Physicochemical Properties
| Molecular Formula | C32H53N7O11 |
| Exact Mass | 711.38 |
| CAS # | 1638156-31-6 |
| PubChem CID | 137444673 |
| Appearance | White to off-white solid powder |
| LogP | -2.7 |
| Hydrogen Bond Donor Count | 6 |
| Hydrogen Bond Acceptor Count | 11 |
| Rotatable Bond Count | 27 |
| Heavy Atom Count | 50 |
| Complexity | 1160 |
| Defined Atom Stereocenter Count | 0 |
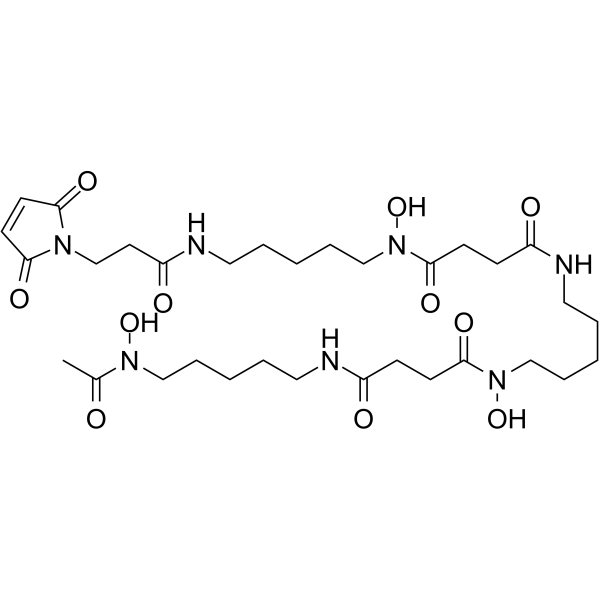
| SMILES | C(N(CCCCCNC(=O)CCN1C(=O)C=CC1=O)O)(=O)CCC(NCCCCCN(C(=O)CCC(NCCCCCN(C(C)=O)O)=O)O)=O |
| InChi Key | WVJJZZJONDZKRF-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C32H53N7O11/c1-25(40)37(48)21-8-2-5-18-33-26(41)11-13-31(46)38(49)22-9-3-6-19-34-27(42)12-14-32(47)39(50)23-10-4-7-20-35-28(43)17-24-36-29(44)15-16-30(36)45/h15-16,48-50H,2-14,17-24H2,1H3,(H,33,41)(H,34,42)(H,35,43) |
| Chemical Name | N-[5-[acetyl(hydroxy)amino]pentyl]-N'-[5-[[4-[5-[3-(2,5-dioxopyrrol-1-yl)propanoylamino]pentyl-hydroxyamino]-4-oxobutanoyl]amino]pentyl]-N'-hydroxybutanediamide |
| Synonyms | Mal-Deferoxamine; 1638156-31-6; N-(3,11,14,22,25,33-hexaoxo-4,10,15,21,26,32-hexaaza-10,21,32- trihydroxytetratriacontane)maleimide; N4-[5-[[4-[[5-(Acetylhydroxyamino)pentyl]amino]-1,4-dioxobutyl]hydroxyamino]pentyl]-N1-[5-[[3-(2,5-dihydro-2,5-dioxo-1H-pyrrol-1-yl)-1-oxopropyl]amino]pentyl]-N1-hydroxybutanediamide; SCHEMBL20682596; |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Bifunctional chelator; In vivo imaging |
| ln Vitro | Zirconium-89 is an emerging radionuclide for positron emission tomography (PET) especially for biomolecules with slow pharmacokinetics as due to its longer half-life, in comparison to fluorine-18 and gallium-68, imaging at late time points is feasible. Desferrioxamine B (DFO), a linear bifunctional chelator (BFC) is mostly used for this radionuclide so far but shows limitations regarding stability. Our group recently reported on fusarinine C (FSC) with similar zirconium-89 complexing properties but potentially higher stability related to its cyclic structure. This study was designed to compare FSC and DFO head-to-head as bifunctional chelators for 89Zr-radiolabeled EGFR-targeting ZEGFR:2377 affibody bioconjugates [1]. |
| ln Vivo | Fsc-zegfr: 2377 and DFO-ZEGFR:2377 were evaluated regarding radiolabeling, in vitro stability, specificity, cell uptake, receptor affinity, biodistribution, and microPET-CT imaging. Both conjugates were efficiently labeled with zirconium-89 at room temperature but radiochemical yields increased substantially at elevated temperature, 85 °C. Both 89Zr-FSC-ZEGFR:2377 and 89Zr-DFO-ZEGFR:2377 revealed remarkable specificity, affinity and slow cell-line dependent internalization. Radiolabeling at 85 °C showed comparable results in A431 tumor xenografted mice with minor differences regarding blood clearance, tumor and liver uptake. In comparison 89Zr-DFO-ZEGFR:2377, radiolabeled at room temperature, showed a significant difference regarding tumor-to-organ ratios. MicroPET-CT imaging studies of 89Zr-FSC-ZEGFR:2377 as well as 89Zr-DFO-ZEGFR:2377 confirmed these findings. In summary we were able to show that FSC is a suitable alternative to DFO for radiolabeling of biomolecules with zirconium-89. Furthermore, our findings indicate that 89Zr-radiolabeling of DFO conjugates at higher temperature reduces off-chelate binding leading to significantly improved tumor-to-organ ratios and therefore enhancing image contrast [1]. |
| Cell Assay |
In Vitro Studies [1] High level EGFR-expressing A431 human epidermoid carcinoma and MDA468 human breast cancer as well as low level EGFR-expressing DU-145 human prostate cancer cell lines were purchased from American Type Culture Collection. Cells were grown in a humidified atmosphere of 95% air/5% carbon dioxide using RPMI-1640 media supplemented with 10% v/v fetal bovine serum (FBS) and 1% v/v Penicillin-Streptomycin-Glutamin (PSG) solution (= complete media). For binding specificity studies, cells were seeded in 6-well plates (4 × 105 cells/dish for A431 and MDA-468; 1 × 106 cells/dish for DU-145) 3 days before the experiment. On the day of the experiment media was aspirated and all dishes were washed with 2 mL of RPMI-1640 media without additives (= incomplete media). Three sets of dishes were blocked by adding 1 mL complete media containing 0.5 μM blocking agent (cetuximab, His6-ZEGFR:2377 affibody molecule or bevacizumab in 100-fold molar excess over radioligand) while three dishes remained unblocked containing 1 mL complete media and the plate was left for 15 min at RT. Hereafter 1 mL of the radioligand solution (5 nM; ∼ 4 × 105 cpm) was added to all dishes and cells were incubated at 37 °C. Incubation media was removed after 1h followed by a washing step with 2 mL of incomplete media. After addition of 500 μL trypsin solution to each well the plate was