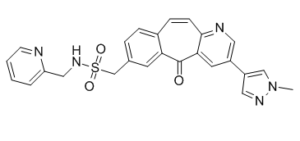
MK-8033 is a novel, potent, selective, ATP competitive small-molecule, dual inhibitor of c-Met/Ron (IC50=1 nM Wt c-Met) under investigation as a treatment for cancer. The goal of developing MK-8033 was to lessen the time-dependent inhibition of CYP3A4 (TDI) that individuals in this structural class exhibit. To obtain MK-8033 and its analogues, a unique two-step protocol for the synthesis of benzylic sulfonamides was created. Crucially, MK-8033 completely inhibits the growth of tumors in a c-Met amplified (GTL-16) subcutaneous tumor xenograft model. This is noteworthy because, unlike c-Met inhibitors that do not preferentially bind to the active kinase conformation, MK-8033 exhibits equal potency against a panel of oncogenic activating mutations of c-Met.
Physicochemical Properties
| Molecular Formula | C25H21N5O3S |
| Molecular Weight | 471.53094 |
| Exact Mass | 471.137 |
| Elemental Analysis | C, 63.68; H, 4.49; N, 14.85; O, 10.18; S, 6.80 |
| CAS # | 1001917-37-8 |
| Related CAS # | MK-8033 hydrochloride;1283000-43-0 |
| PubChem CID | 45142457 |
| Appearance | Light yellow to yellow solid powder |
| Density | 1.39 |
| LogP | 4.635 |
| Hydrogen Bond Donor Count | 1 |
| Hydrogen Bond Acceptor Count | 7 |
| Rotatable Bond Count | 6 |
| Heavy Atom Count | 34 |
| Complexity | 859 |
| Defined Atom Stereocenter Count | 0 |
| SMILES | O=S(NCC1=CC=CC=N1)(CC2=CC=C3C(C(C4=CC(C5=CN(N=C5)C)=CN=C4C=C3)=O)=C2)=O |
| InChi Key | VMJFTOSOFDEKTM-UHFFFAOYSA-N |
| InChi Code | InChI=1S/C25H21N5O3S/c1-30-15-20(13-28-30)19-11-23-24(27-12-19)8-7-18-6-5-17(10-22(18)25(23)31)16-34(32,33)29-14-21-4-2-3-9-26-21/h2-13,15,29H,14,16H2,1H3 |
| Chemical Name | 1-[5-(1-methylpyrazol-4-yl)-2-oxo-7-azatricyclo[9.4.0.03,8]pentadeca-1(11),3(8),4,6,9,12,14-heptaen-14-yl]-N-(pyridin-2-ylmethyl)methanesulfonamide |
| Synonyms | MK-8033; MK8033; MK 8033 |
| HS Tariff Code | 2934.99.9001 |
| Storage |
Powder-20°C 3 years 4°C 2 years In solvent -80°C 6 months -20°C 1 month |
| Shipping Condition | Room temperature (This product is stable at ambient temperature for a few days during ordinary shipping and time spent in Customs) |
Biological Activity
| Targets | Ron (IC50 = 7 nM) |
| ln Vitro |
MK-8033 (Compound 11r, 10 μM) showed a 31% CYP3A4 (cytochrome P450 3A4) inhibition[1]. MK-8033 (1 μM, 2 h) inhibits the c-Met-dependent gastric cancer cell line GTL-16's Y1349 phosphorylation (IC50: 0.03 μM)[1]. MK-8033 (1-10 μM, 72 h) suppresses the growth of GTL-16 cells (IC50: 0.58 μM)[1]. MK-8033 inhibits oncogenic c-Met activation loop mutants with IC50s ranging from 0.6 to 1 nM. It binds more tightly to phosphorylated c-Met (Kd: 3.2 nM) than to its unphosphorylated counterpart (Kd: 10.4 nM).[1] MK-8033 (0.1-10 μM, 2 h) decreases c-Met, ERK, and Akt phosphorylation in EBC-1 and H1993 cells[2]. MK-8033 (1 μM, 1 h) increases the radiation sensitivity of high c-Met-expressing H1993 and EBC-1 cells[2]. MK-8033 (10 μM, 6 h) reduces DNA repair and increases γ-H2Ax levels in comparison to double irradiation in A549 cells[2]. MK-8033 (2 μM, 72 h) causes G-alpha protein mutant UM (uveal melanoma) cells to proliferate less, but induces apoptosis somewhat[3]. |
| ln Vivo |
MK-8033 (Compound 11r, oral administration, 3-100 mg/kg, twice daily for 21 days) inhibits tumor growth in GTL-16 c-Met amplified gastric tumor xenografts[1]. MK-8033 shows good bioavailability (35% for rats, 33% for dogs) and moderate clearance (t1/2: 0.8 h for rats, 3.1 h for dogs)[1]. |
