Physicochemical Properties
| Molecular Formula | C20H24NO4+ |
| Molecular Weight | 342.4145 |
| Exact Mass | 342.169 |
| Elemental Analysis | C, 70.15; H, 7.07; N, 4.09; O, 18.69 |
| CAS # | 2141-09-5 |
| Related CAS # | (+)-Magnoflorine iodide;4277-43-4;(+)-Magnoflorine chloride;6681-18-1 |
| PubChem CID | 73337 |
| Appearance | Solid powder |
| Melting Point | 252ºC |
| LogP | -1.71 |
| Hydrogen Bond Donor Count | 2 |
| Hydrogen Bond Acceptor Count | 4 |
| Rotatable Bond Count | 2 |
| Heavy Atom Count | 25 |
| Complexity | 498 |
| Defined Atom Stereocenter Count | 1 |
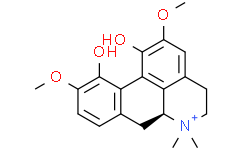
| SMILES | O([H])C1=C(C([H])=C2C([H])([H])C([H])([H])[N+](C([H])([H])[H])(C([H])([H])[H])[C@@]3([H])C([H])([H])C4C([H])=C([H])C(=C(C=4C1=C32)O[H])OC([H])([H])[H])OC([H])([H])[H] |
| InChi Key | YLRXAIKMLINXQY-ZDUSSCGKSA-O |
| InChi Code | InChI=1S/C20H23NO4/c1-21(2)8-7-12-10-15(25-4)20(23)18-16(12)13(21)9-11-5-6-14(24-3)19(22)17(11)18/h5-6,10,13H,7-9H2,1-4H3,(H-,22,23)/p+1/t13-/m0/s1 |
| Chemical Name | (6aS)-2,10-dimethoxy-6,6-dimethyl-5,6,6a,7-tetrahydro-4H-dibenzo[de,g]quinolin-6-ium-1,11-diol |
| Synonyms | α-Magnoflorine; (+)-Magnoflorine; Magnoflorine |
| HS Tariff Code | 2934.99.03.00 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
- In antifungal activity studies, Magnoflorine exhibits inhibitory effects on Candida species (including Candida albicans, Candida tropicalis, Candida krusei), but no specific molecular target (e.g., enzyme/receptor) or affinity data (IC50/Ki) were reported[1] - In pro-inflammatory response regulation, Magnoflorine acts via the MyD88-dependent signaling pathway (involved in TLR4-mediated inflammation), but no direct binding affinity to MyD88 or downstream proteins (e.g., NF-κB, MAPK) was provided[2] |
| ln Vitro |
- Antifungal activity against Candida strains:
- Magnoflorine inhibited the growth of 8 clinical isolates of Candida albicans, with minimum inhibitory concentrations (MICs) ranging from 16 to 64 μg/mL; it also suppressed Candida tropicalis (MIC: 32 μg/mL) and Candida krusei (MIC: 64 μg/mL) growth[1] - At 2×MIC concentration, Magnoflorine reduced the biomass of Candida albicans biofilms by 45% (measured via crystal violet staining) and decreased the viability of biofilm-embedded cells by 38% (XTT reduction assay)[1] - Pro-inflammatory effects in U937 macrophages: - In LPS (1 μg/mL)-activated U937 macrophages, Magnoflorine (10-50 μM) increased TNF-α secretion in a dose-dependent manner: 50 μM Magnoflorine elevated TNF-α levels by 2.8-fold compared to LPS alone (ELISA)[2] - It also upregulated IL-6 (2.1-fold) and IL-1β (1.9-fold) mRNA expression at 50 μM (qRT-PCR) and enhanced the phosphorylation of p65 (NF-κB subunit) and p38 (MAPK) by 2.3-fold and 1.7-fold, respectively (Western blot)[2] - Silencing MyD88 via siRNA abolished the pro-inflammatory effects of Magnoflorine: TNF-α secretion was reduced by 65% compared to non-silenced cells[2] |
| Enzyme Assay |
- Candida growth inhibition assay (MIC determination):
- Candida strains were cultured in RPMI 1640 medium to logarithmic phase, then adjusted to 1×10⁶ CFU/mL.
- Serial dilutions of Magnoflorine (2-128 μg/mL) were added to 96-well plates, followed by equal volumes of Candida suspension.
- Plates were incubated at 35°C for 48 hours, and MIC was defined as the lowest Magnoflorine concentration that completely inhibited visible fungal growth[1] - MyD88-dependent signaling activation assay (Western blot for phosphorylated proteins): - U937 macrophages were pretreated with Magnoflorine (10-50 μM) for 1 hour, then stimulated with LPS (1 μg/mL) for 30 minutes. - Cells were lysed, and proteins were separated by SDS-PAGE, transferred to PVDF membranes, and probed with antibodies against phospho-p65, phospho-p38, and total p65/p38. GAPDH was used as a loading control[2] |
| Cell Assay |
- Candida biofilm inhibition assay:
- Candida albicans was inoculated into 96-well plates and cultured at 35°C for 24 hours to form biofilms.
- Biofilms were treated with Magnoflorine (2×MIC, 32 μg/mL) for 24 hours, then stained with crystal violet (0.1%) for 15 minutes.