kept for 30 min at 37 °C to detach the cells. The cell suspension was diluted to 1 mL by adding 500 μL of incomplete media and after extensive resuspension an aliquot of 10 μL was taken to determine the number of cells/mL via automated cell counter while the rest of each dish was collected in a plastic vial. All dishes were washed with 1 mL incomplete media, the supernatant was added to the plastic vial and samples were measured using an automated gamma-counter. For cellular processing dishes were seeded with A431 and MDA468 cells in a density of 8 × 105 per dish and left to grow overnight. Hereafter, cells were washed with 1 mL incomplete media and 1 mL complete media was added. Finally cells were incubated with 1 mL radioligand solution (5 nM in complete media) at 37 °C for 1, 2, 4, 8, and 24 h in triplicates. At the appointed time, the incubation media was aspirated from the dishes, cells were washed with 2 mL ice-cold incomplete media and the supernatant was discarded. To determine the membrane-bound amount of radiolabeled conjugate cells were treated at RT as follows. First cells were incubated for 15 min with 1 mL acid wash buffer (0.1 M glycine buffer, pH 2.5, containing 4 M urea solution) and after collecting the supernatant this step was repeated with 1 min incubation and acid wash fractions were pooled. After this, cells were rinsed twice with 1.5 mL PBS buffer combining the fractions. Subsequently after PBS buffer collection 1.5 mL of phosphate buffer (pH 8.0) was added to each dish and cells were incubated for 15 min. Phosphate buffer was collected followed by a second basic wash step and phosphate buffer fractions were pooled. To determine the internalized amount of radioligand cells were lysed by adding 1 mL of sodium hydroxide solution (1 M NaOH) and dishes were incubated at 37 °C for at least 45 min. NaOH fractions were collected and the step was repeated without incubation but using a cell scraper. Alkaline fractions were pooled and all fractions were taken for gamma-counter measurement. |
| Animal Protocol |
In Vivo Studies [1] Biodistribution and imaging studies in tumor xenograft bearing female outbred BALB/c nu/nu mice were conducted under xylazine/ketamine anesthesia to minimize animal suffering and number of animals was reduced as much as possible. For tumor xenograft preparation, 1 × 107 A431 cells were injected subcutaneously to the right hind leg and the tumors were grown for 2 weeks. For biodistribution experiments mice were randomized into five groups with four mice. Only one group was pretreated by subcutaneous injection of 10 mg cetuximab per mouse 24 h before the experiment to determine in vivo specificity of 89Zr-FSC-ZEGFR:2377 as specificity of 89Zr-DFO-ZEGFR:2377 has been shown before Zirconium-89 labeled bioconjugates were injected to mice via tail vain (40 kBq in 100 μL PBS per mouse) and the tracer dose was adjusted to 38 μg per mouse with nonlabeled bioconjugate. Distribution of radioactivity among blood and organs was measured 3 and 24 h post injection (p.i). Whole body positron emission tomography (PET)/computed tomography (CT) imaging was performed using Triumph trimodality system at 3 and 24 h p.i. The animals (n = 2 per time point) were injected with 4 MBq of zirconium-89 labeled bioconjugate (dose adjustment with nonlabeled tracer to 38 μg per mouse) and sacrificed by CO2 asphyxiation immediately before being placed in the camera. The CT scans were performed at the following parameters: 80 mm field of view (FOV); 1.48 magnification; one frame and 512 projections for 2.13 min followed by PET scans conducted for 1 h. CT raw files were reconstructed by filter back projection (FBP). PET data were reconstructed using OSEM-3D (20 iterations). CT data were used for scatter and attenuation correction. PET and CT files were fused and analyzed using PMOD v3.510. Coronal and sagittal PET-CT images are presented as maximum intensity projections (MIP) in RGB color scale. |
| References |
[1]. Cyclic versus Noncyclic Chelating Scaffold for 89Zr-Labeled ZEGFR:2377 Affibody Bioconjugates Targeting Epidermal Growth Factor Receptor Overexpression. Mol Pharm. 2018;15(1):175-185. |
| Additional Infomation | The results of this study showed that FSC is a suitable alternative to DFO as chelating scaffold for EGFR-targeting affibody molecules radiolabeled with zirconium-89. Furthermore, our findings indicate that radiolabeling at elevated temperature increases complex stability and tumor-to-organ ratios resulting in considerably improved imaging properties of 89Zr-DFO-bioconjugates. [1] |
Solubility Data
| Solubility (In Vitro) | May dissolve in DMSO (in most cases), if not, try other solvents such as H2O, Ethanol, or DMF with a minute amount of products to avoid loss of samples |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |