Physicochemical Properties
| Molecular Formula | C28H45NO7 |
| Molecular Weight | 507.66 |
| Exact Mass | 507.31960 |
| PubChem CID | 169450195 |
| Appearance | Typically exists as solid at room temperature |
| LogP | 2.9 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 10 |
| Heavy Atom Count | 36 |
| Complexity | 838 |
| Defined Atom Stereocenter Count | 8 |
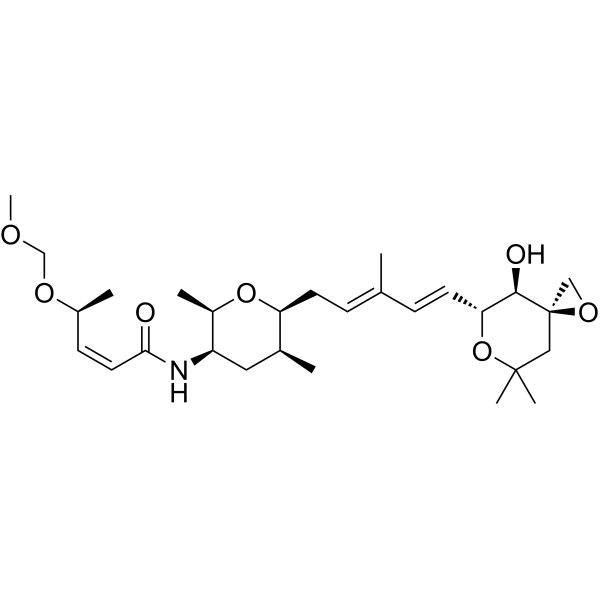
| SMILES | C[C@H]1C[C@H]([C@H](O[C@H]1C/C=C(\C)/C=C/[C@@H]2[C@H]([C@@]3(CC(O2)(C)C)CO3)O)C)NC(=O)/C=C\[C@H](C)OCOC |
| InChi Key | OGCGICPBGMPHIU-DNSRGATHSA-N |
| InChi Code | InChI=1S/C28H45NO7/c1-18(9-12-24-26(31)28(16-34-28)15-27(5,6)36-24)8-11-23-19(2)14-22(21(4)35-23)29-25(30)13-10-20(3)33-17-32-7/h8-10,12-13,19-24,26,31H,11,14-17H2,1-7H3,(H,29,30)/b12-9+,13-10-,18-8+/t19-,20-,21+,22+,23-,24+,26+,28+/m0/s1 |
| Chemical Name | (Z,4S)-N-[(2R,3R,5S,6S)-6-[(2E,4E)-5-[(3R,4R,5R)-4-hydroxy-7,7-dimethyl-1,6-dioxaspiro[2.5]octan-5-yl]-3-methylpenta-2,4-dienyl]-2,5-dimethyloxan-3-yl]-4-(methoxymethoxy)pent-2-enamide |
| Synonyms | Meayamycin D; RNA splicing modulator Meayamycin D; (S,Z)-N-((2R,3R,5S,6S)-6-((2E,4E)-5-((3R,4R,5R)-4-Hydroxy-7,7-dimethyl-1,6-dioxaspiro[2.5]octan-5-yl)-3-methylpenta-2,4-dien-1-yl)-2,5-dimethyltetrahydro-2H-pyran-3-yl)-4-(methoxymethoxy)pent-2-enamide |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Natural product |
| ln Vitro | FR901464 is a cytotoxic natural product that binds splicing factor 3B subunit 1 (SF3B1) and PHD finger protein 5A (PHF5A), the components of the human spliceosome. The amide-containing tetrahydropyran ring binds SF3B1, and it remains unclear how the substituents on the ring contribute to the binding. Here, we synthesized meayamycin D, an analogue of FR901464, and three additional analogues to probe the conformation through methyl scanning. We discovered that the amide-containing tetrahydropyran ring assumes only one of the two possible chair conformations and that methylation of the nitrogen distorts the chair form, dramatically reducing cytotoxicity. Meayamycin D induced alternative splicing of MCL-1, showed strong synergism with venetoclax in drug-resistant lung cancer cells, and was cancer-specific over normal cells. Meayamycin D incorporates an alkyl ether and shows a long half-life in mouse plasma. The characteristics of meayamycin D may provide an approach to designing other bioactive L-shaped molecules [1]. |
| Enzyme Assay |
In Vitro Plasma Stability [1] Mouse CD1 plasma K2 EDTA was prepared in a 2 mL microcentrifuge tube. The compounds were prepared separately as 1 mM solutions in 10% DMSO and added to the plasma as 100× dilutions to give 700 μL at a concentration of compound, 10 μM procaine, meayamycin (meayamycin A), meayamycin D, or meayamycin E, in 0.1% DMSO. The mixture was vortexed for 10 s, capped, and placed in a shaking incubator for 48 h at 125 rpm at 37 °C. At the indicated times an aliquot (70 μL) of the mixture was taken and added to an equal volume of ice-cold MeCN and centrifuged for 15 min at 14,000 relative centrifugal force (rcf) at 4 °C. The supernatant was collected and frozen at −80 °C until sample analysis. The samples were analyzed by LC-MS using Fmoc-l-phenylalanine as an internal standard at a concentration of 25 μM. A standard curve was prepared separately in a matrix-matched solution by 2-fold serial dilution from 10 μM. The decomposition was determined by comparing the ratio of analyte to Fmoc-l-phenylalanine with the ratio of the analyte to Fmoc-l-phenylalanine in the first data point. |
| Cell Assay |
Growth Inhibition Assay [1] All cell lines were maintained in RPMI-1640 media, Waymouth media (DMS53 and DMS114 cells), or F-12K media (FL83B cells) + 10% fetal bovine serum. The cells were mycoplasma-free as determined by the e-Myco PLUS mycoplasma PCR detection kit. The cells were plated in 96-well plates at an initial density of 1500 or 5000 cells per well in culture media (100 μL) and were incubated for 24 h prior to compound addition. The compounds were prepared separately as 10 mM in 100% DMSO or 10 μM in 100% DMSO. Serial dilution in sterile water gave 10× dilutions that were added directly to the cells as 100× dilutions to give the desired concentration of compound, 0.1 nM to 30 μM meayamycin A, meayamycin D, N-methyl meayamycin D, meayamycin E, or N-methyl meayamycin E, in 0.1–0.3% DMSO. The cells were then incubated for additional 72 h. Cell proliferation was measured by using the commercial 3-(4,5-dimethylthiazol-2-yl)-5-(3-carboxymethoxyphenyl)-2-(4-sulfophenyl)-2H-tetrazolium (MTS) dye or 4-[3-(4-iodophenyl)-2-(4-nitrophenyl)-2H-5-tetrazolio]-1,3-benzenesulfonate (WST-1) dye reduction assay. The absorbance at 490 nm (MTS) or 450 nm (WST-1) was measured with a Modulus II Microplate Multimode Reader or Tecan Infinite M1000 PRO Multimode Reader Evaluation of the compounds was performed in duplicate at each concentration. GraphPad Prism 9.4.0 was used to construct dose–response curves and calculate the GI50 values. Immunoblot Analysis [1] Cells were treated with various concentrations of meayamycin analogues for 8 h and then lysed using RIPA buffer (10 mM Tris, pH 7.5, 150 mM NaCl, 1 mM EDTA, 0.1% SDS, 1% IGEPAL, 0.5% sodium deoxycholate) containing phosphatase and protease inhibitors. Approximately 20 μg protein from each cell lysate was resolved on SDS-PAGE gels . Proteins were transferred onto nitrocellulose membranes followed by 1 h incubation at room temperature in blocking solution (1× TBS, 0.1% Tween-20, 5% milk powder). Membranes were incubated overnight at 4 °C with the following antibodies: antiphospho-SF3B1, anti-SF3B1, anti-MCL-1, anti-p27, anti-p21, anti-p53 , and anti-α-tubulin. Proteins were detected using SuperSignal West Pico substrate. Combination Index Analysis [1] Cells were plated in 96-well plates at an initial density of 1500 or 5000 cells per well in RPMI-1640 medium (100 μL) and were incubated for 24 h prior to compound addition. The compounds were prepared separately as 10 mM in 100% DMSO or 10 μM in 100% DMSO. Serial dilution in sterile water gave 10× dilutions that were added directly to the cells alone or in combinations as 100× dilutions to give the desired concentration of compound: 0.5–3 nM meayamycin D, 1.25–10 μM S63845, 25 nM to 8 μM venetoclax, and 0.25–4 μM osimertinib. The cells were then incubated for additional 72 h. Cell proliferation was measured by using the commercial WST-1 dye reduction assay. To determine whether drug combinations were synergistic, we used CompuSyn software to calculate the combination index (CI) using nonconstant drug ratios.48 A CI of less than 1.0 was considered to be indicative of synergism, and this interaction was further classified as strong synergism (CI < 0.3), synergism (CI of 0.3–0.7), and slight to moderate synergism (CI of 0.7–0.9). |
| References | [1]. Synthesis and Conformational Analysis of FR901464-Based RNA Splicing Modulators and Their Synergism in Drug-Resistant Cancers. J Med Chem. 2023 Oct 23;66(21):14497–14512. |
| Additional Infomation | In conclusion, we designed and synthesized four analogues to investigate the six-membered ring conformation of meayamycin. We acknowledge that the Pena group published the cocrystal structure of spliceostatin A-SF3B1-PHF5A during this study; however, their paper was not available during the period of the current study for the design of these analogues. Furthermore, our results substantiate the finding that the bound conformation of spliceostatin occurs in the N-axial conformation. Our results suggest that N-methylation induces twisting of the ring conformation and removing the C12 methyl group from N-methyl meayamycin D does not restore potency, highlighting a necessity for the N–H bond. Conformational analysis suggests that the amide-containing pyran of the natural product exists as a single conformer with the C14–N bond in the axial position. Additionally, structural information from the crystal structure provides insight into the reduced potency of the N-methyl analogues because of their deviated conformations. These results provide evidence for the N-axial chair conformation as the binding conformation and suggest that the amide N–H is essential for both the ring conformation and intracellular modulation of alternative splicing. Two new compounds emerge from this study highlighting linear alkyl ethers as suitable substitutions at the C4′ position while also revealing metabolic labilities of other FR901464 analogues. These new compounds exhibit potency comparable to meayamycin A and improved metabolic stability, the series of which we have named meayamycin D and meayamycin E. Meayamycin D and meayamycin E show modest but promising cancer selectivity against colon cancer cell lines, the generality of which must be further explored. Finally, the 1,2,4,5-all cis substituted tetrahydropyran with a secondary amide at the 4-position may be an L-shaped scaffold that medicinal chemists sought after to present substituents at the 1 and 4 positions with rigidity. |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 1.9698 mL | 9.8491 mL | 19.6982 mL | |
| 5 mM | 0.3940 mL | 1.9698 mL | 3.9396 mL | |
| 10 mM | 0.1970 mL | 0.9849 mL | 1.9698 mL |