| Cell Assay |
Cell Line: EBC-1, H1993 cells, A549 and H460 cells Concentration: 0.1, 1, 10 μM Incubation Time: 2 h Result: Reduced the phosphorylation of c-Met, ERK, and Akt in EBC-1 and H1993 cells in a dose-dependent manner. |
| Animal Protocol |
Human GTL-16 c-Met amplified gastric tumor xenografts 3, 10, 30, and 100 mg/kg Oral administration, twice daily for 21 days |
| References |
[1]. Discovery of 1-[3-(1-methyl-1H-pyrazol-4-yl)-5-oxo-5H-benzo[4,5]cyclohepta[1,2-b]pyridin-7-yl]-N-(pyridin-2-ylmethyl)methanesulfonamide (MK-8033): A Specific c-Met/Ron dual kinase inhibitor with preferential affinity for the activated state of c-Met. J Med Chem . 2013 Mar 28;56(6):2294-310. [2]. C-Met inhibitor MK-8003 radiosensitizes c-Met-expressing non-small-cell lung cancer cells with radiation-induced c-Met-expression. J Thorac Oncol. 2012 Aug;7(8):1211-7. [3]. Simultaneous inhibition of the HGF/MET and Erk1/2 pathways affect uveal melanoma cell growth and migration. PLoS One. 2014 Feb 13;9(2):e83957. |
| Additional Infomation | c-Met Inhibitor MK8033 is an orally bioavailable inhibitor of c-Met, with potential antineoplastic activity. Upon administration, c-Met inhibitor MK8033 binds to and inhibits the autophosphorylation of the c-Met protein, which disrupts c-Met signal transduction pathways and may induce cell death in tumor cells overexpressing or expressing constitutively activated c-Met protein. In addition, MK8033 inhibits Ron (receptor originated from nantes, MST1R). c-Met protein, which is encoded by the proto-oncogene MET, is a receptor tyrosine kinase also known as hepatocyte growth factor receptor (HGFR); this protein is overexpressed or mutated in many tumor cell types and plays key roles in tumor cell proliferation, survival, invasion, and metastasis, and tumor angiogenesis. Ron, a member of the Met family of cell surface receptor tyrosine kinases, is also overexpressed on certain tumor cell types. |
Solubility Data
| Solubility (In Vitro) | DMSO: ≥ 46 mg/mL (~97.6 mM) |
| Solubility (In Vivo) |
Solubility in Formulation 1: ≥ 1 mg/mL (2.12 mM) (saturation unknown) in 10% DMSO + 40% PEG300 + 5% Tween80 + 45% Saline (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear DMSO stock solution to 400 μL of PEG300 and mix evenly; then add 50 μL of Tween-80 to the above solution and mix evenly; then add 450 μL of normal saline to adjust the volume to 1 mL. Preparation of saline: Dissolve 0.9 g of sodium chloride in 100 mL ddH₂ O to obtain a clear solution. Solubility in Formulation 2: ≥ 1 mg/mL (2.12 mM) (saturation unknown) in 10% DMSO + 90% (20% SBE-β-CD in Saline) (add these co-solvents sequentially from left to right, and one by one), clear solution. For example, if 1 mL of working solution is to be prepared, you can add 100 μL of 10.0 mg/mL clear DMSO stock solution to 900 μL of 20% SBE-β-CD physiological saline solution and mix evenly. Preparation of 20% SBE-β-CD in Saline (4°C,1 week): Dissolve 2 g SBE-β-CD in 10 mL saline to obtain a clear solution. (Please use freshly prepared in vivo formulations for optimal results.) |
| Preparing Stock Solutions | 1 mg | 5 mg | 10 mg | |
| 1 mM | 2.1208 mL | 10.6038 mL | 21.2076 mL | |
| 5 mM | 0.4242 mL | 2.1208 mL | 4.2415 mL | |
| 10 mM | 0.2121 mL | 1.0604 mL | 2.1208 mL |