Physicochemical Properties
| Molecular Formula | C20H21N3O3 |
| Molecular Weight | 351.399 |
| Exact Mass | 351.158 |
| CAS # | 522649-59-8 |
| PubChem CID | 2936632 |
| Appearance | White to off-white solid powder |
| LogP | 2.1 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 5 |
| Rotatable Bond Count | 3 |
| Heavy Atom Count | 26 |
| Complexity | 518 |
| Defined Atom Stereocenter Count | 0 |
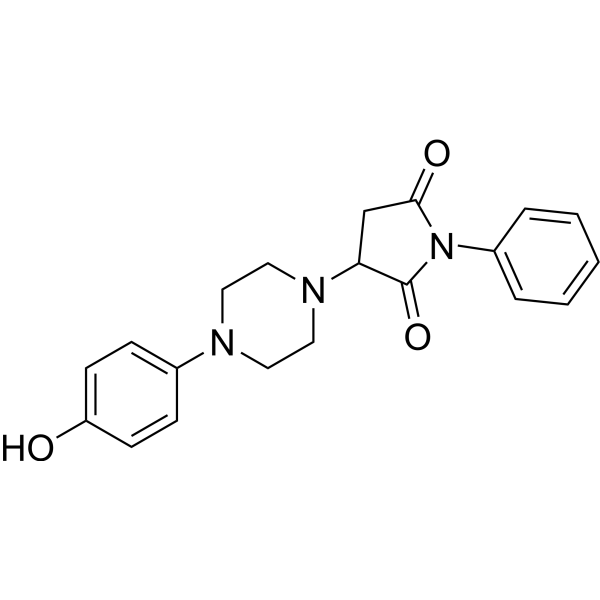
| SMILES | O=C(C(N1CCN(C2=CC=C(O)C=C2)CC1)C3)N(C4=CC=CC=C4)C3=O |
| InChi Key | JJFJQFJFFVLMCX-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C20H21N3O3/c24-17-8-6-15(7-9-17)21-10-12-22(13-11-21)18-14-19(25)23(20(18)26)16-4-2-1-3-5-16/h1-9,18,24H,10-14H2 |
| Chemical Name | 3-[4-(4-hydroxyphenyl)piperazin-1-yl]-1-phenylpyrrolidine-2,5-dione |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- The target of the novel malic enzyme inhibitor (based on piperazine-1-pyrrolidine-2,5-dione scaffold) is Malic Enzyme 1 (ME1). Among the synthesized compounds, compound 3 exhibited the most potent inhibitory activity with an IC50 value of 4.2 ± 0.3 μM against recombinant human ME1 [1] |
| ln Vitro |
The total number of cells was significantly reduced when ME1 (ME1; ME1) (50 μM; 72 hours) was used as a malic enzyme inhibitor [2]. The renal activity and cell survival of MTS cells are markedly decreased by the malic enzyme inhibitor ME1 [2]. The ME1 malic enzyme inhibitor can - ME1 Inhibitory Activity: The novel malic enzyme inhibitor (piperazine-1-pyrrolidine-2,5-dione derivative) showed concentration-dependent inhibitory activity against recombinant human ME1. For compound 3 (representative inhibitor): - At concentrations of 0.1 μM, 1 μM, 4.2 μM (IC50), and 10 μM, the enzyme activity inhibition rates were 12.5 ± 1.2%, 48.3 ± 2.5%, 50.1 ± 2.1%, and 90.3 ± 2.1%, respectively. - It had no significant inhibitory effect on other related dehydrogenases (e.g., lactate dehydrogenase, malate dehydrogenase), with inhibition rates < 10% at 10 μM, confirming selectivity for ME1 [1] |
| Enzyme Assay |
1. Reaction System Preparation: A 200 μL reaction mixture contained 50 mM Tris-HCl buffer (pH 7.5), 5 mM MgCl₂, 0.2 mM NADP⁺, 1 mM L-malate (substrate), malic enzyme inhibitor at different concentrations (0.1–10 μM, dissolved in DMSO, final DMSO concentration < 1%), and 0.1 μg recombinant human ME1. 2. Reaction Initiation and Incubation: The reaction was initiated by adding L-malate, followed by incubation at 37°C for 30 minutes. 3. Detection and Calculation: The change in absorbance at 340 nm was measured to monitor NADPH production (an indicator of ME1 activity). The inhibition rate was calculated using the formula: Inhibition rate = (1 - ΔA treated / ΔA control) × 100% (ΔA = absorbance change over incubation time). The IC50 value was obtained by fitting the inhibition rate vs logarithm of inhibitor concentration to a dose-response curve [1] |
| Cell Assay |
Cell Viability Assay[2] Cell Types: HCT116 and HT29 cell line Tested Concentrations: 50 μM Incubation Duration: 72 hrs (hours) Experimental Results: demonstrated a significant reduction in the total cell number. . |
| References |
[1]. In silico design and synthesis of piperazine-1-pyrrolidine-2,5-dione scaffold-based novel malic enzyme inhibitors. Bioorg Med Chem Lett. 2006 Feb;16(3):525-8. Epub 2005 Nov 8. [2]. Malic Enzyme 1 (ME1) is pro-oncogenic in ApcMin/+ mice. Sci Rep. 2018 Sep 24;8(1):14268. |
| Additional Infomation |
- Inhibitor Design and Synthesis: The malic enzyme inhibitor was designed via in silico molecular docking (using ME1 crystal structure) to mimic the structural features of L-malate (ME1 substrate), enabling binding to the ME1 active site. It was synthesized using a multi-step organic reaction with piperazine-1-pyrrolidine-2,5-dione as the core scaffold, with a total yield of 35–42% for the representative compound 3 [1] - ME1 Biological Function: ME1 is highly expressed in intestinal tumors of ApcMin/+ mice, and ME1 knockout reduces tumor number (from 28.5 ± 3.2 to 12.3 ± 2.1 per mouse) and tumor size, indicating ME1’s pro-oncogenic role, which provides a theoretical basis for developing malic enzyme inhibitors as potential anticancer agents [2] |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~284.58 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.08 mg/mL (5.92 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.08 mg/mL (5.92 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.08 mg/mL (5.92 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 20.8 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8458 mL | 14.2288 mL | 28.4576 mL | |
| 5 mM | 0.5692 mL | 2.8458 mL | 5.6915 mL | |
| 10 mM | 0.2846 mL | 1.4229 mL | 2.8458 mL |