Physicochemical Properties
| Molecular Formula | C6H6O3 |
| Molecular Weight | 126.1100 |
| Exact Mass | 126.031 |
| CAS # | 118-71-8 |
| PubChem CID | 8369 |
| Appearance | White to off-white solid powder |
| Density | 1.3±0.1 g/cm3 |
| Boiling Point | 284.7±40.0 °C at 760 mmHg |
| Melting Point | 160-164 °C(lit.) |
| Flash Point | 127.3±20.8 °C |
| Vapour Pressure | 0.0±1.3 mmHg at 25°C |
| Index of Refraction | 1.561 |
| LogP | 0.08 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 3 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 9 |
| Complexity | 200 |
| Defined Atom Stereocenter Count | 0 |
| InChi Key | XPCTZQVDEJYUGT-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C6H6O3/c1-4-6(8)5(7)2-3-9-4/h2-3,8H,1H3 |
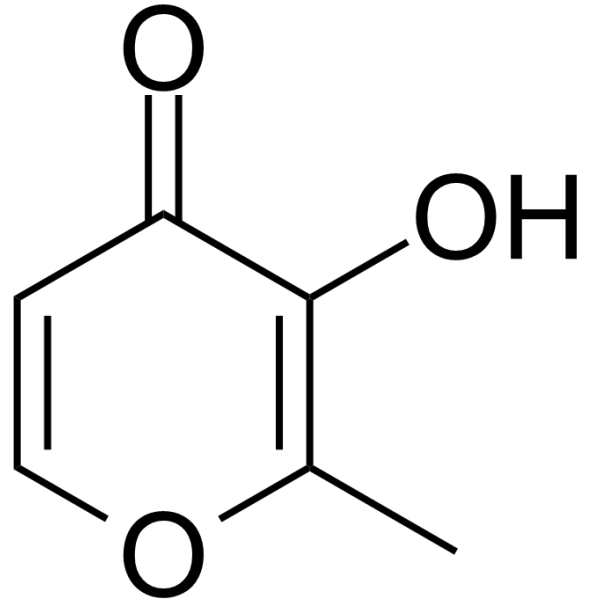
| Chemical Name | 3-hydroxy-2-methylpyran-4-one |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
The literature primarily investigates Maltol's pharmacological effects in the context of diabetic peripheral neuropathy (DPN). It describes antioxidative activity and effects on Na+-K+-ATPase. The mechanisms explored involve modulation of oxidative stress markers and apoptosis-related proteins.[1] |
| ln Vitro |
In RSC96 rat Schwann cells injured by H2O2 (0.6 mM), co-incubation with Maltol at concentrations of 0.1 and 0.5 mM significantly increased cell viability. It also elevated the expression of the anti-apoptotic protein Bcl-XL in a dose-dependent manner and decreased the expression of pro-apoptotic genes BAX and caspase-3.[1] |
| ln Vivo |
In streptozotocin (STZ)-induced diabetic rats, daily oral administration of Maltol at doses of 50, 100, and 200 mg/kg for 12 weeks significantly improved motor nerve conduction velocity (MNCV) by up to 58.2% at 200 mg/kg. It ameliorated thermal and mechanical hyperalgesia, increasing withdrawal thresholds and latencies. Maltol treatment elevated the activity of Na+-K+-ATPase in erythrocytes and sciatic nerves, increased serum levels of total antioxidant capacity (TAOC), glutathione (GSH), and superoxide dismutase (SOD), while decreasing malondialdehyde (MDA). It also modulated apoptosis-related proteins in the sciatic nerve (decreasing BAX and caspase-3, increasing BAG4 and Bcl-XL) and produced a decreasing trend (though not statistically significant) in caspase-3 activity in dorsal root ganglia. Maltol did not significantly affect fasting blood glucose levels or body weight.[1] |
| Cell Assay |
The cell viability assay (CCK-8) was performed on RSC96 cells. Cells were seeded in a 96-well plate and cultured overnight. After treatment with Maltol and/or H2O2 for 6 hours, CCK-8 reagent was added and incubated for another 3 hours. Absorbance was measured at 450 nm to determine cell viability.[1] The expression of apoptosis-related proteins (Bcl-XL) and genes (BAX, caspase-3) in RSC96 cells was assessed. For Western blot, total protein was extracted from cells, quantified, separated by SDS-PAGE, transferred to a PVDF membrane, blocked, and incubated with primary antibodies (e.g., Bcl-XL) and HRP-conjugated secondary antibodies.[1] For quantitative real-time PCR, total RNA was extracted from cells using TRIzol reagent, cDNA was synthesized, and PCR was performed using SYBR Green Master Mix and specific primers for BAX and caspase-3, with GAPDH as an internal standard.[1] |
| Animal Protocol |
Diabetic peripheral neuropathy was induced in male Sprague-Dawley rats by a single intraperitoneal injection of STZ (65 mg/kg) dissolved in citrate buffer (0.1 mol/L, pH 4.5). Diabetic rats (fasting blood glucose ≥ 11.1 mmol/L) were selected 4 weeks post-induction and randomly divided into groups (n=12). Treatment began 4 weeks after diabetes confirmation and lasted for 12 weeks. Maltol was dissolved in 0.5% carboxymethyl cellulose sodium (CMC-Na) solution and administered daily by oral gavage at doses of 50, 100, and 200 mg/kg. Normal and diabetic control groups received the 0.5% CMC-Na vehicle. Body weight and fasting blood glucose were monitored periodically. At the end of the 12-week treatment, assessments included motor nerve conduction velocity (MNCV), thermal and mechanical pain thresholds, collection of blood and tissues (sciatic nerve, dorsal root ganglia) for biochemical (Na+-K+-ATPase, oxidative stress markers), molecular (Western blot, qPCR), and enzymatic (caspase-3 activity) analyses.[1] |
