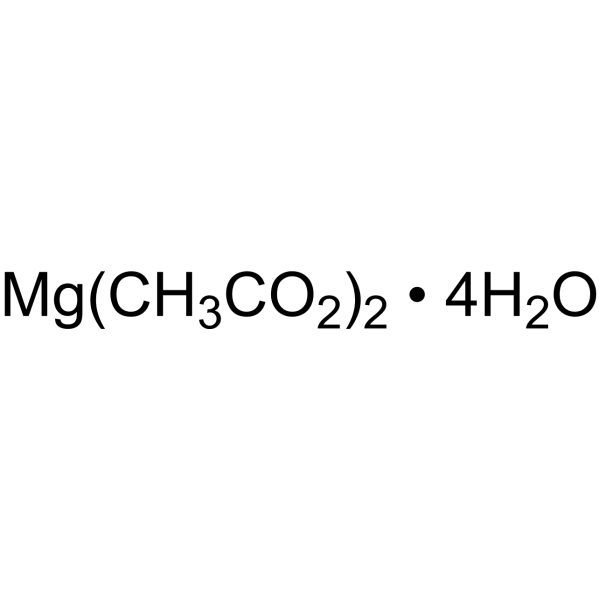Physicochemical Properties
| Molecular Formula | C4H14MGO8 |
| Molecular Weight | 214.4542 |
| Exact Mass | 214.054 |
| CAS # | 16674-78-5 |
| Related CAS # | Acetic acid potassium 99%;127-08-2;Acetic acid;64-19-7;Acetic acid lead;546-67-8 |
| PubChem CID | 134717 |
| Appearance | Typically exists as solid at room temperature |
| Density | 1.454 |
| Boiling Point | 117.1ºC at 760 mmHg |
| Melting Point | 72-75 °C(lit.) |
| Vapour Pressure | 13.9mmHg at 25°C |
| Index of Refraction | n20/D 1.358 |
| Hydrogen Bond Donor Count | 4 |
| Hydrogen Bond Acceptor Count | 8 |
| Rotatable Bond Count | 0 |
| Heavy Atom Count | 13 |
| Complexity | 25.5 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | [Mg+2].[O-]C(C([H])([H])[H])=O.[O-]C(C([H])([H])[H])=O.O([H])[H].O([H])[H].O([H])[H].O([H])[H] |
| InChi Key | XKPKPGCRSHFTKM-UHFFFAOYSA-L |
| InChi Code | InChI=1S/2C2H4O2.Mg.4H2O/c2*1-2(3)4;;;;;/h2*1H3,(H,3,4);;4*1H2/q;;+2;;;;/p-2 |
| Chemical Name | magnesium;diacetate;tetrahydrate |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| ADME/Pharmacokinetics |
Absorption, Distribution and Excretion Intestinal absorption is achieved mainly through passive diffusion. Mainly renal exctretion, where up to 97% of magnesium may be excreted renally during hypermagnesemia. Magnesium ions display approximate volume of distribution of 0.2 to 0.4 L/kg Biological Half-Life Elimination half-life has been reported to be 27.7 hours following an overdose of 400mEq magnesium in an adult. |
| Toxicity/Toxicokinetics |
Protein Binding Protein binding of magnesium ions is about 30% |
| References |
[1]. Thermal dehydration of magnesium acetate tetrahydrate: formation and in situ crystallization of anhydrous glass. J Phys Chem B. 2012 Dec 13;116(49):14477-86. |
| Additional Infomation |
Magnesium acetate tetrahydrate is a hydrated form of anhydrous magnesium acetate salt with the chemical formula of Mg(CH3COO)2 • 4H2O. As a salt form of magnesium, magnesium acetate is one of the bioavailable forms of magnesium and forms a very water soluble compound. Magnesium is an essential element and second most abundant cation in the body that plays a key role in maintaining normal cellular function such as production of ATP and efficient enzyme activity. Magnesium acetate tetrahydrate can be used as an electrolyte supplementation or a reagent in molecular biology experiments. Magnesium Acetate Tetrahydrate is the hydrated acetate salt form of magnesium. Magnesium is a divalent cation essential for a number of biochemical processes involved in nerve signaling, bone mineralization and muscle contractions. About 350 enzymes involved in glycolysis and the Krebs cycle, formation of cyclic-AMP and ATP, cellular signal transduction and protein and nucleic acid synthesis are dependent on magnesium. See also: Magnesium Cation (has active moiety) ... View More ... Drug Indication Used as magnesium salf-containing laxatives to prevent constipation. It can bring synergistic effect to restore normal bowel function when using in combination with aluminum salts that induce bowel retention. Magnesium acetate tetrahydrate is used as a source of water and electrolytes when combined with dextrose and other salts to form intravenous infusions. This injection can be used for patients with carbohydrate or magnesium deficiency, insulin hypoglycemia, constipation or hypertension during pregnancy. Mechanism of Action Magnesium ions electrostatically stabilize the adenylyl cyclase complex and enhance its catalytic actions and production of cAMP. They also regulate the level of phosphorylation in various pathways by formation of transition state of phosphoryl transfer reaction by protein kinases and stabilize ATP binding to protein kinases via electrostatic interactions. Many metabolic enzymes involved in glycolysis and Krebs cycle are magnesium-dependent. Magnesium-containing laxatives cause diarrhea through water retention and increased fecal mass that stimulates peristalsis. When used as an electrolyte supplementation, magnesium acetate tetrahydrate induces diuresis and metabolic alkalinizing effect. Magnesium ions enhance reactivity of arteries to vasoconstrictors, promotes vasoconstriction, and increases peripheral resistance, leading to increased blood pressure through potential competition with calcium ions in the vascular system. Magnesium ions also regulate other ions entering and exiting the cell membrane by acting as a ligand in N-methyl-D-aspartate receptor. Pharmacodynamics Magnesium is an essential cofactor for many enzymatic reactions such as protein synthesis and ATP production. It also participates in adenylyl cyclase pathway and tyrosine kinase signalling pathways. Magnesium may also play a role in regulating glucose metabolism. It serves as an essential cation for a number of biochemical processes involved in nerve signaling, bone mineralization and muscle contractions. |
Solubility Data
| Solubility (In Vitro) | DMSO : ~100 mg/mL (~466.31 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (11.66 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 2.5 mg/mL (11.66 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 3: ≥ 2.5 mg/mL (11.66 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 4.6631 mL | 23.3155 mL | 46.6309 mL | |
| 5 mM | 0.9326 mL | 4.6631 mL | 9.3262 mL | |
| 10 mM | 0.4663 mL | 2.3315 mL | 4.6631 mL |