for general radiation protection.[8] ◉ Effects in Breastfed Infants Relevant published information was not found as of the revision date. ◉ Effects on Lactation and Breastmilk Relevant published information was not found as of the revision date. |
| References |
[1]. Salt Tolerance Enhancement of Liquid Chromatography-Matrix-Assisted Laser Desorption/Ionization-Mass Spectrometry Using Matrix Additive MethylenediphosphonicAcid. Mass Spectrom (Tokyo). 2014;3(1):A0031. |
| Additional Infomation |
Medronic acid is a 1,1-bis(phosphonic acid) consisting of methane substituted by two phosphonic acid groups. It has a role as a bone density conservation agent and a chelator. Medronic Acid is a methylene-substituted bisphosphonate. Medronic acid has affinity for and adheres to the surface of hydroxyapatite crystals in the bone matrix. This agent accumulates at sites of osteoid mineralization and can be used, complexed with radioisotopes such as technetium 99m, in bone imaging. See also: Medronate Disodium (has salt form); Medronate Tetrasodium (has salt form). 1. Medronic acid (also named methylenediphosphonic acid) is used as a matrix additive in liquid chromatography-matrix-assisted laser desorption/ionization-mass spectrometry (LC-MALDI-MS) to enhance the salt tolerance of the detection system[1] 2. The experimental process of applying Medronic acid in LC-MALDI-MS: First, prepare the matrix solution containing Medronic acid (as the additive) and the conventional MALDI matrix; then, after separating the analyte by liquid chromatography, mix the eluent with the prepared matrix solution; next, spot the mixed solution onto the MALDI target plate; finally, perform mass spectrometry detection. The addition of Medronic acid can effectively reduce the interference of salt ions (such as sodium and potassium ions) in the sample on the ionization efficiency of the analyte, thereby improving the detection sensitivity and signal-to-noise ratio of the target compound in the salt-containing sample[1] 3. In the salt tolerance test of the detection system, compared with the conventional MALDI matrix without Medronic acid, the LC-MALDI-MS system with Medronic acid as the matrix additive can maintain stable detection signals when the salt concentration in the sample is increased to 50 mM, while the signal of the conventional system is significantly attenuated or even disappears at a salt concentration of 10 mM[1] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~250 mg/mL (~1420.45 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (11.82 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (11.82 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (11.82 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.6818 mL | 28.4091 mL | 56.8182 mL | |
| 5 mM | 1.1364 mL | 5.6818 mL | 11.3636 mL | |
| 10 mM | 0.5682 mL | 2.8409 mL | 5.6818 mL |