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Groups of two beagle dogs of each sex were given a single intravenous injection of 10 mg/kg bw maltol, and urine samples were collected for 72 hr. An average of 58.5% of the administered dose was excreted as a mixture of sulfate and glucuronic acid conjugates of maltol. About 98% of the total urinary excretion of conjugates occurred within the first 24 hr, males and females excreting an average of 42% and 73% of the administered dose, respectively. Metabolism / Metabolites Maltol and derivatives contain a gamma-pyrone ring system. Gamma-pyrones are relatively basic, and the behavior as a base is partly due to the aromatic character and relative stability of the conjugate acid. As the gamma-pyrone ring also contains a 3-hydroxy substituent, it is expected that maltol and its derivatives will be readily conjugated with glucuronic acid or sulfate. In addition, maltol may form a complex with metal ions (e.g. Fe++), like phenols. |
| Toxicity/Toxicokinetics |
Toxicity Summary IDENTIFICATION AND USE: Maltol is a white crystalline powder. It is used as a flavoring agent, to impart \"freshly baked\" odor and flavor to breads and cakes. It is also used as medication. HUMAN EXPOSURE AND TOXICITY: Maltol at concentrations ranging from 0.1 to 1.5 umol/mL induced sister chromatid exchanges in human lymphocytes. It was suggested that these results were due to an indirect action of maltol and not to its direct reactivity with DNA. ANIMAL STUDIES: Eight female mice were fed a diet containing maltol at a level of 0.5% (w/w) for 21 weeks, calculated to provide an average daily intake of 750 mg/kg bw. At termination, no differences in general health, behavior, body-weight gain or relative liver weights were reported. Gross and microscopic examination revealed no histological abnormalities in the livers of the treated mice when compared with the controls. A three-generation study of reproductive toxicity was conducted, in which groups of 20 male and 20 female rats were given diets containing maltol at concentrations resulting in 100, 200 or 400 mg/kg bw per day. On day 134, animals of the F1 generation showed signs of sialodacryodenitis due to a contagious virus. No deaths occurred, and the signs diminished within 10 days. Maltol had no effect on copulation rate, mating viability index, lactation, offspring sex ratio or 21-day pup survival index. Maltol at concentrations ranging from 0.1 to 1.5 umol/mL induced sister chromatid exchanges in Chinese hamster ovary cells. It was suggested that these results were due to an indirect action of maltol and not to its direct reactivity with DNA. Maltol was weakly mutagenic (two- to threefold increases in number of revertants) in Salmonella typhimurium TA100 at concentrations of 1-3 mg/plate either alone or with metabolic activation. Activity against TA98 was not detected. Maltol tested at concentrations of 0.1-10.0 mg/plate increased the number of revertants in strain TA97 at 1 mg/plate by about twofold. No increase found in the presence of an activation system, or in TA102 alone or with activation. In other studies with S. typhimurium, maltol was not consistently mutagenic when tested at concentrations up to 10,000 ug/plate alone or in the presence of an activation system. Interactions The pyrones, 3-hydroxy-2-methyl-4-pyrone (maltol) and 3-hydroxy-2-ethyl-4-pyrone (ethyl maltol) chelate iron with a high affinity and selectivity. The resulting 1:3 (metal-ligand) complexes, being neutral, are able to partition readily across cell