- Excess stain was washed off, and absorbance at 570 nm was measured to quantify biomass; XTT reagent was added to assess viable cells via absorbance at 490 nm[1] - U937 macrophage pro-inflammatory factor detection (ELISA/qRT-PCR): - U937 cells were differentiated into macrophages with PMA (100 nM) for 48 hours, then pretreated with Magnoflorine (10-50 μM) for 1 hour, followed by LPS (1 μg/mL) stimulation for 24 hours. - Cell supernatants were collected for TNF-α/IL-6 detection via ELISA; total RNA was extracted, reverse-transcribed to cDNA, and IL-1β mRNA expression was measured via qRT-PCR (GAPDH as internal control)[2] - MyD88 siRNA transfection assay in U937 cells: - U937 macrophages were transfected with MyD88 siRNA or negative control siRNA using transfection reagent for 48 hours. - Transfected cells were treated with Magnoflorine (50 μM) and LPS (1 μg/mL) for 24 hours, then TNF-α secretion was detected via ELISA to verify MyD88 dependence[2] |
| Toxicity/Toxicokinetics |
- In U937 macrophages, Magnoflorine (up to 50 μM) did not affect cell viability (MTT assay: >90% viability compared to control), indicating low cytotoxicity at pro-inflammatory active concentrations[2] |
| References |
[1]. Antifungal activity of magnoflorine against Candida strains. World J Microbiol Biotechnol. 2018 Oct 31;34(11):167. [2]. Magnoflorine Enhances LPS-Activated Pro-Inflammatory Responses via MyD88-Dependent Pathways in U937 Macrophages. Planta Med. 2018 Nov;84(17):1255-1264. |
| Additional Infomation |
- Magnoflorine is a natural aporphine alkaloid primarily isolated from plants of the Magnoliaceae family (e.g., Magnolia officinalis), with potential natural product-based antifungal and immunomodulatory activities[1] [2] - The antifungal mechanism of Magnoflorine may involve disruption of Candida cell membrane integrity (suggested by increased membrane permeability in preliminary assays), but no direct evidence (e.g., ergosterol content measurement) was provided[1] - In LPS-activated macrophages, Magnoflorine’s pro-inflammatory effect is TLR4/MyD88-dependent, as it does not enhance inflammation in cells treated with TLR4 inhibitor (TAK-242)[2] |
Solubility Data
| Solubility (In Vitro) | DMSO: ~10 mg/mL (~29.2 mM) |
| Solubility (In Vivo) |
Note: Listed below are some common formulations that may be used to formulate products with low water solubility (e.g. < 1 mg/mL), you may test these formulations using a minute amount of products to avoid loss of samples. Injection Formulations (e.g. IP/IV/IM/SC) Injection Formulation 1: DMSO : Tween 80: Saline = 10 : 5 : 85 (i.e. 100 μL DMSO stock solution → 50 μL Tween 80 → 850 μL Saline) *Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH ₂ O to obtain a clear solution. Injection Formulation 2: DMSO : PEG300 :Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL DMSO → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Injection Formulation 3: DMSO : Corn oil = 10 : 90 (i.e. 100 μL DMSO → 900 μL Corn oil) Example: Take the Injection Formulation 3 (DMSO : Corn oil = 10 : 90) as an example, if 1 mL of 2.5 mg/mL working solution is to be prepared, you can take 100 μL 25 mg/mL DMSO stock solution and add to 900 μL corn oil, mix well to obtain a clear or suspension solution (2.5 mg/mL, ready for use in animals). Injection Formulation 4: DMSO : 20% SBE-β-CD in saline = 10 : 90 [i.e. 100 μL DMSO → 900 μL (20% SBE-β-CD in saline)] *Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Injection Formulation 5: 2-Hydroxypropyl-β-cyclodextrin : Saline = 50 : 50 (i.e. 500 μL 2-Hydroxypropyl-β-cyclodextrin → 500 μL Saline) Injection Formulation 6: DMSO : PEG300 : castor oil : Saline = 5 : 10 : 20 : 65 (i.e. 50 μL DMSO → 100 μLPEG300 → 200 μL castor oil → 650 μL Saline) Injection Formulation 7: Ethanol : Cremophor : Saline = 10: 10 : 80 (i.e. 100 μL Ethanol → 100 μL Cremophor → 800 μL Saline) Injection Formulation 8: Dissolve in Cremophor/Ethanol (50 : 50), then diluted by Saline Injection Formulation 9: EtOH : Corn oil = 10 : 90 (i.e. 100 μL EtOH → 900 μL Corn oil) Injection Formulation 10: EtOH : PEG300:Tween 80 : Saline = 10 : 40 : 5 : 45 (i.e. 100 μL EtOH → 400 μLPEG300 → 50 μL Tween 80 → 450 μL Saline) Oral Formulations Oral Formulation 1: Suspend in 0.5% CMC Na (carboxymethylcellulose sodium) Oral Formulation 2: Suspend in 0.5% Carboxymethyl cellulose Example: Take the Oral Formulation 1 (Suspend in 0.5% CMC Na) as an example, if 100 mL of 2.5 mg/mL working solution is to be prepared, you can first prepare 0.5% CMC Na solution by measuring 0.5 g CMC Na and dissolve it in 100 mL ddH2O to obtain a clear solution; then add 250 mg of the product to 100 mL 0.5% CMC Na solution, to make the suspension solution (2.5 mg/mL, ready for use in animals). Oral Formulation 3: Dissolved in PEG400 Oral Formulation 4: Suspend in 0.2% Carboxymethyl cellulose Oral Formulation 5: Dissolve in 0.25% Tween 80 and 0.5% Carboxymethyl cellulose Oral Formulation 6: Mixing with food powders Note: Please be aware that the above formulations are for reference only. InvivoChem strongly recommends customers to read literature methods/protocols carefully before determining which formulation you should use for in vivo studies, as different compounds have different solubility properties and have to be formulated differently. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.9205 mL | 14.6024 mL | 29.2048 mL | |
| 5 mM | 0.5841 mL | 2.9205 mL | 5.8410 mL | |
| 10 mM | 0.2920 mL | 1.4602 mL | 2.9205 mL |