ML132 (also known as NCGC 00185682) is a potent and selective caspase 1 inhibitor with an IC50 of 0.316 nM. It contains a nitrile-containing propionic acid moiety as an electrophile for covalent attack by the active-site cysteine residue of caspase 1. The syntheses of several cyanopropanate-containing small molecules based on the optimized peptidic scaffold of prodrug VX-765 were accomplished. These compounds were found to be potent inhibitors of caspase 1 (IC(50) values < or =1 nM). Examination of these novel small molecules against a caspase panel demonstrated an impressive degree of selectivity for caspase 1 inhibition over other caspase isozymes. Assessment of hydrolytic stability and selected ADME properties highlighted these agents as potentially useful tools for studying caspase 1 down-regulation in various settings, including in vivo analyses.
Physicochemical Properties
| Molecular Formula | C22H28CLN5O5 | |
| Molecular Weight | 477.941224098206 | |
| Exact Mass | 477.177 | |
| Elemental Analysis | C, 55.29; H, 5.90; Cl, 7.42; N, 14.65; O, 16.74 | |
| CAS # | 1230628-71-3 | |
| Related CAS # |
|
|
| PubChem CID | 44620939 | |
| Appearance | White to light yellow solid powder | |
| LogP | 1.6 | |
| Hydrogen Bond Donor Count | 4 | |
| Hydrogen Bond Acceptor Count | 7 | |
| Rotatable Bond Count | 8 | |
| Heavy Atom Count | 33 | |
| Complexity | 820 | |
| Defined Atom Stereocenter Count | 3 | |
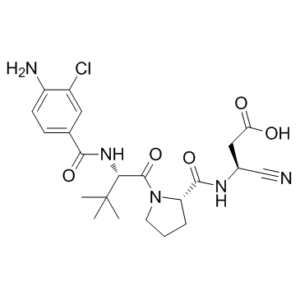
| SMILES | ClC1=C(C=CC(=C1)C(N[C@H](C(N1CCC[C@H]1C(NC[C@@H](C#N)C(=O)O)=O)=O)C(C)(C)C)=O)N |
|
| InChi Key | KENKPOUHXLJLEY-QANKJYHBSA-N | |
| InChi Code | InChI=1S/C22H28ClN5O5/c1-22(2,3)18(27-19(31)12-6-7-15(25)14(23)9-12)21(33)28-8-4-5-16(28)20(32)26-13(11-24)10-17(29)30/h6-7,9,13,16,18H,4-5,8,10,25H2,1-3H3,(H,26,32)(H,27,31)(H,29,30)/t13-,16-,18+/m0/s1 | |
| Chemical Name |
|
|
| Synonyms |
|
|
| HS Tariff Code | 2934.99.9001 | |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month Note: (1). This product requires protection from light (avoid light exposure) during transportation and storage.(2). Please store this product in a sealed and protected environment (e.g. under nitrogen), avoid exposure to moisture. |
|
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Caspase 1 |
| ln Vitro | Researchers examined VRT-043198 (2b), ML132 (NCGC 00185682) (3), NCGC00183434 (4) and the tetrazole NCGC00183681 (16) and the results are displayed in figure 2. VRT-043198 (2b) was confirmed as a potent caspase 1 inhibitor with an IC50 value of 11.5 nM. NCGC00183434 (4) which contains the key cyanopropanoate moiety was found to inhibit caspase 1 with an impressive IC50 value of 0.316 nM. We were further gratified to find that the ethyl ester 3 and tetrazole 16 retained impressive potencies versus caspase 1 (IC50 = 144.7 nM and IC50 = 20.4 nM, respectively). The KI value of 4 was estimated to be 0.4 nM for caspase 1 using a competitive inhibition model [1]. |
| ADME/Pharmacokinetics | Based upon the data provided in this panel, it was clear that these agents represent important new tools for caspase 1 inhibition. However, the contributing functional groups for these agents (i.e. ethyl acetals, aldehydes, nitriles and esters) are all subject to hydrolysis in various conditions. It was paramount to fully understand their stability profile to appreciate their utility as molecular probes or even clinically used agents. Therefore, we examined 1, 2b, 3/ML132 (NCGC 00185682), 4 and 16 within an aqueous degradation study at neutral (pH 7), acidic (pH 2), and basic (pH 8) conditions. The study was conducted by monitoring the degradation of each agent by LCMS analysis at various time points over 96 hours (Figure 3). The prodrug 1 showed moderate degradation in water with over 50% of the compound decomposed after 48 hours. This degradation was amplified in both basic and acidic conditions. Conversely, the active agent 2b was very stable in both neutral and acidic conditions and its degradation at pH 8 was moderate. The potent 4 was exceedingly stable in basic conditions and its stability in neutral and acidic conditions was moderate to good (degradation of 50% in both conditions after 72 hours). The ethyl ester 3/ML132 (NCGC 00185682) was exceptionally stable in neutral and acidic conditions (no degradation noted), however, it was fully degraded in basic conditions after 22 hours (presumably due to saponification of the ester). Finally, the tetrazole 16 was found to be resistant to degradation in all conditions. Interestingly, this data suggests that 1 may have a short half-life as an oral agent due to its instability in acidic conditions such as those found in the gastric environment (40% degradation after 3.5 hours at pH 2). In contrast, this data highly suggests that 3/ML132 (NCGC 00185682) and 16 will be suitable reagents for all manner of examinations (cell based and in vivo studies) and even the highly active 4 will persist beyond 24 hours.[1] |
| References | [1]. A highly potent and selective caspase 1 inhibitor that utilizes a key 3-cyanopropanoic acid moiety. ChemMedChem.2010 May 3;5(5):730-8. |
| Additional Infomation |
(3S)-3-[[[(2S)-1-[(2S)-2-[[(4-amino-3-chlorophenyl)-oxomethyl]amino]-3,3-dimethyl-1-oxobutyl]-2-pyrrolidinyl]-oxomethyl]amino]-3-cyanopropanoic acid is a peptide. Herein, we examine the potential of a nitrile-containing propionic acid moiety as an electrophile for covalent attack by the active-site cysteine residue of caspase 1. The syntheses of several cyanopropanate-containing small molecules based on the optimized peptidic scaffold of prodrug VX-765 were accomplished. These compounds were found to be potent inhibitors of caspase 1 (IC(50) values < or =1 nM). Examination of these novel small molecules against a caspase panel demonstrated an impressive degree of selectivity for caspase 1 inhibition over other caspase isozymes. Assessment of hydrolytic stability and selected ADME properties highlighted these agents as potentially useful tools for studying caspase 1 down-regulation in various settings, including in vivo analyses. [1] |
Solubility Data
| Solubility (In Vitro) |
|
|||
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 7 mg/mL (14.65 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 70.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. Solubility in Formulation 2: ≥ 6.25 mg/mL (13.08 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 62.5 mg/mL clear DMSO stock solution to 400 μL PEG300 and mix evenly; then add 50 μL Tween-80 to the above solution and mix evenly; then add 450 μL normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 3: ≥ 6.25 mg/mL (13.08 mM) (saturation unknown) in 10% DMSO + 90% Corn Oil (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 62.5 mg/mL clear DMSO stock solution to 900 μL corn oil and mix evenly. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.0923 mL | 10.4616 mL | 20.9231 mL | |
| 5 mM | 0.4185 mL | 2.0923 mL | 4.1846 mL | |
| 10 mM | 0.2092 mL | 1.0462 mL | 2.0923 mL |