membranes and thus may facilitate iron transport across the intestinal wall. Absorption of radioactive iron ((59)Fe) in the presence of these pyrones was investigated in male rats 1, 2, 4 and 6 hr after intraduodenal administration of a 7 micrograms dose and compared with that of (59)Fe given as the sulfate, gluconate, fumarate or complexed to EDTA. Total body absorption and distribution were calculated from the (59)Fe content of various tissue samples. With all the iron preparations used, blood levels of (59)Fe were highest 1 hr after injection whilst the (59)Fe content at the major site of deposition, i.e. the bone marrow, increased up to 6 hr. No (59)Fe was found in the urine. Total body absorption of (59)Fe was significantly higher from the pyrones than from the other four preparations. Over the dose range 0.7-700 micrograms, the proportion of (59)Fe absorbed from both iron maltol and iron sulphate decreased with increasing dose. Enhanced (59)Fe uptake from maltol was evident at 0.7-70 micrograms but not at 700 micrograms suggesting that use of these pyrones will not result in iron overload. Absorption of (59)Fe given into the stomach was slower in onset but was sustained longer presumably via a steady delivery of iron to the duodenum from the gastric reservoir. Neurofilamentous tangles have been induced in cultured neurons from rat brain hemispheres by application of both aluminum and maltol. Quantitative evaluation revealed a significantly higher percentage of tangle containing neurons when using the aluminum-maltol mixture than after application of aluminum alone. Tangles were found to be consistently stained with monoclonal antibodies to neurofilament proteins but failed to react with polyclonal antibodies against microtubule-associated proteins 1, 2 and tau. Aluminum (Al) has been observed to cause neurofilament protein accumulation in both experimental animals and cultured cells. Impairment of axonal transport is thought to be a mechanism of toxicity. Inhibition of the degradation of neurofilament proteins, however, resulting in accumulation of these proteins may be an alternative mechanism for Al toxicity. In the present study, the effect of calcium (Ca) on the proteolysis of the neurofilament triplet proteins by calcium-activated neutral proteases (CANP) was studied in the isolated sciatic nerve explants. The extent of the degradation was found to be dependent on the Ca concentration. The effect of Al chloride, -citrate and -maltol on the calcium-induced degradation was studied. No effect of any of the Al compounds was observed, suggesting that the metal may exert its neurotoxic effect via a mechanism other than impairment of neurofilament proteolysis. Maltol itself was found to enhance the effect of Ca on the degradation of neurofilament proteins, probably by facilitating the movement of Ca across the neuronal membrane. Deposition of aluminum in the body is responsible for the development of dialysis-related diseases in patients with renal dysfunction and may play a role in the development of certain neurodegenerative disorders. Although citric acid is known to be a strong enhancer of gastrointestinal absorption of aluminum, its effect on aluminum distribution and accumulation is not yet clear. Maltol has been shown to increase the neurotoxicity of aluminum, but little is known about its effect on aluminum deposition in the body. To elucidate the role of citric acid and maltol in aluminum accumulation and toxicity, rats were loaded intraperitoneally during a 7-day period with different amounts of aluminum chloride in absence or presence of citric acid or maltol before analysis of aluminum in serum, brain, bone, and urine. Coadministration of citric acid led to relatively reduced serum levels, as compared with aluminum and aluminum-maltol treatment. This is explained by both tissue elimination and enhanced renal elimination. ... Maltol was shown to be a strong enhancer of aluminum