Physicochemical Properties
| Molecular Formula | C11H21N |
| Molecular Weight | 167.294 |
| Exact Mass | 167.167 |
| CAS # | 60-40-2 |
| Related CAS # | Mecamylamine hydrochloride;826-39-1 |
| PubChem CID | 4032 |
| Appearance | Typically exists as solid at room temperature |
| Density | 0.91 |
| Boiling Point | 189.3ºC at 760 mmHg |
| Melting Point | 245-256ºC |
| Flash Point | 58.1ºC |
| Index of Refraction | 1.4875 |
| LogP | 2.811 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 1 |
| Rotatable Bond Count | 1 |
| Heavy Atom Count | 12 |
| Complexity | 197 |
| Defined Atom Stereocenter Count | 0 |
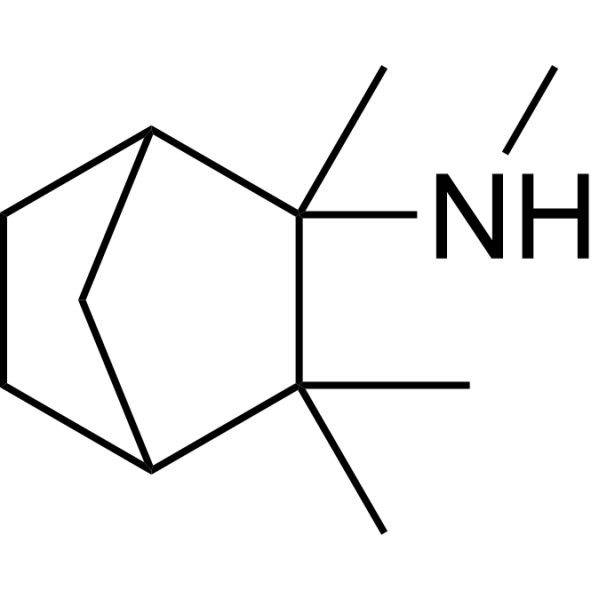
| SMILES | CC1(C)C2CCC(C2)C1(C)NC |
| InChi Key | IMYZQPCYWPFTAG-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C11H21N/c1-10(2)8-5-6-9(7-8)11(10,3)12-4/h8-9,12H,5-7H2,1-4H3 |
| Chemical Name | N,2,3,3-tetramethylbicyclo[2.2.1]heptan-2-amine |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ln Vitro | Mecamylamine (0.5-9 μM) in bath solution increases the frequency of firing in 5-HT DRN (dorsal raphe nucleus) neurons that have been identified [1]. Mecamylamine increases glutamatergic and decreases GABAergic input to 5-HT DRN neurons when administered in bath solution at concentrations of 0.5–9 μM [1]. In spiral cut strips of rabbit aorta, mecamylamine (1 mM, 5 min) inhibits histamine receptors and histamine-induced contraction [2]. Nicotine's neuroprotective effects are lessened by mecamylamine (10 μM, 48 h) [3]. Mecamylamine (1-100 nM, 30 min) in HDMVEC attenuates the formation of endothelial tubes in a dose-dependent manner [4]. |
| ln Vivo | Using a CNV mouse model, mecamylamine (subcutaneous pump, 50 mg/kg/day, 2 days) inhibits choroidal neovascularization (CNV) [4]. Dependent on β2 and α7 subtypes, mecamylamine (ip, 0.5-1 mg/kg) has antidepressant-like effects in the tail suspension test (TST) and forced swim test (FST) in C57BL/6J mice. base[5]. |
| Cell Assay |
Western Blot Analysis[3] Cell Types: SCG Neuronal Tested Concentrations: 10 μM Incubation Duration: 48 hrs (hours) Experimental Results: diminished nicotine-promoted increase in ERK1/2. |
| Animal Protocol |
Animal/Disease Models: Choroidal neovascularization (CNV) mouse model [1] Doses: 50 mg/kg/day, 2 days Route of Administration: subcutaneouspump implanted under the back skin, 200 μL, average pump speed 0.5 μL/h. Experimental Results: In the absence of nicotine, the development of CNV at the site of Bruch's membrane rupture was inhibited. Animal/Disease Models: C57BL/6J mice [5] Doses: 0.5-1 mg/kg Route of Administration: intraperitoneal (ip) injection Experimental Results: No effect on β2 gene knockout mice and α7 gene knockout mice, but diminished wild-type littermates Mouse immobility time in FST. |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Mecamylamine is almost completely absorbed from the gastrointestinal tract Mecamylamine is excreted slowly in the urine in the unchanged form. The rate of its renal elimination is influenced markedly by urinary pH. Alkalinization of the urine reduces, and acidification promotes, renal excretion of mecamylamine. Mecamylamine crosses the blood-brain and placental barriers. |
| Toxicity/Toxicokinetics |
Protein Binding 40% |
| References |
[1]. Mechanisms of stimulatory effects of mecamylamine on the dorsal raphe neurons. Brain Res Bull. 2020 Nov;164:289-298. [2]. The influence of mecamylamine on contractions induced by different agonists and on the role of calcium ions in the isolated rabbit aorta. J Pharmacol Exp Ther. 1976 Apr;197(1):57-65. [3]. Nicotine is neuroprotective to neonatal neurons of sympathetic ganglion in rat. Auton Neurosci. 2019 Jan;216:25-32. [4]. Mecamylamine suppresses Basal and nicotine-stimulated choroidal neovascularization. Invest Ophthalmol Vis Sci. 2008 Apr;49(4):1705-11. [5]. The nicotinic antagonist mecamylamine has antidepressant-like effects in wild-type but not beta2- or alpha7-nicotinic acetylcholine receptor subunit knockout mice. Psychopharmacology (Berl). 2006 Dec;189(3):395-401. |
| Additional Infomation |
Mecamylamine is a primary aliphatic amine. A nicotinic antagonist that is well absorbed from the gastrointestinal tract and crosses the blood-brain barrier. Mecamylamine has been used as a ganglionic blocker in treating hypertension, but, like most ganglionic blockers, is more often used now as a research tool. Mecamylamine has been reported in Wettsteinia inversa with data available. A nicotinic antagonist that is well absorbed from the gastrointestinal tract and crosses the blood-brain barrier. Mecamylamine has been used as a ganglionic blocker in treating hypertension, but, like most ganglionic blockers, is more often used now as a research tool. See also: Mecamylamine Hydrochloride (has salt form); Dexmecamylamine (narrower). Drug Indication For the treatment of moderately severe to severe essential hypertension and in uncomplicated cases of malignant hypertension Mechanism of Action Mecamylamine is a ganglionic blocker which prevents stimulation of postsynaptic receptors by acetylcholine released from presynaptic nerve endings. The hypotensive effect of Mecamylamine is attributed to reduction in sympathetic tone, vasodilation, and reduced cardiac output, and is primarily postural. Pharmacodynamics Mecamylamine is a potent, oral antihypertensive agent and ganglion blocker, and is a secondary amine. Mecamylamine is indicated for the management of moderately severe to severe essential hypertension and in uncomplicated cases of malignant hypertension. Mecamylamine reduces blood pressure in both normotensive and hypertensive individuals. A small oral dosage often produces a smooth and predictable reduction of blood pressure. Although this antihypertensive effect is predominantly orthostatic, the supine blood pressure is also significantly reduced. Mecamylamine crosses the blood-brain and placental barriers. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 5.9776 mL | 29.8882 mL | 59.7764 mL | |
| 5 mM | 1.1955 mL | 5.9776 mL | 11.9553 mL | |
| 10 mM | 0.5978 mL | 2.9888 mL | 5.9776 mL |