ML239 was originally identified as a potent and selective inhibitor of breast cancer stem cells with an IC50 of 1.16 μM, but recent studies (2016 Nature 12(2):109-16.) suggested that it most likely acts through activation of fatty acid desaturase 2 (FADS2). ML239 was discovered from high-throughput screen (HTS) with the National Institute of Health–Molecular Libraries Small Molecule Repository (NIH–MLSMR) compound collection which identified a class of acyl hydrazones to be selectively lethal to breast cancer stem cell (CSC) enriched populations. Medicinal chemistry efforts were undertaken to optimize potency and selectivity of this class of compounds. The optimized compound was declared as a probe (ML239) with the NIH Molecular Libraries Program and displayed greater than 20-fold selective inhibition of the breast CSC-like cell line (HMLE_sh_Ecad) over the isogenic control line (HMLE_sh_GFP).
Physicochemical Properties
| Molecular Formula | C₁₃H₁₀CL₃N₃O₂ | |
| Molecular Weight | 346.6 | |
| Exact Mass | 344.983 | |
| Elemental Analysis | C, 45.05; H, 2.91; Cl, 30.68; N, 12.12; O, 9.23 | |
| CAS # | 1378872-36-6 | |
| Related CAS # |
|
|
| PubChem CID | 135999136 | |
| Appearance | Solid powder | |
| Density | 1.5±0.1 g/cm3 | |
| Index of Refraction | 1.634 | |
| LogP | 3.57 | |
| Hydrogen Bond Donor Count | 2 | |
| Hydrogen Bond Acceptor Count | 3 | |
| Rotatable Bond Count | 5 | |
| Heavy Atom Count | 21 | |
| Complexity | 372 | |
| Defined Atom Stereocenter Count | 0 | |
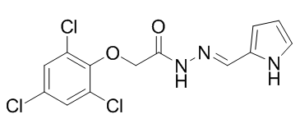
| SMILES | O=C(N/N=C/C1=CC=CN1)COC2=C(Cl)C=C(Cl)C=C2Cl |
|
| InChi Key | NVEDPFICKAIHKD-NGYBGAFCSA-N | |
| InChi Code | InChI=1S/C13H10Cl3N3O2/c14-8-4-10(15)13(11(16)5-8)21-7-12(20)19-18-6-9-2-1-3-17-9/h1-6,17H,7H2,(H,19,20)/b18-6+ | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets |
Fatty acid desaturase 2 (FADS2) [2] |
||
| ln Vitro |
With an IC50 of 1.16 μM and approximately 24-fold selectivity against the control cell line, ML239 (Compound 7j) is a strong and selective inhibitor of breast cancer stem cells[1]. It is highly probable that ML239 suppresses breast cancer stem-like cells by activating fatty acid desaturase 2 (FADS2). NCIH661 cells are cytotoxic to ML239; cytotoxicity is reduced by FADS2 knockdown, and cytotoxicity is further reduced in cancer cell lines (CCLs) by the FADS2 inhibitor SC-26196[2]. Exhibited selective cytotoxicity to breast cancer stem cell (CSC)-enriched populations. The optimized ML239 showed greater than 20-fold selective inhibition of the breast CSC-like cell line (HMLE_sh_Ecad) compared to the isogenic control line (HMLE_sh_GFP) [1] - Most likely acts through activation of FADS2. In NCIH661 large cell lung carcinoma (LCLC) cells, FADS2 knockdown reduced the cytotoxicity of ML239 [2] - Treatment of NCIH661 cells with 2 μM ML239 for 24 hours altered the levels of 183 cellular lipids. Co-treatment with ML239 (2 μM) and SC-26196 (a FADS2 inhibitor) resulted in significant changes in lipid species compared to single-agent treatment (p < 0.001, two-way ANOVA with Bonferroni correction) [2] |
||
| ln Vivo |
|
||
| Cell Assay |
Selective cytotoxicity assay in breast CSC-like cells and control cells: Breast CSC-like cell line (HMLE_sh_Ecad) and isogenic control line (HMLE_sh_GFP) were treated with ML239 at various concentrations. Cell viability was evaluated to determine the selective inhibitory effect of ML239, and the selectivity ratio (inhibition of HMLE_sh_Ecad vs. HMLE_sh_GFP) was calculated to confirm greater than 20-fold selectivity [1] - FADS2 knockdown and cytotoxicity assay in NCIH661 cells: NCIH661 cells were transfected with FADS2 knockdown constructs or control constructs. After successful knockdown of FADS2, the cells were treated with different concentrations of ML239. Cell viability was measured to assess the impact of FADS2 knockdown on ML239 cytotoxicity, with each point representing the mean of n = 2 independent experiments [2] - Lipid profiling assay in NCIH661 cells: NCIH661 cells were treated with 2 μM ML239 alone, 2 μM SC-26196 alone, or the combination of both for 24 hours. DMSO was used as a vehicle control. Levels of 183 cellular lipids were detected and quantified. Statistical analysis (two-way ANOVA with Bonferroni correction) was performed to identify significantly changed lipid species, with n=9 for DMSO, ML239, and SC-26196 groups, and n=3 for the co-treatment group [2] - Gene expression and sensitivity correlation assay: Basal transcript levels (∼19,000 genes) across 823 human cancer cell lines were correlated with the sensitivity patterns of ML239 (expressed as areas under concentration-response curves, AUCs). Z-scored Pearson correlation coefficients were calculated to identify selective outlier transcripts, and FADS2 was found to be correlated with ML239 sensitivity [2] |
||
| Animal Protocol |
|
||
| References |
[1]. Identification of a selective small molecule inhibitor of breast cancer stem cells. Bioorg Med Chem Lett. 2012 May 15;22(10):3571-4. [2]. Correlating chemical sensitivity and basal gene expression reveals mechanism of action. Nat Chem Biol. 2016 Feb;12(2):109-16. |
||
| Additional Infomation |
N'-(2-pyrrolylidenemethyl)-2-(2,4,6-trichlorophenoxy)acetohydrazide is an aromatic ether. ML239 is a small-molecule inhibitor belonging to the acyl hydrazone class [1] - It was identified through a high-throughput screen (HTS) with the National Institute of Health-Molecular Libraries Small Molecule Repository (NIH-MLSMR) compound collection, based on its selective lethality to breast cancer stem cell (CSC)-enriched populations [1] - Medicinal chemistry optimization was performed to enhance the potency and selectivity of the acyl hydrazone class of compounds, leading to the development of ML239, which was declared as a probe with the NIH Molecular Libraries Program [1] - The mechanism of action of ML239 was inferred through correlating its sensitivity patterns with basal gene expression across 823 human cancer cell lines, revealing a correlation with FADS2 expression and confirming FADS2 as a key mediator of its cytotoxicity [2] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 2.5 mg/mL (7.21 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 25.0 mg/mL clear DMSO stock solution to 900 μL of corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.8852 mL | 14.4259 mL | 28.8517 mL | |
| 5 mM | 0.5770 mL | 2.8852 mL | 5.7703 mL | |
| 10 mM | 0.2885 mL | 1.4426 mL | 2.8852 mL |