accumulation in serum, brain, and bone. The rise of aluminum in these target tissues was dose dependent. For more Interactions (Complete) data for Maltol (7 total), please visit the HSDB record page. Non-Human Toxicity Values LD50 Chicken oral 3720 mg/kg LD50 Guinea pig oral 1410 mg/kg LD50 Rabbit oral 1620 mg/kg LD50 Mouse sc 820 mg/kg For more Non-Human Toxicity Values (Complete) data for Maltol (6 total), please visit the HSDB record page. The in vivo study reported that repeated oral administration for 12 weeks did not significantly affect body weight, suggesting no overt systemic toxicity at the tested doses (50-200 mg/kg) in this model. The in vitro study noted that at concentrations of 0.1 and 0.5 mM, Maltol protected RSC96 cells from H2O2-induced injury, but the literature also mentions (in the discussion) that other studies have reported cytotoxic/apoptotic effects of Maltol at higher concentrations (e.g., 1.2 mM) or in different cell types, indicating that its effects may be concentration and cell type-dependent.[1] |
| References |
[1]. Maltol, a food flavor enhancer, attenuates diabetic peripheral neuropathy in streptozotocin-induceddiabetic rats. Food Funct. 2018 Dec 13;9(12):6287-6297. |
| Additional Infomation |
Maltol is a white crystalline powder with a fragrant caramel-butterscotch odor. pH (5% aqueous solution) 5.3. (NTP, 1992) 3-hydroxy-2-methyl-4-pyrone is a member of 4-pyranones. It has a role as a metabolite. Maltol has been reported in Bolbostemma paniculatum, Streptomyces, and other organisms with data available. 3-Hydroxy-2-methyl-4-pyrone is a metabolite found in or produced by Saccharomyces cerevisiae. Mechanism of Action Maltol (3-hydroxy-2-methyl-4-pyrone) produced reactive oxygen species as a complex with transition metals. Maltol/iron complex inactivated aconitase the most sensitive enzyme to oxidative stress. The inactivation of aconitase was iron-dependent, and prevented by TEMPOL, a scavenger of reactive oxygen species, suggesting that the maltol/iron-mediated generation of superoxide anion is responsible for the inactivation of aconitase. Addition of maltol effectively enhanced the ascorbate/copper-mediated formation of 8-hydroxy-2'-deoxyguanosine in DNA. Oxidation of ascorbic acid by CuSO(4) was effectively stimulated by addition of maltol, and the enhanced oxidation rate was markedly inhibited by the addition of catalase and superoxide dismutase. These results suggest that maltol can stimulate the copper reduction coupled with the oxidation of ascorbate, resulting in the production of superoxide radical which in turn converts to hydrogen peroxide and hydroxyl radical. Cytotoxic effect of maltol can be explained by its prooxidant properties: maltol/transition metal complex generates reactive oxygen species causing the inactivation of aconitase and the production of hydroxyl radical causing the formation of DNA base adduct. ... We examined the ability of maltol to induce the cytochrome P450 1a1 (Cyp1a1), an enzyme known to play an important role in the chemical activation of xenobiotics to carcinogenic derivatives. Our results showed that treatment of Hepa 1c1c7 cells with maltol significantly induced Cyp1a1 at mRNA, protein, and activity levels in a concentration-dependent manner. The RNA synthesis inhibitor, actinomycin D, completely blocked the Cyp1a1 mRNA induction by maltol, indicating a requirement of de novo RNA synthesis through transcriptional activation. In addition, maltol induced aryl hydrocarbon receptor (AhR)-dependent luciferase reporter gene expression in stably transfected H1L1.1c2 cells, suggesting an AhR-dependent mechanism. This is the first demonstration that the food flavoring agent, maltol, can directly induce Cyp1a1 gene expression in an AhR-dependent manner and represents a novel mechanism by which maltol promotes carcinogenicity and toxicity. Maltol has antioxidant properties, presumably through its ability to complex metal ions such as Fe++ and to promote the formation of reduced glutathione (GSH). Maltol at a concentration of 130 umol/L inhibited iron-mediated lipid peroxidation and increased scavenging of reactive oxygen species by enhancing the supply of NADPH required for regeneration of GSH. Maltol inhibited the formation of thiobarbituric acid-reactive substances when incubated with rat liver microsomes in the presence of Fe++ and ascorbate. Maltol at concentrations of 130-140 umol/L also effectively inhibited the inactivation of NADP-isocitrate dehydrogenase, the principal NADPH-generating enzyme, by Fe++. Maltol significantly increased the oxidation of Fe++, while dimethylpyrone had no effect. The latter results suggest that the 3-hydroxy substituent in maltol is necessary to promote Fe++ oxidation. Therapeutic Uses /EXPL THER/ /The objective of the study was/ to evaluate the neuroprotective and neurite outgrowth effects of maltol, a natural aroma compound, on retinal ganglion cells (RGCs) under oxidative stress in vitro. Mouse primary RGCs were isolated using immunopanning-magnetic separation and exposed to H2O2 in the presence of maltol. The cell viability and apoptosis were determined by using adenosine 5'-triphosphate (ATP) assay and terminal deoxynucleotidyl transferase (TdT)-mediated deoxyuridine triphosphate (dUTP) nick end labeling (TUNEL), respectively. Neurite outgrowth was assessed by immunofluorescence for alpha-tubulin. The activation of nuclear factor-kappaB (NF-kappaB) was also evaluated using immunofluorescence. When the RGCs were exposed to 20 uM of H2O2 for 16 hr, their viability dropped to 40.3 +/- 3.4%. However, the maltol treatment restored the cells in a dose-dependent manner. The viability recovered to 73.9 +/- 5.1% with 10 uM of maltol and even reached 175.1 +/- 11.3% with 2 mM of maltol, as measured by ATP assay. This oxidative stress significantly increased the number of TUNEL-positive RGCs, but the maltol drastically reduced the proportion of those apoptotic cells. The oxidative stress hampered the neurite outgrowth of the RGCs, whereas maltol restored their ability to sprout neurites. Regarding NF-kappaB, the active form of phosphorylated NF-kappaB (pNF-kappaB) increased the oxidative stress level but the maltol treatment again reduced it to an unstressful level. Our data revealed that maltol attenuated the oxidative stress-induced injury in the primary mouse RGCs. Its neuroprotective and neurite outgrowth effects seemed to be related to NF-kappaB signaling. Maltol has potential as a new neuroprotective therapeutic agent for oxidative stress-related ocular diseases, including glaucoma. Maltol (3-hydroxy-2-methyl-4-pyrone) is a food flavor enhancer and preservative with known antioxidative properties. This study is the first to report its protective effects against diabetic peripheral neuropathy (DPN) in an STZ-induced rat model. The proposed mechanism of action involves its antioxidant activity (scavenging free radicals, improving oxidative stress markers) and anti-apoptotic effects (modulating Bcl-2 family proteins and caspase-3) in peripheral nerves and Schwann cells. The benefits were independent of blood glucose control. The study suggests Maltol as a potential novel therapeutic candidate for DPN.[1] |
Solubility Data
| Solubility (In Vitro) |
DMSO : ~100 mg/mL (~792.96 mM) H2O : ~1.82 mg/mL (~14.43 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (19.82 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (19.82 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (19.82 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. Solubility in Formulation 4: 3.33 mg/mL (26.41 mM) in PBS (add these co-solvents sequentially from left to right, and one by one), clear solution; with ultrasonication (<60°C). (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 7.9296 mL | 39.6479 mL | 79.2959 mL | |
| 5 mM | 1.5859 mL | 7.9296 mL | 15.8592 mL | |
| 10 mM | 0.7930 mL | 3.9648 mL | 7.9296 mL |